|
 |
|
|
Abstract
Ethanol sclerotherapy for the treatment of low-flow vascular malformations can cause catastrophic cardiopulmonary complications, including pulmonary embolism and pulmonary hypertension, that can result in right heart failure and fatal arrhythmias, leading to death. We here report a case of abrupt cardiovascular collapse that developed immediately following ethanol sclerotherapy in 31-year-old female patient who had a large arteriovenous malformation in her leg. Anesthesiologists should be aware of the fatal cardiopulmonary complications that are associated with ethanol sclerotherapy and consider the use of invasive hemodynamic monitoring, such as pulmonary artery pressure monitoring, when large doses of ethanol are required.
Sclerotherapy with alcohol is the preferred therapy for low-flow arteriovenous malformations (AVM) because it is the most effective, rarely demonstrating recurrence, and is associated with few complications [1,2,3,4]. However, ethanol sclerotherapy can produce minor local complications such as skin blistering, ulcerations, scar formation, and local nerve damage [5,6]. Furthermore, ethanol sclerotherapy may cause lethal cardiopulmonary complications including pulmonary embolism, pulmonary hypertension, and cardiac arrhythmias that may lead to cardiovascular collapse or even death [4,5,7]. Here, we report the case of a 31-year-old female patient who was resuscitated after sudden cardiovascular collapse immediately following ethanol sclerotherapy.
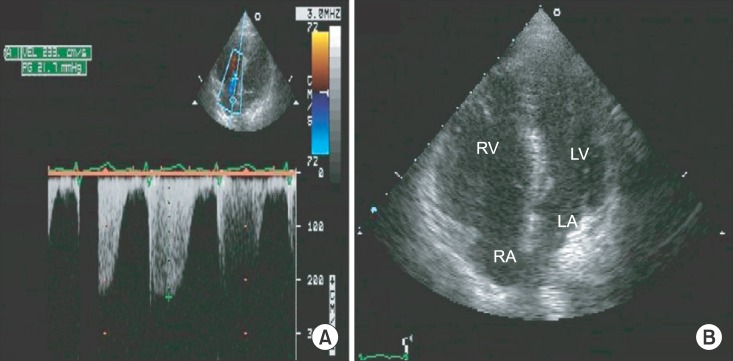
A 31-year-old female patient with a height of 164 cm and weight of 60 kg was admitted to undergo ethanol sclerotherapy for a large painful vascular malformation in her left leg. Magnetic resonance imaging revealed an AVM measuring 28 × 7 × 4 cm in her left calf (Fig. 1). All preoperative examinations, including laboratory tests, electrocardiography, and chest X-ray, were normal. Vital signs before general anesthesia were stable: her blood pressure was 130/80 mmHg and her heart rate was 82 beats/min. Following the application of routine hemodynamic monitoring (3-lead electrocardiogram, noninvasive blood pressure, and pulse oximetry), anesthesia was induced using a bolus intravenous injection of 120 mg propofol and 40 mg rocuronium. A 20-gauge catheter was inserted into the radial artery to monitor her arterial blood pressure continuously. The target controlled infusion of 2% propofol and remifentanil (2-3 µg/ml and 2-5 ng/ml of the effect-site concentrations, respectively) was applied to maintain anesthesia. Immediately following the injection of ethanol, the patient's blood pressure and heart rate rose slightly to 160/100 mmHg and 100 beats/min, respectively. Thereafter, the patient's blood pressure and heart rate were stable throughout the procedure while she was in the prone position. A total dose of 50 ml of 99% ethanol was injected into the lesion over a period of 30 minutes without the use of a tourniquet. As soon as the patient was turned from prone to supine after finishing the procedure, an abrupt drop in her arterial blood pressure occurred, 40-50 mmHg systolic and 10-15 mmHg diastolic blood pressure with a simultaneous increase in her heart rate to 100-110 beats/min. To restore blood pressure, 20 mg of ephedrine was immediately injected, but the patient's heart rate rather decreased abruptly to < 30 beats/min. Chest compressions were performed along with the simultaneous injection of 50 µg of epinephrine. A few minutes later, the patient's blood pressure and heart rate recovered to 180/100 mmHg and 100-110 beats/min. At that moment, the arterial blood gas analysis showed metabolic acidosis with otherwise normal findings (pH 7.29, PaCO2 28 mmHg, PaO2 466 mmHg, SaO2 100%, base excess -11.6, Hematocrit 38%, Na+ 137 mEq/l, K+ 3.7 mEq/l, and Ca2+ 1.11 mmol/l). The patient was transferred to the intensive care unit with the endotracheal tube in place. Transthoracic echocardiograph (TTE) and embolism computed tomography (CT) were performed to confirm pulmonary hypertension or pulmonary embolism. There was no evidence of pulmonary embolism on CT. In addition, the peak tricuspid regurgitation jet velocity on TTE was 2.3 m/s, indicating no evidence of pulmonary hypertension at that moment (Fig. 2A). However, TTE revealed dilated right ventricle (Fig. 2B) and severely decreased right ventricular function.
