|
 |
|
|
Abstract
Background
We investigated the effects of the combined administration of nefopam, a N-methyl-D-aspartate receptor antagonist and low dose remifentanil, on early postoperative pain and analgesic requirement.
Methods
Fifty patients scheduled to undergo mastoidectomy and tympanoplasty were randomized to be given either nefopam 40 mg mixed with normal saline 100 ml (Group N) or an equal amount of normal saline (Group C) before anesthesia induction. Anesthesia was maintained with 5-6 vol% desflurane and remifentanil 0.05-0.15 ┬Ąg/kg/min during the surgery. Postoperative pain was controlled by titration of ketorolac in the postanesthesia care unit (PACU) and ward. We evaluated the intraoperative remifentanil dose, recovery profiles, ketorolac demand in the PACU and ward, numeric rating scale (NRS) for pain at time intervals of every 10 min for 1 h in the PACU, 6, 12, 18 and 24 h in a ward, as well as the time to first analgesic requirement in the PACU and ward.
Results
Ketorolac demand and NRS in the PACU were significantly lower in Group N than Group C (P = 0.002, P = 0.005, respectively). The time to first analgesic requirement in the PACU in Group N were significantly longer than Group C (P = 0.046). There were no significant differences in intraoperative remifentanil dose, ketorolac demand, NRS, and the time to first analgesic requirement in the ward between the groups.
Remifentanil is an ultra-short-acting analgesic which causes a predictable and fast recovery due to rapid metabolism [1], and can prevent several complications of opioid including respiratory depression [2].
However, many studies have shown opioid-induced hyperalgesia (OIH) and acute opioid tolerance especially when remifentanil is used [3,4]. The OIH and acute opioid tolerance may be related to the high-dose of remifentanil administered during surgery [5,6], but some studies have shown that low-dose of remifentanil also could increase postoperative pain [3,7]. Consequently, there is controversy about the dosage of remifentanil to be used in surgery. The occurrence of OIH and acute opioid tolerance is mainly known to be due to the activation of N-methyl-D-aspartate (NMDA) receptors [8]. Thus, some studies have suggested that ketamine and magnesium prevent remifentanil-induced OIH by blocking NMDA receptors, thereby decreasing postoperative pain [9,10].
Nefopam is a non-narcotic analgesic that acts centrally by inhibiting 5-hydroxytryptamine and noradrenaline uptake [11]. It also reduces the presynaptic release of glutamate which is associated with pain and centrally inhibits postsynaptic NMDA receptors [12]. Nefopam has been used for postoperative pain control [13] and patient-controlled analgesia [14] in several surgeries. Tirault et al. [15] reported that pretreatment of nefopam could prevent postoperative morphine overconsumption which occurred after a large dose of remifentanil and propofol anesthesia in major abdominal surgery. However, there has been no report about the effectiveness of nefopam in middle ear surgery which usually requires a low-dose remifentanil infusion instead of nitrous oxide. Taken together, we speculated that nefopam infusion combined with low dose remifentanil could reduce early postoperative analgesic demand regardless of the occurrence of the remifentanil-induced pronociceptive effect. We investigated the effect of nefopam on the analgesic demand dose as a primary end point and the postoperative pain score and the time to first analgesic requirement as secondary end points in patients undergoing middle ear surgery with desflurane anesthesia and low dose remifentanil infusion.
The study was conducted with a prospective, randomized controlled and double-blinded method for postoperative assessment. This study was approved by the Institutional Review Board and registered in the UMIN clinical trials registry. All procedures were in accordance with the Declaration of Helsinki. We obtained informed consent from 50 patients with the American Society of Anesthesiologists physical status I-II, aged 20-65, who were scheduled to undergo elective mastoidectomy and tympanoplasty. The exclusion criteria included hypersensitivity to nefopam or remifentanil , both of which were used in this study.
