Introduction
Securing the airway is a vital component in the clinical practice of anesthesia. Difficult intubation (DI) is prone to potential complications, ranging from minimal airway edema to life-threatening events. Predicting DI during the preoperative assessment is a key challenge, as no single clinical predictor is sufficiently valid for predicting the outcomes. Various imaging techniques have been under consideration for evaluation of the airways, but each has specific limitations, such as radiation exposure, high cost, and procedure time, etc.
Ultrasonography is a non-invasive and quick bedside tool that allows easy visualization of the neck anatomy and assessment of the airway [
1,
2]. Various ultrasonography-related parameters such as tongue thickness (TT), the invisibility of hyoid bone (VH), mandible condylar mobility, and anterior neck soft tissue thickness from the skin to the thyrohyoid membrane (ST) and the hyoid bone (SH), respectively, have the potential of predicting difficult airway. The current literature is, however, limited to small studies, restricted further by the low incidence of DI [
3–
5]. Thus, the validity of ultrasonography-based parameters in predicting DI requires further exploration. We hypothesized that upper airway ultrasonographic parameters including TT, SH, ST, and VH would reliably predict DI during preoperative assessment in patients without anticipated difficult airway. We preferred these parameters considering the ease and rapidity in locating their anatomical landmarks to allow precise measurements, their potential ability to predict DI, and the limited role of clinical screening in their evaluation. Our primary aim was to evaluate the effectiveness of aforesaid ultrasonographic parameters in predicting DI by comparing them between the DI and easy intubation (EI) groups. We also analyzed the validity of various models with combined ultrasonography-based parameters in predicting DI.
Materials and Methods
After obtaining institutional ethical approval (AIIMS/IEC/18/85) and written informed consent, patients of both sexes classified as American Society of Anesthesiologists physical status I–III, aged 18 to 60 years, undergoing tracheal intubation for surgery under general anesthesia, were included in this prospective, observational, cohort study conducted between August 2018 to July 2019 (Indian Clinical Trial Registry No: CTRI/2018/07/014786). This clinical research was done following the ethical principles for medical research involving human subjects in accordance with the Helsinki Declaration 2013. An experienced investigator conducted a difficult airway assessment during the preoperative visit. Those with upper airway anatomical anomaly, trauma, or tumor; history of a difficult airway; and difficult airway on pre-anesthetic check-up and those requiring deviation in the study protocol were excluded. A modified Mallampati test (MMP) grade of 3 or 4, small thyromental distance (< 6.5 cm), and small inter-incisor distance (< 3 cm) indicate the presence of difficult airway.
All enrolled patients underwent a duly explained ultrasonographic examination (Logic eR7, GE Medical Systems Co. Ltd., China) of the upper airway in the pre-operative room. A skilled investigator (≥ 5 years of experience in airway ultrasonography) performed the procedure and recorded the measurements. For sublingual ultrasonography, patients were positioned in the sitting, neutral head position [
4]. A curved ultrasound probe (4.2–10.0 MHz in a sterile cover) was placed intra-orally under the patient's tongue in a longitudinal orientation (perpendicular to the face) and advanced backwards as far as the patient felt comfortable (
Fig. 1A). The hyoid bone was noted in the obtained image (
Fig. 1D). For submandibular ultrasonography, all patients were asked to remain in a supine and extended neck posture, to keep their mouth closed, and to remain quiet, with the tip of tongue relaxed and just touching the incisors [
6]. A curvilinear ultrasound probe (2–5 MHz) was positioned beneath the mentum along the mid-sagittal plane, adjusted to obtain a clear image of the tongue contour (
Fig. 1B). The maximum vertical length from the tongue surface to the submental skin was noted and defined as TT (
Fig. 1E). In the same position, the transducer was rotated to 90° and SH and ST, were measured (
Figs. 1C,
1F and
1G).
