During onset and recovery of neuromuscular blockade, the action of NMBAs is determined by competitive binding of ACh and NMBAs to the postsynaptic nAChRs at the neuromuscular junctions [
11]. The conventional strategy for neuromuscular blockade reversal involves the use of pharmacological tools to increase ACh levels, thereby increasing binding at postsynaptic nAChRs by inhibiting acetylcholinesterase near postsynaptic nAChRs. In contrast, sugammadex binds and chelates the ŌĆśguestŌĆÖ molecules at a 1 : 1 ratio; thus, sugammadex has no effects on the release and levels of ACh at the neuromuscular junctions, while reversing neuromuscular blockade [
1]. Several receptors mediate and modulate the release of ACh at neuromuscular junctions. In general, ACh release in variable neuronal signal freuqencies is modulated through fine tuning of several receptors at the presynaptic membrane by ACh and adenosine [
12]. The TOF ratio is the most common and valuable measurement during neuromuscular monitoring, which is performed by four stimuli of 2 Hz [
2]. With this low-frequency stimulation, presynaptic muscarinic M
1AChRs and adenosine A
1 receptors are predominant and modulate the release of adequate amounts of ACh [
12,
13]. ACh then transmits the signal to the post synaptic junction via postsynaptic nAChRs, which affects the presynaptic neuronal nAChRs, which in turn have a positive feedback on ACh release. During non-depolarizing NMBA-induced neuromuscular blockade, the TOF fade and TOF ratio are both considered phenomena related to presynaptic neuronal nAChRs [
3,
14,
15]. A recent study reported that these phenomena are related to the postsynaptic receptor type [
15]. Those authors conducted an in-vivo experiment; the TOF fade occurred only under blockade of the postjunctional nAChRs with ╬▒-bungarotoxin (╬▒-BTX) or ╬▒-conotoxin, but not under that of the presynaptic nAChR with specific blockers alone. Moreover, co-administration of ╬▒-BTX or ╬▒-conotoxin during presynaptic nAChR blockade resulted in a prominent TOF fade. Other studies reported that the presynaptic nAChR regulates the amount of ACh per neural stimulus [
15,
16]. Faria et al. [
15] reported that blockade with dihydro-╬▓-erythrodine (Dh╬▓E) was effective in decreasing the level of prejunctional ACh release, which was consistent with the findings from another study using a cell culture model. Decrease of ACh release causes the onset of a neuromuscular blockade, which was slow under blockade of only the postjunctional nAChRs, but accelerated under that of prejunctional AChRs with Dh╬▓E [
17]. Therefore, the postsynaptic and presynaptic actions of NMBAs on nAChRs affect the efficacy of neuromuscular blockade [