All patients were premedicated with intramuscular midazolam 2 mg and glycopyrrolate 0.2 mg 1 h before surgery. In the operating room, patients were given a noninvasive blood pressure, pulse oximetry, electrocardiography and entropy monitor (M-Entropy plug-in module, Datex-Ohmeda, Finland). All patients were randomly allocated by a simple randomization using a computer-generated random number table into one of the two groups. Before patients arrived in the operating room, a study drug (nefopam or normal saline) solution was prepared by a nurse who was not involved in this study. In the nefopam group (Group N), nefopam 40 mg mixed with normal saline 100 ml was slowly administered during 20 min before anesthesia induction [16]. In the control group (Group C), an equal amount of normal saline was administered during the same period. The study drugs were given to patients by an anesthesiologist who was blinded to the treatment group and who was assigned to provide anesthesia for the patients. There were no significant differences in the demographic data between the two groups (Table 1).
Anesthesia was induced with intravenous thiopental sodium 5 mg/kg and rocuronium 0.6 mg/kg and maintained with 5-6 vol% desflurane in 50% oxygen in air adjusted to keep entropy values 40-60. Remifentanil was initially administered with a rate of 0.1 ┬Ąg/kg/min before induction and infused at 0.05-0.15 ┬Ąg/kg/min to keep hemodynamic variables suitable to induced hypotension (around systolic blood pressure 90-100 mmHg) in all patients. Muscle relaxant was only administered during the induction of anesthesia.
The remifentanil infusion and desflurane were stopped at the end of surgery. Intraoperative remifentanil infusion dose, end-tidal desflurane concentration (Et DES) at the end of surgery, and the duration of the surgery were recorded. Recovery profiles (the times from desflurane stop to eye opening and extubation) were also checked. To reverse the neuromuscular block, pyridostigmine 10 mg and glycopyrrolate 0.4 mg were intravenously administered when self-respiration was recovered. Extubation was performed when the patient obeyed commands and recovered a regular self-respiration.
In a postanesthesia care unit (PACU), a pain score was taken using a numeric rating scale (NRS) every 10 min for 1 h by another anesthesiologist, blinded to the treatment group. Postoperative pain (a NRS Ōēź 4) was controlled by repetitive intravenous injections of ketorolac 10 mg according to the NRS which was examined every 10 min in a PACU. In a ward, the same blinded anesthesiologist obtained pain scores using NRS at postoperative 6, 12, 18, and 24 h, and if patients complained of pain that required analgesics, ketorolac 30 mg was administered intravenously. No one who was involved in patient management or data collection were aware of the group assignment.
The time to first analgesic requirement and a ketorolac demand dose were assessed in a PACU and ward, respectively. Adverse events including nausea and vomiting, dizziness, headache, sedation, respiratory depression, sweating, dry mouth, tachycardia, hypertension, and restlessness were also assessed in a PACU and ward during the postoperative 24 h.
A primary endpoint in this study was the ketorolac demand dose in a PACU after surgery. A sample size calculation was based on a pilot study performed with 5 cases in each group. In the pilot study, a ketorolac demand dose in a PACU after surgery in Group N was 30% less than that in Group C. Therefore, assuming that nefopam would reduce a ketorolac demand dose by 30%, the required number of patients for each group was 22 (╬▒ = 0.05, power = 0.8). Considering a 10% drop-out rate, we enrolled 25 patients in each group, resulting in a total of 50 patients.
Statistical analyses were performed with SigmaStat software version 4.0 (Systat Software, Inc., Point Richmond, CA, USA). Student's t-test or Mann-Whitney U test was used to compare mean values of age, height, weight, body mass index, surgical time, total infusion time and dose of remifentanil, Et DES at the end of surgery, ketorolac demand doses and the times to first analgesic requirement in a PACU and ward, the times from desflurane stop to eye opening and extubation between the groups. Sex and incidences of adverse events during the postoperative 24 h between the groups were compared using a chi-squared test or Fisher's exact test. Pain scores, assessed using a NRS, in a PACU and ward were compared using a two-way repeated measures analysis of variance. The data are expressed as a mean ┬▒ standard deviation, median (range), or number of patients. A P value < 0.05 was considered statistically significant.
