|
 |
| Korean J Anesthesiol > Volume 75(2); 2022 > Article |
|
Abstract
Background
Subanesthetic intravenous (IV) ketamine acts as an analgesic and has opioid-sparing effects, particularly for acute postoperative pain; however, its effectiveness in children is understudied. The primary aim of this study was to evaluate the non-inferiority of subanesthetic IV ketamine vs. caudal bupivacaine for postoperative analgesia in children undergoing infra-umbilical surgery.
Methods
Children aged < 6 years were enrolled in this single-blind study and randomized to receive either subanesthetic IV ketamine (0.3 mg/kg) or caudal 0.125% bupivacaine (1 ml/kg) along with general anesthesia. Postoperative pain was assessed using the FLACC scale at 30 minutes and 1, 2, 3, and 6 h post-operation. Intra- and postoperative opioid consumption, time to extubation, postoperative vomiting, agitation, sedation, and inflammatory markers were also assessed.
Results
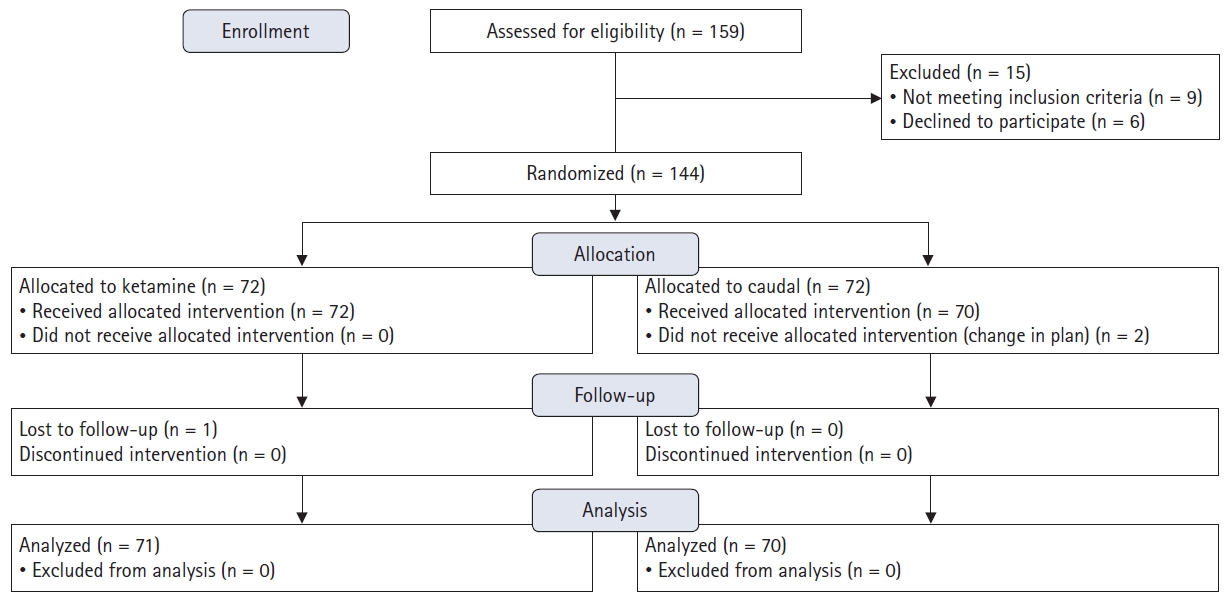
Altogether, 141 children completed the study (ketamine group: n = 71, caudal group: n = 70) The cumulative proportion of children without significant postoperative pain (FLACC score < 4) in the first 6 h post-surgery was 45.1% in the ketamine group vs. 72.9% in the caudal group (P < 0.001). More children in the ketamine group required an additional dose of intraoperative fentanyl (33.8% vs. 5.7%, P < 0.001) and postoperative tramadol (54.9% vs. 27.1%, P < 0.001). However, postoperative agitation, sedation, and other secondary outcomes were similar between the groups.
Caudal epidurals are commonly performed for postoperative pain management in children undergoing abdominal and lower limb surgeries [1,2]. However, apart from procedural failures, potential complications include inadvertent dural punctures, infections, and local anesthetic systemic toxicity. Moreover, in children with coagulopathy, pilonidal cysts, spinal dysraphism, or local site infections, caudal epidurals are contraindicated [2,3]. Caudal epidurals are also not recommended for some surgeries, such as hypospadias repair, since it can cause penile vasodilation, which increases blood loss and may result in fistula formation [4].
Various clinical studies support the use of subanesthetic intravenous (IV) ketamine as an analgesic, particularly for acute pain in the perioperative setting [5]. Ketamine has been added to local anesthetics in caudal epidurals to prolong postoperative analgesia in pediatric patients; however, this does not eliminate procedural risks, and concerns have also been raised that additives in the epidural or intrathecal space may cause neurotoxicity [5]. One previous study found that the efficacy of caudal bupivacaine and caudal ketamine were comparable for postoperative analgesia in children undergoing inguinal hernia surgery, with better postoperative recovery profiles in the caudal ketamine group [6]. However, whether IV subanesthetic ketamine provides a level of postoperative analgesia that is similar to that of caudal bupivacaine in children is unknown.
Hence, we hypothesized that the analgesic effect of subanesthetic IV ketamine is non-inferior to that of caudal bupivacaine in children undergoing infra-umbilical surgeries. If confirmed, the results of this study would provide evidence for an easier method of postoperative analgesia in children without the risk of complications associated with caudal epidurals. In addition, it may obviate the need for multimodal analgesia and polypharmacy, such as non-opioid and opioid analgesics in children who are unable to receive caudal epidurals [7]. The primary aim of this study was thus to evaluate the efficacy of a subanesthetic dose of IV ketamine (0.3 mg/kg) vs. caudal 0.125% bupivacaine (1 ml/kg) for postoperative analgesia in children. The secondary aims of this study were to evaluate the differences in intra- and postoperative opioid consumption and the safety of ketamine vs. caudal bupivacaine through assessing the time to extubation, postoperative vomiting (POV), postoperative agitation, and sedation and comparing the baseline and postsurgical inflammatory markers between the two groups.
The study was a prospective, single-blind, non-inferiority, randomized controlled trial conducted in a tertiary care teaching hospital for two years. Children who were aged < 6 years; with American Society of Anesthesiologists physical status (ASA-PS) classification I & II; and scheduled for elective infra-umbilical, inguinoscrotal, or lower limb surgery were enrolled. The exclusion criteria were as follows: children with cardiovascular diseases, clotting disorders, a known drug allergy to either ketamine or bupivacaine, those with contraindications to caudal anesthesia, and those whose parents/guardians refused to provide consent.