18]. Considering these results, the relationship between pre- and postsynaptic receptor function is an important determinant of the TOF fade and the TOF ratio. The conventional strategy for neuromuscular block reversal with anticholinesterase administration is not capable of eliminating NMBAs at the neuromuscular junction, leading to prolonged neuromuscular blocking activities after the initial administration of anticholinesterase. The TOF fade and the TOF ratio are prominent for the duration of NMBA action at the postsynaptic nAChRs. Sugammadex is not able to inactivate neuromuscular blockade at the neuromuscular junction [
1,
7], but causes immediate reduction of the NMBA concentration outside the neuromuscular junction. This causes rapid transfer of NMBAs, due to the concentration gradient across the neuromuscular junction [
1]. Thus, reduction of NMBA concentrations and their activities at the neuromuscular junction attenuates the TOF fade and increases the TOF ratio, which explains the results of our
ex-vivo experiment. We sorted the six groups into two categories to enable clear description and easy understanding for readers: a high-dose group, with sugammadex at Ōēź 2 times the equimolar dose of rocuronium; and a low-dose group, with sugammadex at the level of or less than the equimolar dose of rocuronium. In the high-dose groups, a rapid drop of rocuronium concentration in the Krebs buffer solution allowed rapid exit of rocuronium molecules from the neuromuscular junction, and thereby, achieved attenuation and disappearance of TOF fade even in the early recovery period. In the low-dose group, rocuronium molecules remained at the neuromuscular junction under sugammadex, and the TOF fade and the TOF ratio were maintained until rocuronium, was adequately decreased, due to the concentration gradient across the neuromuscular junction. We obtained similar results in the low-dose groups to that of spontaneous recovery. In
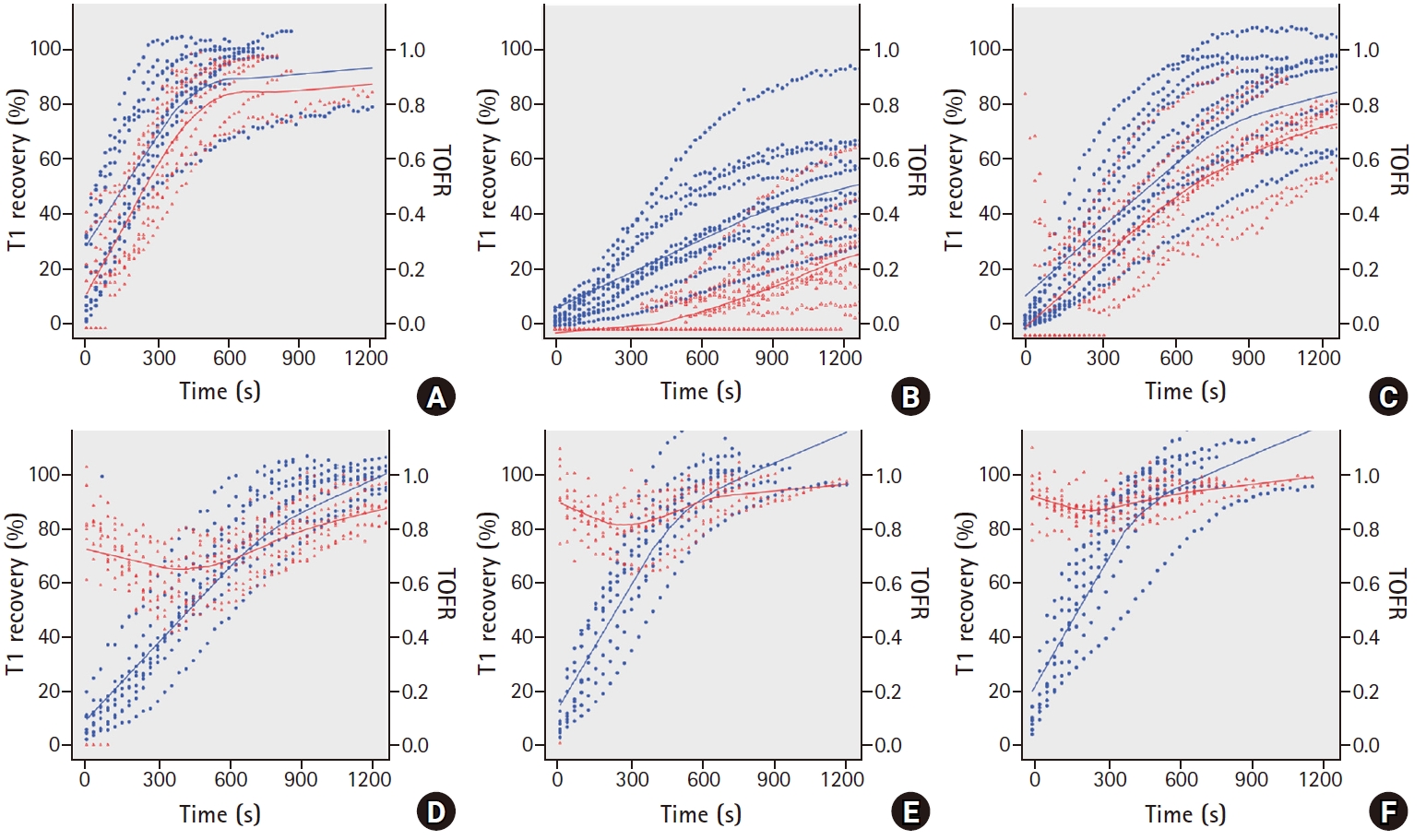
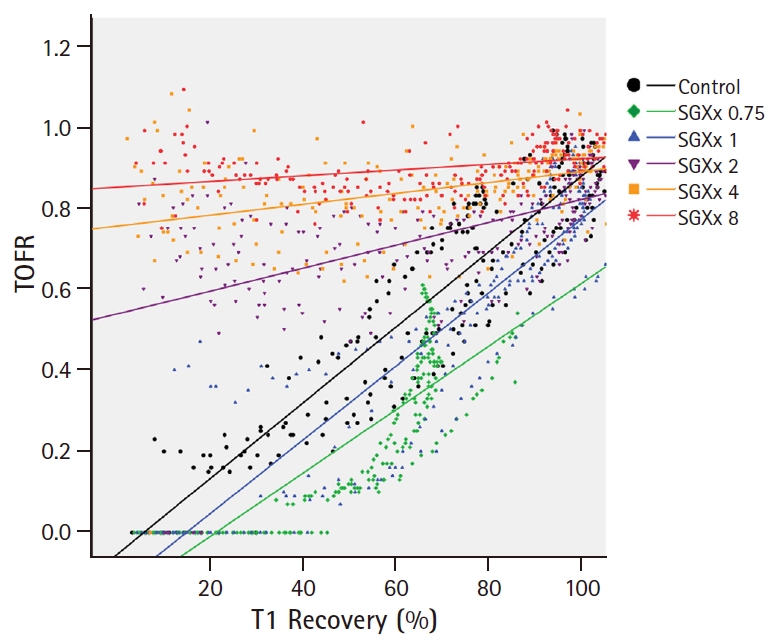
Fig. 3A, as the TOF ratio recovered in parallel with T1 recovery and showed prominent TOF fade in the low-dose group, y-axis values (TOF ratio) often start low and converge at 1. In contrast, TOF fade was attenuated and the TOF ratio was high even in the early recovery of T1 in the high-dose group. As such, the slopes of regression curves, ╬®, of low-dose groups were steeper than those of high-dose groups. In
Fig. 4, we demonstrated the simultaneous progression of % T1 recovery and the TOF ratio by time. We generated one value (TOFR/T1 product) by using T1 recovery and the TOF ratio, which were converted by % value. In the low dose group, as the T1 recovery preceded the TOF ratio, TOFR/T1 was Ōēż 1. As such, it was well-represented by the equation of y = ╬╗x. In contrast, those in the high dose group were Ōēź 1 and showed a decay pattern, because TOF fade was attenuated and the TOF ratio was higher than T1 recovery.
Our study has several limitations. First, we conducted an