Of the 50 patients originally enrolled in the study, 6 patients were excluded for violations of the study protocol. The results from the remaining 44 patients were evaluated, with 22 patients finally included in Group N, and 22 patients in Group C (Fig. 1).
There were no significant differences in the surgical time, total infusion time, dose and rate of remifentanil, and Et DES at the end of surgery (Table 1). Ketorolac demand in the PACU was significantly lower in Group N than that in Group C (P = 0.005) and the time to the first analgesic requirement in the PACU was significantly longer in Group N than in Group C (P = 0.046).
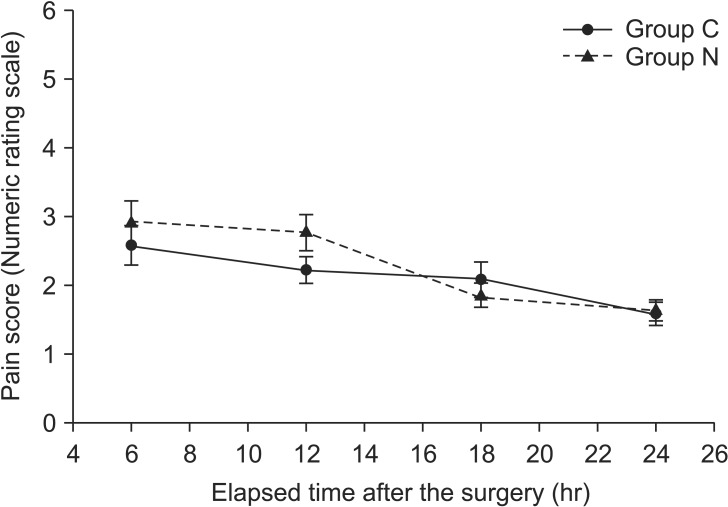
However, there were no significant differences in the time to first analgesic requirement and ketorolac demand in the ward between the groups (P = 0.608, P = 0.794, respectively). Eye opening time after surgery was significantly shorter in Group C than that in Group N (P = 0.027), but the extubation time was not different between the groups (P = 0.167, Table 2). The NRS score in the PACU was significantly lower in Group N than in Group C (P = 0.002, Fig. 2). But there was no significant difference in the NRS score in the ward between the groups (P = 0.494, Fig. 3). A pattern of change over time in the NRS score in both groups was similar in the PACU (P = 0.322, Fig. 2) and ward (P = 0.210, Fig. 3).
In descending order of incidence rate, nausea and vomiting, dizziness, and headache occurred as adverse events during the postoperative 24 h. But there were no significant differences between the groups regarding adverse events (Table 3).
In this study, the ketorolac demand dose was larger, the NRS score was higher, and the time to first analgesic requirement was shorter in Group C than in Group N in the PACU. These results indicate that remifentanil infusion under desflurane anesthesia may cause pain sensitization in the immediate postoperative period and thus require of the first postoperative analgesic earlier. In addition, the administration of nefopam combined with remifentanil infusion can reduce the degree of pain and decrease analgesic demand in the same period. Thus we suggest that nefopam may prevent remifentanil from increasing pain sensitivity, which may be caused by blocking central sensitization due to the inhibition of NMDA receptors.
We designed this study in consideration of the following several issues associated with patients undergoing middle ear surgeries. First, middle ear surgeries usually cause mild to moderate pain to patients [17]. Second, the surgeries usually require remifentanil infusion instead of nitrous oxide for a good surgical outcome and the induced hypotension associated with a good surgical field [18]. But, remifentanil infusion can develop paradoxical and severe pain after opioid administration [4]. Many studies have been conducted regarding the prevention of this severe pain due to remifentanil infusion. Continuous infusion of low dose ketamine, a non-competitive NMDA receptor antagonist decreased early postoperative pain by preventing remifentanil-induced hyperalgesia [9]. Pretreatment with pregabalin which is a close structure of gabapentin and wound infiltration with magnesium sulfate prevented OIH due to remifentanil by blocking NMDA receptors [10,19]. Nefopam is widely used as an analgesic agent for postoperative pain control and it is also a NMDA receptor antagonist [12]. Third, the surgeries are frequently associated with postoperative nausea and vomiting [20]. Therefore, considering the possibility of increased pain due to remifentanil-induced pronociceptive effects and a tendency to induce nausea and vomiting of due to the surgeries, we chose nefopam as a proper analgesic for middle ear surgery in this study. The dose of nefopam was set at 40 mg due to a concern about remifentanil-induced pronociceptive effect and a reference that the median effective analgesic dose of nefopam after moderately painful surgery was 28 mg [21]. As a result, nefopam administration combined with remifentanil infusion reduced pain and analgesic consumption in the immediate postoperative period, which indicates that nefopam may prevent the remifentanil-induced pronociceptive effect. Although