On postoperative day 1, the patient regained her consciousness and was discharged five days after the operation without any complications.
Sclerotherapy is now accepted as the treatment of choice for AVM [6,8,9]. Several sclerosants have been developed but absolute ethanol is favored over other embolic agents because of its relatively good results and low chance of recanalization [2,10]. Despite its benefits, however, ethanol sclerotherapy frequently produces minor local complications, including tissue necrosis, peripheral nerve injuries, skin blisterings, and ulcers [5,6]. Moreover, if ethanol is absorbed into systemic circulation, it can cause fatal complications, including acute alcohol intoxication, red blood cell hemolysis, pulmonary embolism, pulmonary vasospasm that may lead to right heart failure, cardiac arrhythmias due to changes in the cardiac conduction system and even death in severe cases [4,5,7].
According to a previous study, systemic ethanol contamination during sclerotherapy occurs in over 80% of cases [1]. The plasma ethanol level is directly proportional to the amount of ethanol injected but is independent of the AVM morphology, venous drainage, and injection technique [1]. The pulmonary arterial ethanol level is correlated with a single dose of ethanol, and the pulmonary arterial pressure (PAP) immediately elevates following a bolus injection of ethanol [11]. Ultimately, larger doses of ethanol result in more systemic complications. The accepted practice over the past few years has been to limit the total volume of ethanol administered over a procedure to < l ml/kg [2].
In our present case, 50 ml of 99% ethanol was injected into a 60 kg patient. The total dose of ethanol that was injected was nearly the maximum dose allowable for this patient. The large amount of ethanol that was injected might have carried a potential risk of cardiovascular compromise, or the right heart failure was caused by the acute development of severe pulmonary hypertension. We could not find any direct evidence of pulmonary hypertension because the normal peak tricuspid regurgitation jet velocity on our TTE examination was observed. Nevertheless, we speculate that the right heart failure mainly contributed to the development of cardiovascular collapse in this case, that is, a systematically absorbed ethanol may result in severe pulmonary hypertension acutely, leading to right heart failure which was inferred from a dilated right ventricle and severely poor right ventricular function on TTE examination. The normal peak tricuspid regurgitation jet velocity would likely manifest due to right ventricle dilatation which persisted as the trace of acute severe pulmonary hypertension. As a result of right heart failure, left ventricular filling and output decreased, eventually leading to cardiovascular collapse which necessitated the prompt resuscitation.
To minimize the systemic absorption of alcohol and maximize its sclerosing effects, operators have developed several new techniques, such as the occlusive method that maintains alcohol within the lesion [12]. However, the same risks still exist [13]. Invasive hemodynamic monitoring such as PAP monitoring recommended when a large amount of alcohol is expected to be used [5,13]. In the case of increased PAP, PAP-lowering agents, including nitroglycerine, inhaled nitric oxide, prostaglandin E1 or prostaglandin I2, should be considered [4].
In conclusion, we report the patient who was resuscitated from cardiovascular collapse that was probably due to acute right heart failure following ethanol sclerotherapy. Anesthesiologists should always pay attention to the total dose of ethanol administered, not exceeding 1 ml/kg, during this type of treatment. When a large amount of alcohol is expected to be injected, the anesthesiologist should consider using invasive PAP monitoring and preparing PAP-lowering agent to treat acute pulmonary hypertension and make thorough preparation for managing severe cardiovascular complications.
References
1. Hammer FD, Boon LM, Mathurin P, Vanwijck RR. Ethanol sclerotherapy of venous malformations: evaluation of systemic ethanol contamination. J Vasc Interv Radiol 2001; 12: 595-600. PMID: 11340138.