The Institutional Ethics Committee approved the study (AIIMS, Bhubaneswar, T/IM-F/17-18/16; Chairman: Dr. Suresh Chandra Dash; date of approval 9th January 2018), and written informed consent was obtained from each participant’s parent or guardian. The study was registered prospectively in the Clinical Trials Registry of India (CTRI) (trial registration number: CTRI/2018/02/011822; Principal Investigator: Dr. Alok Kumar Sahoo; study start date 1st March 2018) and the procedures were conducted in accordance with the Helsinki Declaration-2013.
The children were randomized into two groups (Caudal and Ketamine) using computer-generated randomization codes. Allocation concealment was performed using opaque sealed envelopes, which were opened by the attending anesthesiologists once the patients were received in the preoperative holding area on the day of the surgery. Since a sham caudal group was not planned, the anesthesiologists were not blinded to group allocation.
Monitoring, anesthesia, fluid, and temperature management were standardized for both groups. Children with a previous IV line were given inj. midazolam (0.1 mg/kg) IV 5 min before parental separation, and children without an IV line were given oral midazolam (0.5 mg/kg) 30 min before parental separation.
Anesthesia was induced with inj. glycopyrrolate (10 μg/kg), fentanyl (2 μg/kg), and propofol (2 mg/kg), IV. Muscle relaxation was achieved using inj. atracurium (0.5 mg/kg), IV. The airway was secured using an appropriately-sized tracheal tube or supraglottic airway device. Anesthesia was maintained using a mixture of oxygen in air and sevoflurane at 1 minimum alveolar concentration. After induction, 2–3 ml of blood was collected to measure interleukin-6 (IL-6) and tumor necrosis factor-α (TNF-α) levels (baseline) since both cytokines are markers of inflammation and are associated with the pro-nociceptive activity.
Following the induction of anesthesia, children randomized to the ketamine group received a sub-anesthetic dose of inj. ketamine (0.3 mg/kg) IV, and those randomized to the caudal group were administered a caudal epidural using the loss of resistance technique containing 1 ml/kg of 0.125% bupivacaine to a maximum dose of 2 mg/kg. inj. fentanyl (0.25 μg/kg) IV was supplemented if the heart rate and/or blood pressure responses increased > 20% from baseline. In addition, all children received 15 mg/kg of inj. acetaminophen IV at induction, which was scheduled every 8 h for the first 24 h post-operation.
After surgery, inj. glycopyrrolate (10 μg/kg) and neostigmine (50 μg/kg) IV were administered to antagonize the neuromuscular blockade. The time to extubation after the neuromuscular blockade antagonists were administered, and the incidence of postoperative agitation (measured using the Watcha scale) in the first 30 min after extubation were recorded. A Watcha scale score ≥ 2 was used to indicate the presence of emergence agitation [8]. A second blood sample was drawn 3 h post-surgery to determine postoperative inflammatory marker levels.
In the postoperative anesthesia care unit (PACU), the pain was assessed using the Face, Legs, Arm, Cry, Consolability (FLACC) scale at 30 min and 1 and 2 h, and subsequently in the surgical ward at 3 and 6 h post-operation [9]. The license to use the FLACC scale was obtained from the University of Michigan Office of Technology (License Agreement #9709-umich). Children with FLACC scores ≥ 4 at any point in the first 6 h post-operation were reported as having a positive pain response. Children with a positive pain response received IV rescue analgesia with inj. tramadol (1 mg/kg) IV.
The Ramsay sedation score (RSS) was used to assess sedation in the PACU, with an RSS > 3 signifying excessive sedation. POV was another adverse effect considered. All postoperative outcome assessments were performed by trained nursing personnel who were blinded to the groups. The study period lasted from induction of anesthesia to 6 h post-operation.
The primary aim was to evaluate the non-inferiority of subanesthetic ketamine (0.3 mg/kg) vs. 0.125% caudal bupivacaine (1 ml/kg) for postoperative pain. The primary outcome was the cumulative proportion of children with a FLACC score ≥ 4 at 6 h post-operation.
The secondary aims were to evaluate the differences in intra-and postoperative opioid requirements and the safety of ketamine vs. caudal bupivacaine in children. The secondary outcomes assessed were the time to extubation after neuromuscular block antagonism, POV, postoperative agitation, sedation, and baseline and postsurgical inflammation.
A sample size of 72 was required for each group to prove non-inferiority with a margin of 5% (beyond which it is not considered clinically meaningful). The response rate in the caudal analgesia group (i.e., the proportion of children with a FLACC score < 4 at 6 h post-operation) was set at 96% [10], and the response rate in the IV ketamine group was set at 90%, since this would be clinically significant for pain relief. The sample size was powered at 80%, allowing for an alpha error of 0.05, and a drop-out rate of 10% was considered.