several studies reported that OIH or acute opioid tolerance occurs more in cases of high-dose remifentanil infusion [8,22], a small dose of remifentanil infusion of effect-site target concentration 2 ng/ml (an infusion rate of 0.1 ┬Ąg/kg/min) could cause early postoperative pain increase [23]. Angst et al. [3] also reported that withdrawal after remifentanil exposure with an infusion rate of 0.1 ┬Ąg/kg/min (plasma concentration ranging between 2.7 and 2.9 ng/ml) increased the pain response compared to before remifentanil infusion. In this study, all patients were given a small dose of remifentanil infusion during the surgery and especially the patients of Group C were given a very small dose of 0.05 ┬▒ 0.03 ┬Ąg/kg/min. Nevertheless, interestingly, they complained of more pain and required higher analgesic dose associated with pain sensitization compared to the patients of Group N.
The exact mechanism of OIH or acute opioid tolerance has not been clearly demonstrated yet [24], although many reports mainly support that it may involve a central sensitization in the spinal cord through activation of dorsal horn NMDA systems [25,26]. Likewise, nefopam is a serotonin and catecholamine reuptake inhibitor [27] as well as a noncompetitive NMDA receptor antagonist [28]. Nefopam also directly interacts with ╬▒2-adrenoceptors [27]. Taken together, further detailed and complex studies will be needed to determine the exact mechanism of the analgesic effect of nefopam combined with remifentanil as well as the relation between remifentanil dosage and OIH or acute opioid tolerance.
In this study, eye opening time after surgery was significantly shorter in Group C than in Group N. This result may originate from the use of nefopam because there were no differences in the demographic and anesthetic data including the dose and rate of remifentanil, and Et DES at the end of surgery between the groups in our study. In light of some studies using other NMDA antagonists that reported that there were no differences in recovery times of the groups, one mechanism of action of nefopam may be associated with the delayed eye opening time, but it remains to be examined.
Adverse events associated with the drugs used in this study rarely uncommonly occurred. They were easily treatable and the incidences of them were comparable between the groups. In particular, adverse events associated with nefopam including sweating, dry mouth, tachycardia, hypertension, malaise, nausea or vomiting and restlessness were not observed or were only minor, in agreement with the study by Durrieu et al. [29].
A limitation in this study is that the reduced pain and analgesic consumption in Group N could have been caused by the analgesic effect of nefopam, and not its anti-pronociceptive effect, and a low dose of remifentanil infusion could not have induced OIH. The plasma half-life of nefopam ranges from 3 to 5 hours, and peak plasma concentrations are reached 15-20 min after intravenous injection [30]. In this study, the NRS score and the time to first analgesic requirement of Group N were significantly lower than those of Group C in the PACU (that is, in an immediate postoperative period) but not in the ward. The difference may be due to the analgesic effect of nefopam considering that its half-life of 3-5 h almost corresponded precisely with the time from the start of nefopam infusion to the patient's arrival in the ward. If we had designed another patient group which was given only desflurane anesthesia without remifentanil infusion in this study, we would have been able to identify a remifentanil-induced pronociceptive effect by comparing the desflurane only group and the desflurane with remifentanil group as some studies did [10,19].
Another limitation in this study is that it could not determine the most effective dose of nefopam for reduction of pain sensitized by remifentanil. Thus, further studies designed with different doses of nefopam groups will be needed to elucidate the optimal dosage.
In conclusion, nefopam administration combined with low dose remifentanil infusion reduces pain and analgesic consumption in an immediate postoperative period in patients undergoing middle ear surgery under desflurane anesthesia.
References
1. Mandel JE. Considerations for the use of short-acting opioids in general anesthesia. J Clin Anesth 2014; 26(1 Suppl): S1-S7. PMID: 24485553.