Thereafter, the patients were moved to the operation theatre, and standard monitors were applied. After adequate pre-oxygenation, general anesthesia was induced with propofol (1–2 mg/kg IV), midazolam (0.05 mg/kg IV), fentanyl (0.004 mg/kg IV), and vecuronium (0.1 mg/kg IV). After 3 minutes of mask ventilation, an experienced investigator (≥ 5 years of experience in intubation procedure) performed the laryngoscopy (Macintosh blade size 3 or 4) in the sniffing position. To facilitate laryngeal view, external laryngeal manipulation was allowed, and intubation was attempted. In case of failed attempts, standard protocols were followed as per unanticipated DI guidelines [
7]. ‘DI’ was defined as the placement of the endotracheal tube by using conventional laryngoscopy that required > 2 attempts, lasted > 10 min, or required alternate methods [
7]. The ‘time taken for intubation’ was defined as the time point from initiation of the first direct laryngoscopy attempt to confirmation of successful endotracheal intubation by continuous waveform capnography. The difficult airway cart included intubating stylet (IS), McCoy blade (MB), intubating laryngeal mask airway (ILMA), video laryngoscope (VL), light wand (LW), fiberoptic bronchoscope, and percutaneous cricothyroidotomy. The endpoint of the study was tracheal intubation, based on which all the included patients were categorized as EI or DI.
Clinical airway assessment data and ultrasonography-based data were categorized according to the group for statistical analysis and interpretation. The investigator who performed the ultrasonographic examination was blinded to the preoperative airway assessment data, intubation procedure, and group allocation. Another investigator carried out the preoperative airway assessment, intubation procedure and group allocation, but was blinded to ultrasonographic parameters. Another investigator blinded to study protocols performed the data analysis.
Statistical analysis
The sample size was calculated using the sample size calculator of the University of California, San Francisco, US [
8]. Taking an alpha error of 5%, power of 80%, the incidence of unanticipated DI as 5% (a weighted average of data from the literature [1–9.5%]), the sample size was calculated as 1,030 (EI: 978, DI: 52), considering an effect size of 0.8 (estimated from initial pilot observations) for the primary outcome [
3,
9]. Statistical analysis was performed using MedCalc software version 19.0.7 (Acacialaan, Belgium). The results were presented as descriptive statistics, summarized as mean (SD) or number (percentage). Data were analyzed by logistic regression, receiver operator characteristic (ROC) curve, and Youden index to calculate the diagnostic validity profile of the outcome variables. The continuous variables were compared by unpaired student t-test. The categorical variables were compared by Chi-square test/Fisher’s exact test. A P < 0.05 was considered significant.
Results
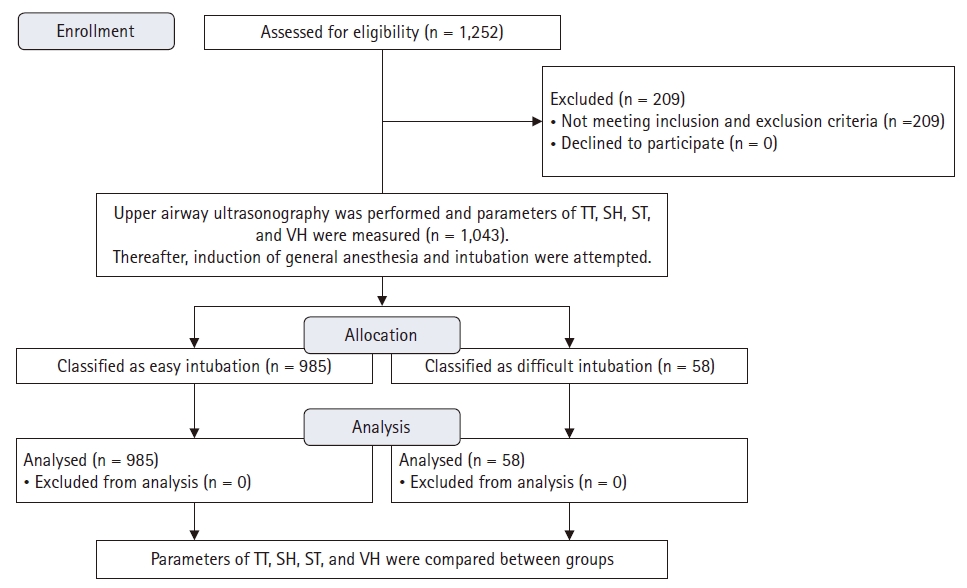
We assessed 1,252 patients for eligibility, of which 1,043 were included in the study (no dropouts). The demographic profile was comparable among the groups (
Table 1). In all, 985 patients were classified as EI, as 58 patients as DI (
Fig. 2). The EI group had significantly lower MMP grade than the DI group. On direct laryngoscopy, the EI group had a Cormack-Lehane (CL) grade of 1 or 2, while DI group had a CL grade of 2 or 3 with some grade 4 cases as well, which varied significantly on intergroup comparison (
Table 1). Further, 802 patients could be intubated in the first attempt, while the remaining required repeat attempts or alternate methods, such as IS+MB in 27 patients, ILMA in 24 patients, VL in 10 patients, and LW in one patient, with success rates of 96.3%, 91.6%, 90%, and 100%, respectively.