The Shapiro-Wilk test was used to assess the normality of the data. The parametric data were analyzed using unpaired t-tests, while the non-parametric data were analyzed using the Mann-Whitney U test. Categorical data were analyzed using the chi-square test. Regression techniques were used to determine the association between the variables. Statistical significance was set at P < 0.05 (two-tailed). Data were analyzed using R software (version 3.5, R studio, Switzerland).
A total of 159 children were considered over a 1.5 yr period, 141 of which completed the study. A total of 71 children were finally included in the ketamine group and 70 in the caudal group (Fig. 1). We excluded two children from the caudal group due to a change in the surgical plan and one from the ketamine group due to inaccurate data collection.
Baseline characteristics, including age, sex, and weight, were comparable between the groups (Table 1). Most of the surgeries had a short duration, and the durations were similar between the groups. The infra-umbilical surgeries conducted were as follows: inguinal hernia repair, hypospadias and chordee correction, undescended testis correction, and various other surgeries (Table 1).
The proportion of children with a FLACC scale score < 4 in the first 6 h post-operation was 45.1% in the ketamine group and 72.9% in the caudal group (P < 0.001) (Fig. 2, Table 2). The mean FLACC scale score in the caudal group was significantly lower at 30 min and at 1, 2, and 3 h post-operation (P < 0.001) (Fig. 3). The lower limit of the confidence interval (–0.47) for the difference between the groups crossed the non-inferiority limit of –0.05, suggesting that subanesthetic IV ketamine was inferior to caudal analgesia. Mixed effect modeling of the FLACC responses at various time points with the groups as covariant showed a significant difference in the response trends between the two groups across all time points (P < 0.05).
The proportion of children requring an additional dose of intraoperative fentanyl in the ketamine group was 33.8% compared to 5.7% in the caudal group (P < 0.001). The mean dose of fentanyl was also significantly higher in the ketamine group (Table 2). The standardized mean difference between the two groups for fentanyl consumption was 0.56. The proportion of children requiring postoperative tramadol was also significantly higher in the ketamine group in the first 6 h post-operation (54.9% vs. 27.1%, P < 0.001) (Table 2).
The time to tracheal extubation was comparable between the groups (Table 2). Postoperative agitation and sedation were also similar between the groups (Table 2). Only four children, three in the ketamine group and one in the caudal group, experienced POV in this study. Serum TNF-α and IL-6 levels were lower in the ketamine group than in the caudal group, although the difference was not statistically significant at any of the time points (Table 2).
The main finding of this study was that sub-anesthetic ketamine was inferior to caudal epidural analgesia with bupivacaine for postoperative pain relief measured using the FLACC scale score for the first 6 h post-operation in children undergoing infra-umbilical surgery under general anesthesia. The FLACC scale score at 6 h post-operation was similar between the groups, possibly due to the caudal effect wearing off. However, the degree of early postoperative inflammation and other secondary outcomes, such as POV, sedation, and postoperative agitation, were similar between the groups.
Ketamine causes spinal inhibition of nociceptive transmission [11]. Subanesthetic ketamine has been widely used for postoperative pain relief, but controversy still exists regarding the optimal dose, duration, and timing of administration [12,13]. Our results are different from those reported by Naguib et al. [6] in their study of 50 children in which they found comparable analgesic efficacy of low-dose caudal ketamine (0.5 mg/kg) with caudal 0.25% bupivacaine (1 ml/kg). In their study, the analgesic efficacy of caudal ketamine was observed until 24 h post-operation. Significantly, a greater number of the children in the caudal ketamine group were calm and cheerful postoperatively at 60 and 90 min compared to the caudal bupivacaine group. Differences between our study results vs. those of Naguib et al. could have been due to the route of administration and the dose of ketamine (0.3 mg/kg in our study vs. 0.5 mg/kg in the study by Naguib et al. [6]). The lack of difference in postoperative recovery profiles in our study could be similarly explained by differences in the pharmacodynamics between the two routes. Thus, the equivalence of the two routes for ketamine likely does not exist.