2. Thompson JP, Rowbotham DJ. Remifentanilan-- opioid for the 21st century. Br J Anaesth 1996; 76: 341-343. PMID: 8785129.



3. Angst MS, Koppert W, Pahl I, Clark DJ, Schmelz M. Short-term infusion of the mu-opioid agonist remifentanil in humans causes hyperalgesia during withdrawal. Pain 2003; 106: 49-57. PMID: 14581110.


4. Chu LF, Angst MS, Clark D. Opioid-induced hyperalgesia in humans: molecular mechanisms and clinical considerations. Clin J Pain 2008; 24: 479-496. PMID: 18574358.


5. Lee C, Kim YD, Kim JN. Antihyperalgesic effects of dexmedetomidine on high-dose remifentanil-induced hyperalgesia. Korean J Anesthesiol 2013; 64: 301-307. PMID: 23646238.



6. Lee H. Opioid-induced hyperalgesia is a paradox for perioperative physician. Korean J Anesthesiol 2013; 64: 1-2. PMID: 23372877.



7. Cho AR, Kim HK, Kim KH, Jung KY, Kim WS, Kwon JY. Effect of remifentanil on postoperative pain in gynecologic surgery with sevoflurane anesthesia. Korean J Anesthesiol 2008; 55: 182-189.

8. Wilder-Smith OH, Arendt-Nielsen L. Postoperative hyperalgesia: its clinical importance and relevance. Anesthesiology 2006; 104: 601-607. PMID: 16508408.


9. Hong BH, Lee WY, Kim YH, Yoon SH, Lee WH. Effects of intraoperative low dose ketamine on remifentanil-induced hyperalgesia in gynecologic surgery with sevoflurane anesthesia. Korean J Anesthesiol 2011; 61: 238-243. PMID: 22025947.



10. Lee C, Song YK, Jeong HM, Park SN. The effects of magnesium sulfate infiltration on perioperative opioid consumption and opioid-induced hyperalgesia in patients undergoing robot-assisted laparoscopic prostatectomy with remifentanil-based anesthesia. Korean J Anesthesiol 2011; 61: 244-250. PMID: 22025948.



11. Rosland JH, Hole K. The effect of nefopam and its enantiomers on the uptake of 5-hydroxytryptamine, noradrenaline and dopamine in crude rat brain synaptosomal preparations. J Pharm Pharmacol 1990; 42: 437-438. PMID: 1979627.


12. Novelli A, Diaz-Trelles R, Groppetti A, Fern├Īndez-S├Īnchez MT. Nefopam inhibits calcium influx, cGMP formation, and NMDA receptor-dependent neurotoxicity following activation of voltage sensitive calcium channels. Amino Acids 2005; 28: 183-191. PMID: 15714253.


13. Evans MS, Lysakowski C, Tram├©r MR. Nefopam for the prevention of postoperative pain: quantitative systematic review. Br J Anaesth 2008; 101: 610-617. PMID: 18796441.



14. Kim K, Kim WJ, Choi DK, Lee YK, Choi IC, Sim JY. The analgesic efficacy and safety of nefopam in patient-controlled analgesia after cardiac surgery: A randomized, double-blind, prospective study. J Int Med Res 2014; 42: 684-692. PMID: 24691459.


15. Tirault M, Derrode N, Clevenot D, Rolland D, Fletcher D, Debaene B. The effect of nefopam on morphine overconsumption induced by large-dose remifentanil during propofol anesthesia for major abdominal surgery. Anesth Analg 2006; 102: 110-117. PMID: 16368814.


16. Kim YM, Lim BG, Kim H, Kong MH, Lee MK, Lee IO. Slow injection of nefopam reduces pain intensity associated with intravenous injection: a prospective randomized trial. J Anesth 2014; 28: 399-406. PMID: 24201414.


17. Wittekindt D, Wittekindt C, Meissner W, Guntinas-Lichius O. Postoperative pain assessment after middle ear surgery. HNO 2012; 60: 974-984. PMID: 22767198.


18. Lim BG, Lim SH. Comparison of isoflurane-nitrous oxide anesthesia and isoflurane-remifentanil anesthesia during tympanoplasty. Korean J Anesthesiol 2008; 54: 152-159.

19. Jo HR, Chae YK, Kim YH, Chai HS, Lee WK, Choi SS, et al. Remifentanil-induced pronociceptive effect and its prevention with pregabalin. Korean J Anesthesiol 2011; 60: 198-204. PMID: 21490822.



20. Yoon DG, Jung SH, Ha MH, Song NW. Comparison of ramosetron and ondansetron for preventing nausea and vomiting after middle ear surgery under general anesthesia with sevoflurane and remifentanil. Korean J Anesthesiol 2009; 56: 408-412.