The means of the upper airway ultrasonography-based parameters TT, SH, and ST were significantly greater in the DI group than in the EI group (P < 0.001, respectively) (
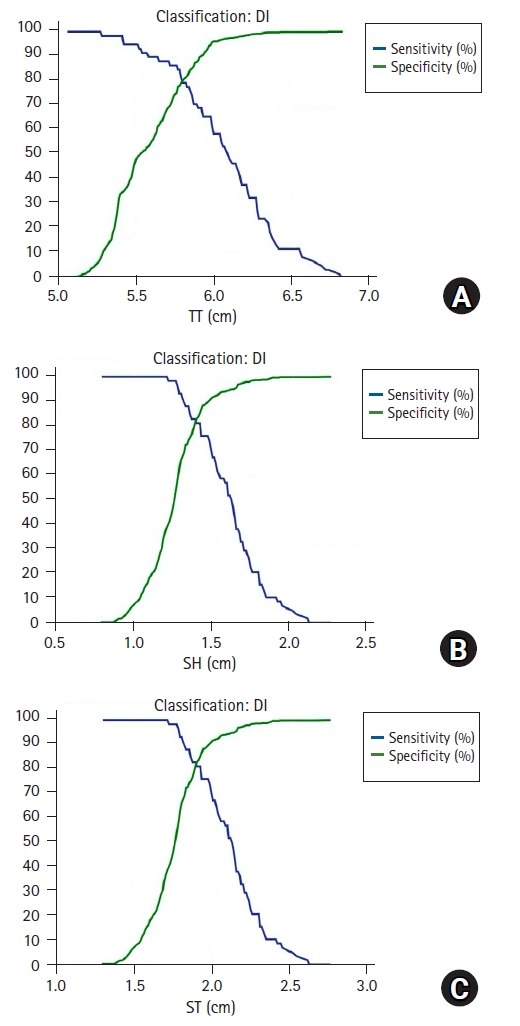
Table 2). The VH was 14.4% in the EI group versus 72.4% in the DI group (P < 0.001). For validity analysis, an ROC curve was plotted for each ultrasonographic parameter. The optimal criterion for TT to predict DI was found to be > 5.8 cm (sensitivity: 84.5%, specificity: 78.1%) with an area under the curve (AUC) of 0.880. For SH, the optimal value was > 1.4 cm (sensitivity: 81%, specificity: 85.2%), with an AUC of 0.898; for ST, it was > 2.4 cm (sensitivity: 75.9%, specificity: 85.2%), with an AUC of 0.885. The sensitivity and specificity for VH were 72.4% and 85.6%, respectively, with an AUC of 0.790 (
Table 2). We also plotted a graph to determine the changes in sensitivity and specificity for each threshold value of TT, SH, and ST. An increase in TT, SH, and ST thresholds led to an increase in the specificity but a decrease in the sensitivity for identifying DI (
Fig. 3). The TT, SH, ST, and VH had the accuracies of 78.4%, 85.0%, 84.7%, and 84.9%, respectively. On univariate analysis, the odds ratio (OR) for TT was 1.06, indicating a 6% increase in the log-odds of DI per millimeter increase in TT. Similarly, the ORs for SH, ST, and VH were 1.07, 1.10, and 15.58, respectively. A wide CI (8.52, 28.47) was observed for VH, indicating a low level of precision (
Table 2). A subgroup analysis was also performed to compare the ultrasonographic parameters in those with CL grade 2 and a similar difference was observed between the ultrasonographic parameters.