ex-vivo experiment and disregarded the pharmacokinetic component of rocuronium action, since the phrenic nerve-hemidiaphragm tissue specimens were examined in an organ bath filled with Krebs buffer solution. The overall recovery time to > 95% T1 was > 30 minutes in the low-dose group and was shortened to < 15 minutes in the high-dose group. In clinical settings, however, the recovery time is Ōēż 5 minutes [
19,
20] considering the dose of sugammadex used. Reports have indicated that administration of 2 mg/kg sugammadex for moderate neuromuscular blockade and 4 mg/kg for deep neuromuscular blockade achieved recovery times of Ōēż 3 and 5 minutes to a TOF ratio of > 0.9, respectively [
20ŌĆō
22]. The discrepancies of recovery patterns between the
in-vivo and
ex-vivo approaches suggest that the results should be interpreted differently considering that NMBA-induced neuromuscular blockade is fully and rapidly recovered in clinical settings, which might hinder our results make the blockade disappeared without notice. As such, although the postoperative residual block is still a problem, even in the new era of sugammadex-induced recovery from a neuromuscular block [
23,
24], we should cautiously judge the clinical implications of the findings of the current
ex-vivo study. Second, this study focused on the nicotinic AChR subtype at the presynaptic and postsynaptic junctions. Sugammadex has no action at the neuromuscular junction, and rocuronium has no action on the other receptors at the neuromuscular junction; this study only focused on the nAChRs, which are the primary action site of ACh and NMBAs during neuromuscular blockade. However, as we described above, several receptors modulate ACh release in different environments [
12,
13], and we tried to maintain a consistent environment and neural stimulation throughout the study period. We used TOF stimulation of four stimuli of 2 Hz supramaximal stimulation, which is the same mode used for clinical neuromuscular monitoring. The method of obtaining the phrenic nerve-hemidiaphragm tissue specimen had the disadvantage of temporary hypoxia and damage to the tissue specimen during preparation due to thorax extraction from the rat. To minimize these drawbacks, we made attempts to oxygenate the rat, to remove the thorax immediately after the aorta was cut, and then performed trimming of the specimen in a petri dish containing Krebs buffer solution aerated with a mixture of 95% O
2 and 5% CO
2.
In conclusion, a high dose of sugammadex rapidly reversed the neuromuscular block induced by rocuronium. However, the recovery pattern of the TOF ratio differ according to the dose of sugammadex, particularly when the amount of sugammadex is high enough compared to that of rocuronium used. In that condition, a high TOF ratio may be achieved even without full recovery of the T1 twitch tension. The TOF ratio alone might be insufficient to indicate full recovery of neuromuscular blockade without full recovery of the T1 twitch. Therefore, clinicians should use an appropriate dose of sugammadex and wait for full recovery of both the TOF ratio and T1 twitch.