21. Delage N, Maaliki H, Beloeil H, Benhamou D, Mazoit JX. Effective dose (ED50) of nefopam and ketoprofen in postoperative patients: a study of interaction using sequential analysis and isobolographic analysis. Anesthesiology 2005; 102: 1211-1216. PMID: 15915035.


22. Shin SW, Cho AR, Lee HJ, Kim HJ, Byeon GJ, Yoon JW, et al. Maintenance anaesthetics during remifentanil-based anaesthesia might affect postoperative pain control after breast cancer surgery. Br J Anaesth 2010; 105: 661-667. PMID: 20876698.



23. Vinik HR, Kissin I. Rapid development of tolerance to analgesia during remifentanil infusion in humans. Anesth Analg 1998; 86: 1307-1311. PMID: 9620525.


24. Borgland SL. Acute opioid receptor desensitization and tolerance: is there a link? Clin Exp Pharmacol Physiol 2001; 28: 147-154. PMID: 11207668.


25. Jordan B, Devi LA. Molecular mechanisms of opioid receptor signal transduction. Br J Anaesth 1998; 81: 12-19. PMID: 9771268.



26. Kissin I, Bright CA, Bradley EL Jr. Acute tolerance to continuously infused alfentanil: the role of cholecystokinin and N-methyl-D-aspartatenitric oxide systems. Anesth Analg 2000; 91: 110-116. PMID: 10866896.


27. Girard P, Coppe MC, Verniers D, Pansart Y, Gillardin JM. Role of catecholamines and serotonin receptor subtypes in nefopam-induced antinociception. Pharmacol Res 2006; 54: 195-202. PMID: 16750379.


28. Fernandez-Sanchez MT, Diaz-Trelles R, Groppetti A, Manfredi B, Brini AT, Biella G, et al. Nefopam, an analogue of orphenadrine, protects against both NMDA receptor-dependent and independent veratridine-induced neurotoxicity. Amino Acids 2002; 23: 31-36. PMID: 12373515.


29. Durrieu G, Olivier P, Bagheri H, Montastruc JL. Overview of adverse reactions to nefopam: an analysis of the French Pharmacovigilance database. Fundam Clin Pharmacol 2007; 21: 555-558. PMID: 17868209.


30. Aymard G, Warot D, D├®molis P, Giudicelli JF, Lechat P, Le Guern ME, et al. Comparative pharmacokinetics and pharmacodynamics of intravenous and oral nefopam in healthy volunteers. Pharmacol Toxicol 2003; 92: 279-286. PMID: 12787260.