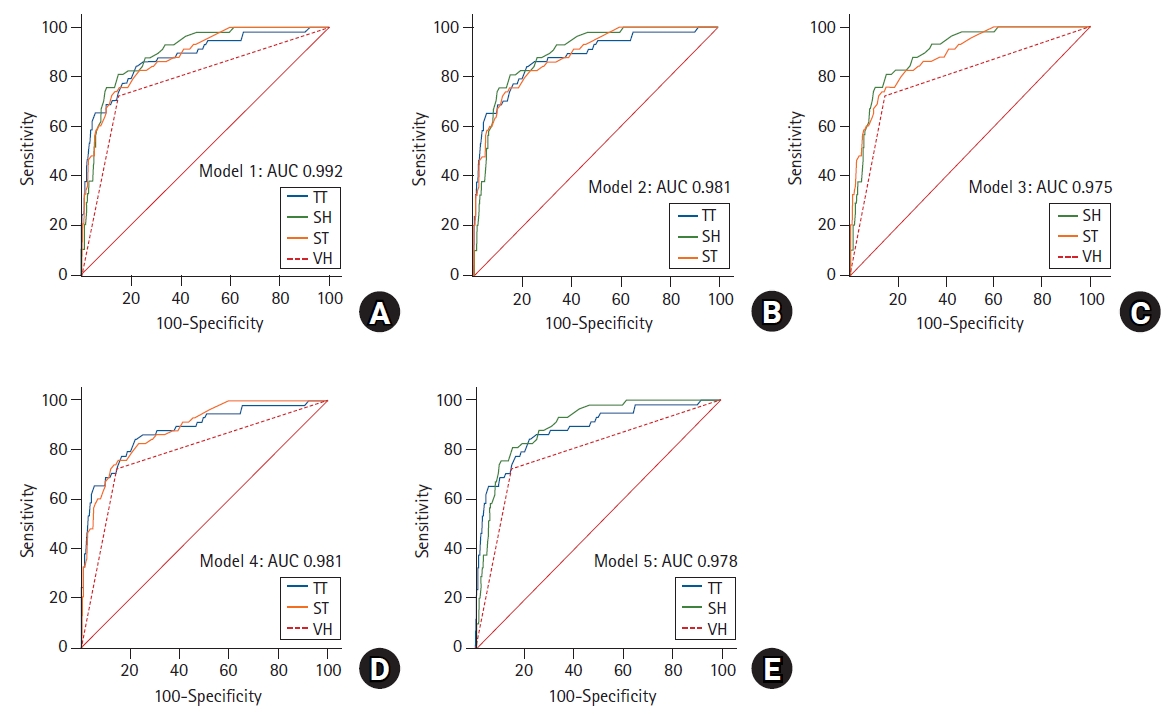
The validity of five models based on combined ultrasonography-based parameters was also assessed through multiple logistic regression-derived ROC analysis (
Table 3,
Fig. 4). ‘Model 1’, which included all the four ultrasonographic parameters, had the highest accuracy with an AUC of 0.992. ‘Model 2’, which included TT, SH, and ST, and ‘Model 4’, with TT, ST, and VH, had an AUC of 0.981. ‘Model 3’, which included SH, ST, and VH, had an AUC of 0.975. ‘Model 5’, which included TT, SH, and VH, had an AUC of 0.978. On evaluating the relative contribution of each parameter, VH had the largest OR, though a wide CI was observed for the OR values of VH. ST was the second variable to have a strong impact on the diagnostic validity of the models (
Table 3).
Discussion
This study demonstrated the ability of individual upper airway ultrasonography-based parameters and of models using a combination of these paraments in predicting DI. All the upper-airway ultrasonographic parameters varied significantly between the EI and DI groups. The SH parameter had the highest accuracy, while the TT was least accurate in predicting DI. Among the five models, ‘Model 1’ with all the four ultrasonographic parameters had the highest validity in terms of the AUC.