Our study findings were similar to those of Dix et al. [14], wherein 75 children were given an IV pre-incisional bolus (0.5 mg/kg) either alone or in combination with a postoperative infusion of ketamine (4 µg/kg/min) vs. saline placebo during appendicectomy. The authors did not find any improvement in pain score (at rest or with movement) or a decrease in morphine consumption in the ketamine group. They concluded that ketamine might not have the same opioid-sparing effect in children as in adults [14]. Another explanation for the lack of effect in our study could be that the patients received pre-emptive ketamine. It has been suggested that ketamine may block N-methyl-D-aspartate receptors more effectively in adults if it has been previously opened by an intense or noxious stimulus or the so-called foot-in-the-door blockade phenomenon [15]. A similar phenomenon may occur in pediatric patients. Thus, the timing of administration, that is, pre-emptive vs. postsurgical, may lead to different responses.
IL-6 and TNF-α are major proinflammatory acute-phase proteins that are secreted in response to the tissue damage caused by surgery. These cytokines are known to modulate inflammation and nociception and possibly contribute to pain intensification. Increased levels of these cytokines are associated with increased inflammation and pain [16]. Previous studies have shown that ketamine has an anti-inflammatory effect and reduces the levels of proinflammatory cytokines TNF-α and IL-6 by immunomodulation [17-20]. This regulatory action was more pronounced when ketamine was administered before the noxious stimulus. In our study, the similar postsurgical cytokine levels between the ketamine and caudal groups suggest that even though ketamine was inferior to caudal bupivacaine for postoperative analgesia, the anti-inflammatory response was similar. The 3-hour postoperative window for the sampling of postoperative inflammatory markers used in this study is consistent with previous studies, since TNF-α decays by the third hour after exposure to toxins/surgery, while IL-6 levels are detected in the blood by the second hour and peak at 12–24 hours post-surgery [21,22]. We are unable to comment on the apparent dissociation of the inflammatory and nociceptive action, but it is possible that the peak effect may have been missed in the two groups due to differences in the time of rising and peak between the markers.
The adverse effects were comparable between the groups, indicating that IV subanesthetic ketamine is safe. Ketamine can cause excessive sedation and agitation; however, this effect is more pronounced at an anesthetic dose [23]. In our patients, we found that extubation time, sedation, and postoperative agitation scores were similar between the groups. Our findings are similar to those of Sinha and Sood [24], who found that caudal ketamine does not increase or cause agitation. Thus, sub-anesthetic ketamine appears to have a good safety profile in this patient population.
The main limitation of our study was the absence of a placebo group; however, our aim was not to show the superiority of ketamine versus placebo for postoperative analgesia but rather to show the non-inferiority of the intervention with caudal epidurals to test our hypothesis. Second, we only followed the patients’ FLACC scale scores for the first 6 h post-operation, as the effects of even “kiddie” caudals wear off in a sizeable proportion of children after this time [25]. The action of a bolus dose of subanesthetic ketamine is unlikely to have a long duration, which is also a potential limitation of this study. However, in children, there is very limited evidence regarding the postsurgical administration of subanesthetic ketamine, and thus, we are unable to comment on whether the timing of ketamine administration would be more appropriate after surgery in this patient population. Another limitation was that some of the outcomes, such as intraoperative opioids and time to extubation, were not blinded.
In conclusion, a subanesthetic dose of IV ketamine is inferior to caudal analgesia with bupivacaine in children aged < 6 years undergoing infra-umbilical surgeries. Unless there is a specific contraindication, children in this age group should continue to receive caudal epidurals for postoperative analgesia for these surgeries. Further studies to evaluate the effect of ketamine infusions or post-surgery subanesthetic administration should be considered.
NOTES
Author Contributions
Alok Kumar Sahoo (Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Supervision; Writing – original draft; Writing – review & editing)
Satyajeet Misra (Conceptualization; Investigation; Methodology; Project administration; Supervision; Writing – review & editing)
Bikram Kishore Behera (Conceptualization; Methodology; Project administration; Supervision; Writing – review & editing)
Anand Srinivasan (Data curation; Investigation; Resources; Software; Validation)
Sritam Swarup Jena (Methodology; Project administration; Resources; Supervision)
Manoj Kumar Mohanty (Project administration; Resources; Supervision)
Fig. 2.
Cumulative Face, Legs, Activity, Cry, Consolability (FLACC) scale scores between the groups for the first 6 h post-operation.