Fig.┬Ā1
A flow chart describing patient recruitment, randomization, and withdrawal. Initially, 50 patients were randomly assigned to either the nefopam group which was given nefopam together with remifentanil 5 min before anesthesia induction (Group N) or the control group which was given only remifentanil 5 min before anesthesia induction (Group C). Finally, 44 patients (22 in Group N and 22 in Group C) completed this study.
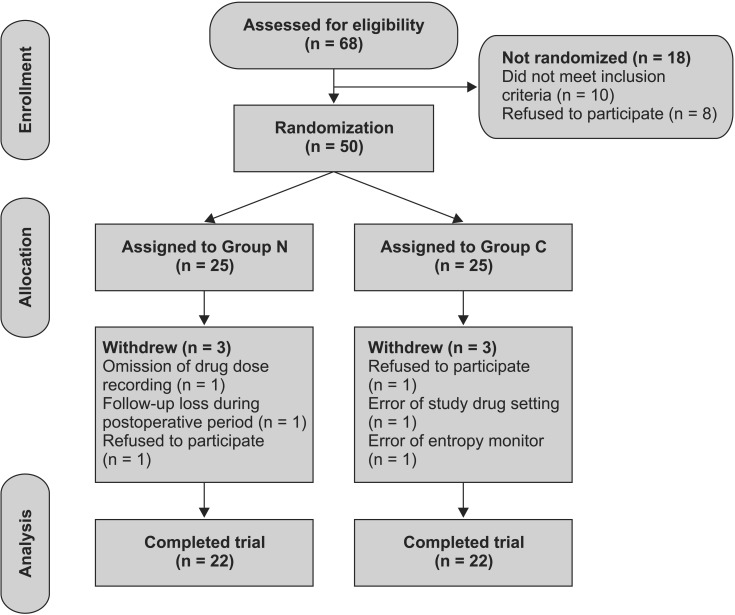
Fig.┬Ā2
The numeric rating scale (NRS) for pain in the postanesthesia care unit. A similar pattern of changes over time in the NRS was observed in both groups (P = 0.322). There was a significant difference between Group N and Group C (P = 0.002). Group C: the control group which was given only remifentanil 5 min before anesthesia induction. Group N: the nefopam group which was given nefopam together with remifentanil 5 min before anesthesia induction.
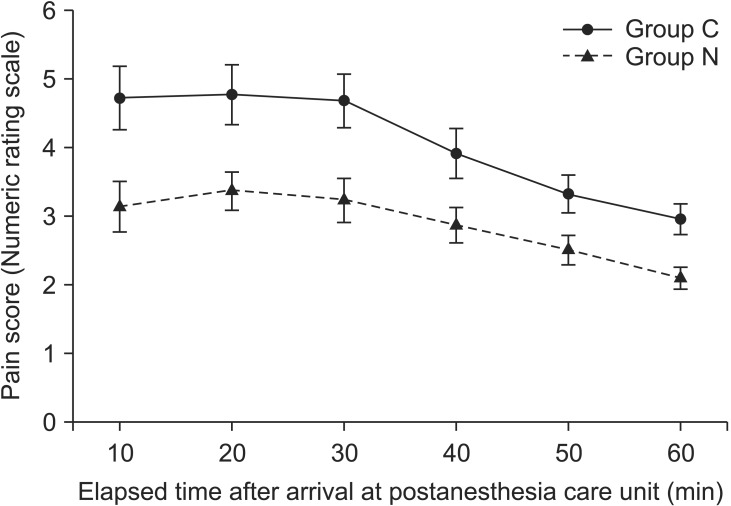
Fig.┬Ā3