2. Yakes WF, Rossi P, Odink H. How I do it. Arteriovenous malformation management. Cardiovasc Intervent Radiol 1996; 19: 65-71. PMID: 8729181.


3. Doppman JL, Pevsner P. Embolization of arteriovenous malformations by direct percutaneous puncture. AJR Am J Roentgenol 1983; 140: 773-778. PMID: 6601389.


4. Shin BS, Kim CS, Lee BD, Sim WS, Kang JG, Kim HS. Anesthetic management of pulmonary arterial pressure change during the ethanol sclerotherapy of peripheral arteriovenous malformations. Korean J Anesthesiol 2004; 47: 356-360.

5. Mitchell SE, Shah AM, Schwengel D. Pulmonary artery pressure changes during ethanol embolization procedures to treat vascular malformations: can cardiovascular collapse be predicted? J Vasc Interv Radiol 2006; 17: 253-262. PMID: 16517770.


6. Berenguer B, Burrows PE, Zurakowski D, Mulliken JB. Sclerotherapy of craniofacial venous malformations: complications and results. Plast Reconstr Surg 1999; 104: 1-11. PMID: 10597669.


7. Gelczer RK, Charboneau JW, Hussain S, Brown DL. Complications of percutaneous ethanol ablation. J Ultrasound Med 1998; 17: 531-533. PMID: 9697961.


8. Yakes WF, Luethke JM, Parker SH, Stavros AT, Rak KM, Hopper KD, et al. Ethanol embolization of vascular malformations. Radiographics 1990; 10: 787-796. PMID: 2217971.


9. Jin Y, Lin X, Li W, Hu X, Ma G, Wang W. Sclerotherapy after embolization of draining vein: a safe treatment method for venous malformations. J Vasc Surg 2008; 47: 1292-1299. PMID: 18387776.


10. Do YS, Yakes WF, Shin SW, Lee BB, Kim DI, Liu WC, et al. Ethanol embolization of arteriovenous malformations: interim results. Radiology 2005; 235: 674-682. PMID: 15858106.


11. Ko JS, Kim JA, Do YS, Kwon MA, Choi SJ, Gwak MS, et al. Prediction of the effect of injected ethanol on pulmonary arterial pressure during sclerotherapy of arteriovenous malformations: relationship with dose of ethanol. J Vasc Interv Radiol 2009; 20: 39-45. PMID: 19028113.


12. Ierardi AM, Mangini M, Vaghi M, Cazzulani A, Carrafiello G, Mattassi R. Sclerotherapy of peripheral venous malformations: a new technique to prevent serious complications. Vasc Endovascular Surg 2010; 44: 282-288. PMID: 20403950.


13. Ko JS, Kim CS, Shin BS, Kim MJ, Lee JH, Kim KH, et al. Changes in pulmonary artery pressures during ethanol sclerotherapy for arteriovenous malformations: identifying the most vulnerable period. Clin Radiol 2011; 66: 639-644. PMID: 21513926.


- TOOLS