|
 |
| Korean J Anesthesiol > Volume 76(2); 2023 > Article |
|
Abstract
Background
Methods
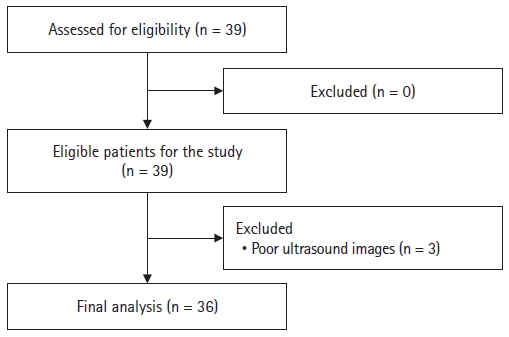
Results
Conclusions
NOTES
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author Contributions
Hyun-Jung Shin (Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Validation; Writing ŌĆō original draft; Writing ŌĆō review & editing)
Heeyeon Kim (Investigation; Methodology)
Sung-Hee Han (Conceptualization; Investigation; Methodology)
Sang-Hwan Do (Conceptualization; Methodology)
Hyo-Seok Na (Conceptualization; Data curation; Supervision; Writing ŌĆō review & editing)
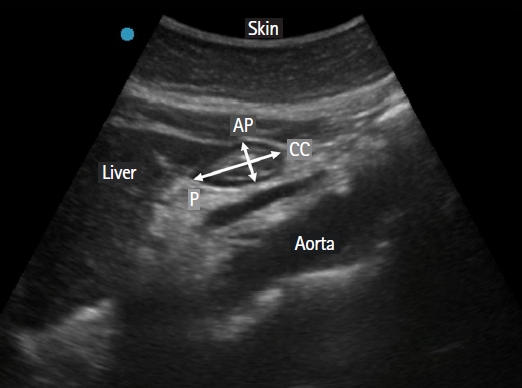
Fig.┬Ā1.
Table┬Ā1.
Table┬Ā2.
Values are presented as mean ┬▒ SD, median (Q1, Q3), or number of the patients (%). TKA: total knee arthroplasty, CSA: cross-sectional area, RLD: right-lateral decubitus, GV: gastric volume. GVs were presented in calculated total volume and in unit volume per body weight, respectively. Paired t-test for CSA in a semi-recumbent position. Wilcoxon signed rank test for CSA in RLD and GV. McNemar-Bowker test for the comparison of qualitative assessment of GV.
Table┬Ā3.
| n = 36 | |
|---|---|
| Tramadol (mg) | 522.2 ┬▒ 434.9 |
| MED of tramadol (mg) | 52.2 ┬▒ 43.5 |
| Morphine (mg) | 1.7 ┬▒ 4.6 |
| Total MED (mg) | 53.9 ┬▒ 42.8 |