The numeric rating scale (NRS) for pain in the ward. A similar pattern of changes over time in the NRS was observed in both groups (P = 0.210). There was no significant difference between Group N and Group C (P = 0.494). Group C: the control group which was given only remifentanil 5 min before anesthesia induction. Group N: the nefopam group which was given nefopam together with remifentanil 5 min before anesthesia induction.
Table┬Ā1
Demographic and Clinical Data
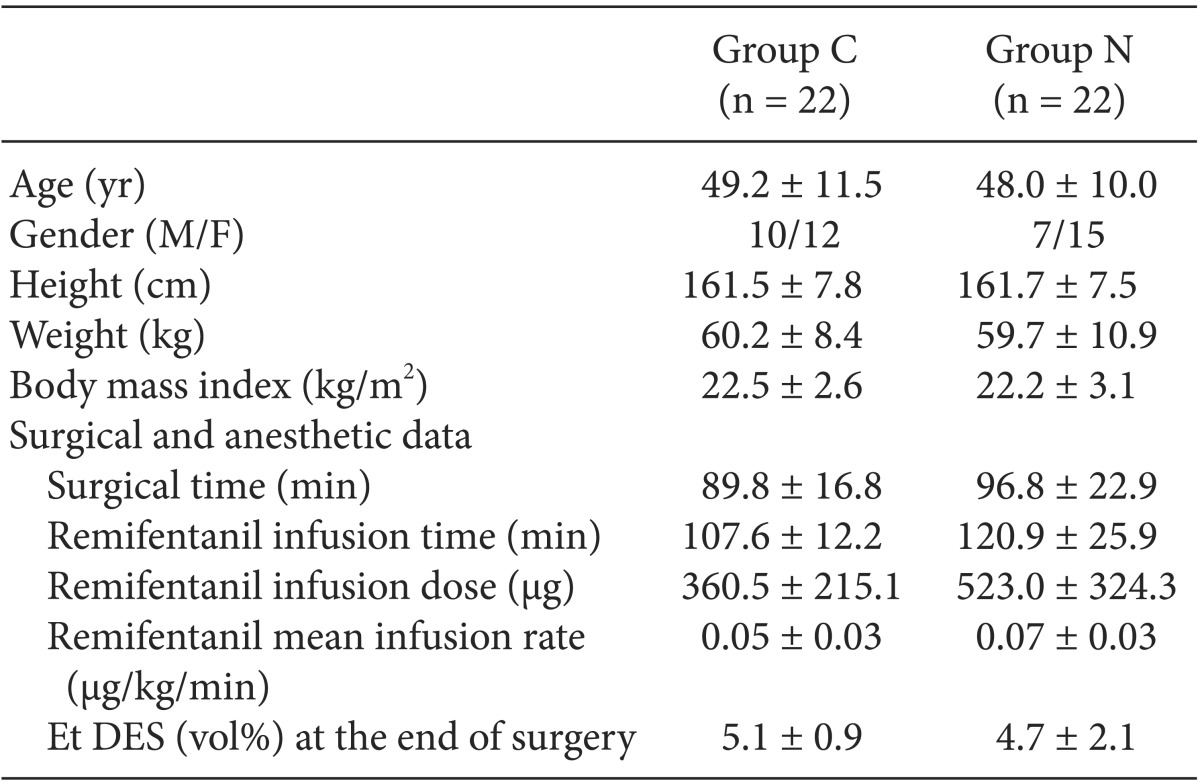
Values are mean ┬▒ SD or number of patients. There were no significant differences between the two groups. Group C: the control group which was given only remifentanil 5 min before anesthesia induction. Group N: the nefopam group which was given nefopam together with remifentanil 5 min before anesthesia induction. Et DES: end-tidal desflurane concentration.
Table┬Ā2
Recovery Profiles and Analgesic Profiles in the PACU and Ward
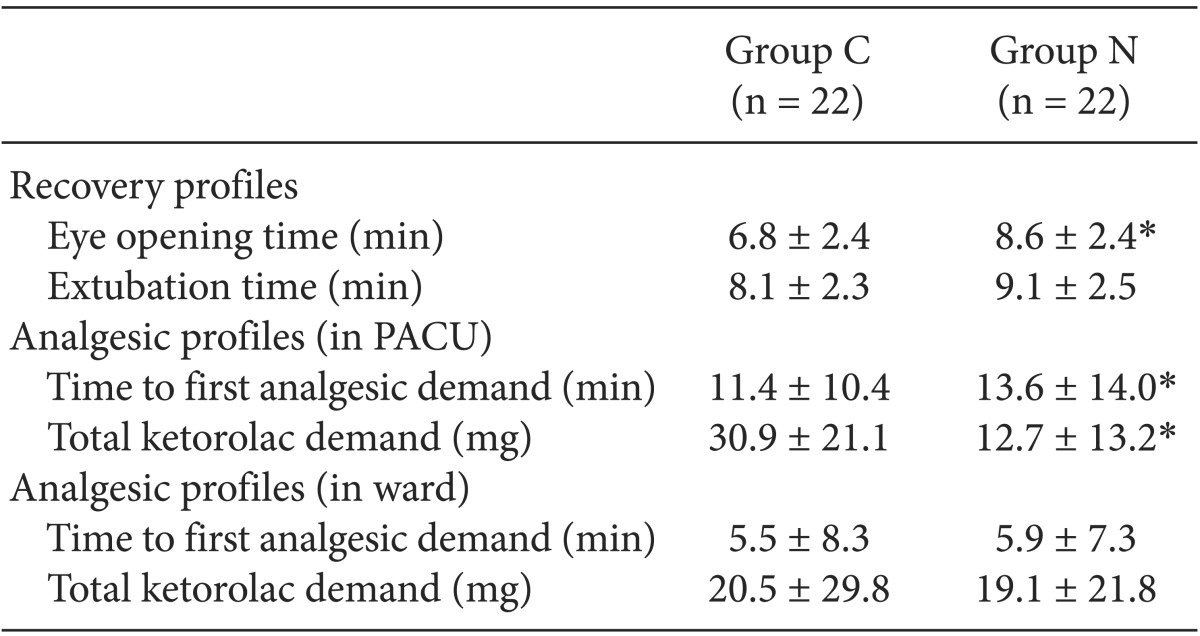
Values are mean ┬▒ SD or number of patients. Group C: the control group which was given only remifentanil 5 min before anesthesia induction. Group N: the nefopam group which was given nefopam together with remifentanil 5 min before anesthesia induction. PACU: postanesthesia care unit. *P < 0.05 vs. group C.
- TOOLS