Fig. 3.
Mean Face, Legs, Activity, Cry, Consolability (FLACC) scale scores over time according to group.
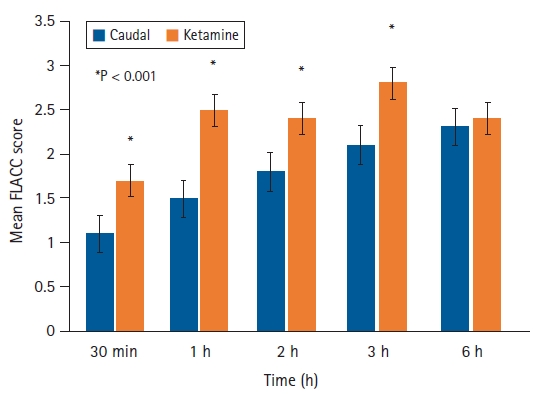
Table 1.
Baseline Characteristics
Table 2.
Primary and Secondary Outcome Parameters
References
1. de Beer DA, Thomas ML. Caudal additives in children-solutions or problems? Br J Anaesth 2003; 90: 487-98.


2. Wiegele M, Marhofer P, Lönnqvist PA. Caudal epidural blocks in paediatric patients: a review and practical considerations. Br J Anaesth 2019; 122: 509-17.