For optimal viewing of the glottis during direct laryngoscopy, the soft tissues in the neck need to be mobilized adequately. Adhikari et al. [
10] evaluated the anterior neck soft tissue thickness at different ultrasonographic planes and observed that ST and SH correlated strongly with difficult laryngoscopy. We used similar levels for predicting DI and obtained a threshold limit of 1.4 cm for SH and 2.4 cm for ST. Wu et al. [
11] found an SH cut-off of 1.28 cm while Adhikari et al. [
10] obtained an ST limit of 2.8 cm, for predicting difficult laryngoscopy. Yadav et al. [
5] reported cut off values of 0.66 and 2.03 cm, respectively, for SH and ST. It appears that different cut-off targets are required for predicting difficult laryngoscopy and intubation. The small sample sizes in the above studies could have attributed to such variation.
Previous studies have correlated increased TT to difficult laryngoscopy [
5,
6]. However, the measurements varied significantly according to the anatomic level of the ultrasonographic scans. We measured the TT in the mid-sagittal plane to obtain values of the thickest portion of entire tongue contour. The ROC analysis showed that TT > 5.8 cm predicted risk of DI with a sensitivity of 84.5% and specificity of 78.1%, with a 6% increase in the log-odds of DI per millimeter increase in TT values. Yao and Wang [
6] observed that a cut-off value of TT as 6.1 cm predicted DI. The observed variation could be due to a difference in baseline demographics and ethnicities of the studied population. The increased TT also relates to the high MMP grade. Our study had a higher proportion of patients with MMP grade 2 in the DI group (mean TT of 6.1 cm) than in the EI group (mean TT of 5.8 cm). A larger TT is expected to mask the visibility of the faucal pillars, which justifies our results. Though MMP grade 1 or 2 patients are classified as EI, previous studies have shown that it as an inadequate stand-alone test for predicting difficult airway [
12]. We excluded the patients with MMP grades 3 and 4 to identify the threshold for those in which difficult airway could not be anticipated by clinical examination. As no clinical predictor is sufficient to predict DI, our results serve to complement the pre-anesthetic workup in anticipating DI. The CL grades of 1 and 2 are considered easy laryngoscopy, but 26 patients with CL grade 2 were classified as DI in our study. The factors that contributed to DI in these patients included visibility of only the posterior part of the glottis or arytenoid during direct laryngoscopy, deep-seated larynx, or reduced space within the oropharynx [
13]. These patients were intubated at the third attempt by IS with MB, ILMA, or VL. Hui and Tsui [
4] observed a correlation between VH and difficult laryngoscopy. We observed a similar pattern in the DI group, although VH was also observed in 14% of the EI group. The caudal displacement of the hyoid bone reduced its visibility on ultrasonography probe, possibly because of the hypopharyngeal position of the tongue or short rami of the mandible, which compromises the view of the glottis during direct laryngoscopy [
4].
We also analyzed the validity of five different models with different combinations of the studied ultrasonographic parameters. The predictive ability of the combinations was significantly better than that of the individual parameters, as evidenced by an increased AUC (0.975, 0.992). ‘Model 1’ had the highest AUC (0.992) ‘Models 2 and 4’ were the next best (AUC 0.981), while the ‘Model 3’ had the lowest AUC (0.975). Considering the inclusion of three parameters in a single submandibular window, with no need of intraoral probe placement, and an acceptable AUC, ‘Model 2’ seems to be a viable option. To analyze the individual contribution of each variable in the model, we calculated the OR. Taking into account the wide CI in OR values of VH, indicating a low precision level, ST appears to have a significant impact on the validity of models. A large sample size may, however, efficiently delineate the impact of VH. Future studies can attempt to design a scoring system/formula based on combined ultrasonography-based variables, considering the weightage of each ultrasonographic parameter in predicting DI. This study can serve as the base for such trials.
Our study had several limitations. We only analyzed the anatomic parameters of the airway without taking into account the impact of functional components like head positioning, degree of neck extension, skills of performer, etc. which may alter the difficulties encountered during the intubation procedure. To avoid the associated bias, we standardized all the functional parameters in our study. Patients in the EI and DI groups were distributed unevenly, which may have affected the validity of results, but this was as expected, considering the low incidence of unanticipated DI. We could not randomize the patients, but the investigators were blinded. Thus, we expect that the above limitations did not severely distort the results.
In conclusion, the SH had better accuracy than the remaining three ultrasonographic parameters included in the study. Although the individual parameters showed limited validity, a model combining all the four parameters offered better diagnostic profile than each one of them.