3. Suresh S, Lonnqvist PA. Regional anesthesia in children. In: Miller’s Anaesthesia. 9th ed. Edited by Gropper MA Cohen NH Eriksson LI Fleisher LA Leslie K Winer-Kronish JP: Philadelphia, Elsevier Health Sciences. 2019, pp 2368-419.
4. Taicher BM, Routh JC, Eck JB, Ross SS, Wiener JS, Ross AK. The association between caudal anesthesia and increased risk of postoperative surgical complications in boys undergoing hypospadias repair. Paediatr Anaesth 2017; 27: 688-94.



5. Amiri HR, Espandar R, Sanatkar M. Comparing caudal and intravenous ketamine for supplementation of analgesia after Salter innominate osteotomy. J Child Orthop 2012; 6: 479-83.



6. Naguib M, Sharif AM, Seraj M, el Gammal M, Dawlatly AA. Ketamine for caudal analgesia in children: comparison with caudal bupivacaine. Br J Anaesth 1991; 67: 559-64.


8. Lee CA. Paediatric emergence delirium: an approach to diagnosis and management in the postanaesthesia care unit. J Perioper Crit Intensive Care Nurs 2017; 3: 140.

9. Merkel SI, Voepel-Lewis T, Shayevitz JR, Malviya S. The FLACC: a behavioral scale for scoring postoperative pain in young children. Pediatr Nurs 1997; 23: 293-7.

10. Dalens B, Hasnaoui A. Caudal anesthesia in pediatric surgery: success rate and adverse effects in 750 consecutive patients. Anesth Analg 1989; 68: 83-9.


11. Hirota K, Lambert DG. Ketamine: its mechanism(s) of action and unusual clinical uses. Br J Anaesth 1996; 77: 441-4.


12. Sheehy KA, Lippold C, Rice AL, Nobrega R, Finkel JC, Quezado ZM. Subanesthetic ketamine for pain management in hospitalized children, adolescents, and young adults: a single-center cohort study. J Pain Res 2017; 10: 787-95.



13. Martindale SJ, Dix P, Stoddart PA. Double-blind randomized controlled trial of caudal versus intravenous S(+)-ketamine for supplementation of caudal analgesia in children. Br J Anaesth 2004; 92: 344-7.


14. Dix P, Martindale S, Stoddart PA. Double-blind randomized placebo-controlled trial of the effect of ketamine on postoperative morphine consumption in children following appendicectomy. Paediatr Anaesth 2003; 13: 422-6.


16. De Kock M, Loix S, Lavand’homme P. Ketamine and peripheral inflammation. CNS Neurosci Ther 2013; 19: 403-10.



17. Loix S, De Kock M, Henin P. The anti-inflammatory effects of ketamine: state of the art. Acta Anaesthesiol Belg 2011; 62: 47-58.

18. Morris P, Ali K, Merritt M, Pelletier J, Macedo LG. A systematic review of the role of inflammatory biomarkers in acute, subacute and chronic non-specific low back pain. BMC Musculoskelet Disord 2020; 21: 142.



19. Chen MH, Li CT, Lin WC, Hong CJ, Tu PC, Bai YM, et al. Rapid inflammation modulation and antidepressant efficacy of a low-dose ketamine infusion in treatment-resistant depression: a randomized, double-blind control study. Psychiatry Res 2018; 269: 207-11.


20. Kawasaki C, Kawasaki T, Ogata M, Nandate K, Shigematsu A. Ketamine isomers suppress superantigen-induced proinflammatory cytokine production in human whole blood. Can J Anaesth 2001; 48: 819-23.


21. Watt DG, Horgan PG, McMillan DC. Routine clinical markers of the magnitude of the systemic inflammatory response after elective operation: a systematic review. Surgery 2015; 157: 362-80.


22. Bantis A, Tsakaldimis G, Zissimopoulos A, Giannakopoulos S, Kalaitzis C, Pitiakoudis M, et al. Can tumor necrosis factor- α and interleukin-6 be used as prognostic markers of infection following ureteroscopic lithotripsy? ISRN Urol 2014; 2014: 457063.

23. Lee EN, Lee JH. The effects of low-dose ketamine on acute pain in an emergency setting: a systematic review and meta-analysis. PLoS One 2016; 11: e0165461.