The incidence of pneumothorax in association with laparoscopic surgery has been reported to vary between 0.01-3% [1-3]. However, many difficulties present in anesthetized patients during laparoscopic surgery because of increased end-tidal carbon dioxide (ETCO2) and maximal airway pressure (Pmax) levels. In contrast with pneumothorax caused by various kinds of lung injury, capnothorax that occurs due to CO2-pneumoperitoneum often resolves spontaneously within a short period of time [2,3]. Therefore, if capnothorax can be diagnosed during laparoscopic surgery, the insertion of a chest tube may not be required, which avoids another invasive procedure. Here, we report two cases in which capnothorax were diagnosed at the bedside using ultrasonography. Spontaneous resolution of this condition was observed using ultrasonography, most particularly the verification of the "lung point" sign.
Case Reports
Case 1
A 40-year-old woman, 169 cm, 57 kg, underwent laparoscopic right adrenalectomy due to an adrenal pheochromocytoma. Preoperative evaluation of the patient was unremarkable. General anesthesia was induced using thiopental sodium, fentanyl, vecuronium and sevoflurane. A 7.5 Fr 3-lumen catheter was inserted under ultrasonographic guidance (linear probe: UST-5546, 5-10 MHz, machine: Prosound SSD-4400®, Aloka Medical, Co., Ltd., Tokyo, Japan) into the right internal jugular vein, and lung sliding was confirmed in both thoraces. Next, the patient was placed into the left lateral decubitus position. Before incision, ETCO2, Pmax, and peripheral oxygen saturation (SpO2) were determined to be 33 mmHg, 17 cmH2O, and 100%, respectively. Pneumoperitoneum was established using CO2, intraabdominal pressure was maintained < 15 mmHg, and Pmax was 23 cmH2O at the time. Thirty minutes after inducing pneumoperitoneum, the ETCO2 and Pmax levels gradually increased to 43 mmHg and 30 cmH2O, respectively. Thereafter, SpO2 decreased acutely to 92%. On auscultation, the breath sounds from the left lung field were normal. However, the right side of the chest demonstrated no sounds. Immediately, the fraction of inspired oxygen (FiO2) increased from 0.5 to 0.8, and the endotracheal tube was withdrawn back to 2 cm in order to rule out endobronchial intubation. Because the depth of the endotracheal tube was 18 cm at the incisor teeth and confirmed the depth of endotracheal tube by cuff palpation at the sternal notch, we supposed that it was enough to rule out one-lung intubation without using bronchoscope. SpO2 was maintained at between 98-100%, though the right thorax was still silent. Pneumothorax was strongly suspected, and the chest was examined using ultrasonography. Lung sliding and seashore sign were demonstrated in the left lung (Fig. 1A), however there was no sliding with barcode sign in the right lung (Fig. 1B). Although the surgeon simultaneously inspected the diaphragm in detail, no apparent injuries or defects were found. Because the vital signs were stable and SpO2 and ETCO2 were normal, as indicated by an increased FiO2 and respiratory rate, the operation was laparoscopically completed.
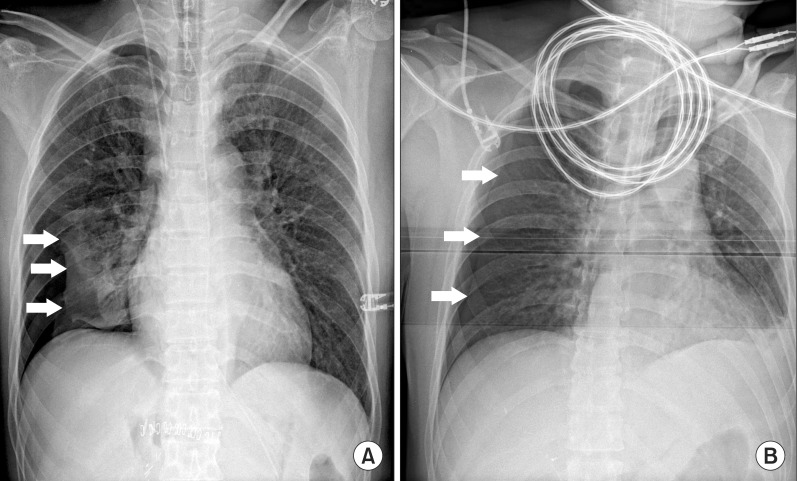
When the operation was concluded, a positive end-expiratory pressure (PEEP) of 5 cmH2O was applied to inflate the collapsed lung. The patient was examined using ultrasonography whilst in the supine position, and at the same time, portable chest radiography was called to assess a pneumothroax. The ultrasonographic probe was placed on the right lower lateral chest wall between anterior and mid axillary line (Fig. 2), and we identified the "lung point" sign, which can be used to diagnose pneumothorax [4]. Also, chest radiography indicated a < 30% collapse of the right lung (Fig. 3A). As the right hemithorax was continuously examined using ultrasonography, auscultation of the right lung improved, as well as the appearance of lung sliding sign and the disappearance of the lung point sign in whole right hemithorax within about 30 minutes after the end of the operation. No hemodynamic instability was noted, and SpO2 was maintained at 100% using only medical air. Hence, pneumthorax was considered to be capnothorax, and the patient was extubated without the insertion of a chest tube. The patient was transferred to the postanesthetic care unit (PACU) and did not complain about any respiratory disturbance.
Case 2
A 39-year-old man, 165 cm, 63 kg, was scheduled for living right donor nephrectomy via hand-assisted laparoscopic surgery (HALS). The patient was normal on preoperative evaluation and presented without a specific medical history. The induction of anesthesia with thiopental sodium, fentanyl, vecuronium and desflurane was uneventful. The patient was placed in the left lateral decubitus position. After CO2-pneumoperitoneum was induced, the intraabdominal pressure was maintained < 14 mmHg, and ETCO2, Pmax, and SpO2 were 33 mmHg, 23 cmH2O and 100%, respectively. When the upper pole of the kidney was approached, the diaphragm was injured by the dissector, which was immediately repaired. The surgeon inquired about the status of the right lung because the right hemidiaphragm was bulging. The lung sounds in the right chest were decreased on auscultation, and ETCO2 and Pmax were increased to 42 mmHg and 30 cmH2O, respectively. However, SpO2 was maintained at 100%. At that point, we evaluated the right thorax using ultrasonography (linear probe: L25x, 6-12 MHz, machine: M-turbo System®, Sonosite Inc., Bothell, WA, USA) in the operative field, and the "lung point" sign was found at the right lower anterior axillary line (Fig. 1C). Considering the location of the lung point sign, a small amount of pneumothorax was expected. However, because the hemodynamic parameters were maintained in a normal range, the operation was continued via HALS and a PEEP of 5 cmH2O was applied.
At the end of the surgery, the surgeon requested a portable chest radiography and we concurrently examined the patient using ultrasonography in the supine position. Chest radiography indicated a pneumothorax < 20% (Fig. 3B), and the range of the pneumothorax which was indicated by the lung point sign was gradually diminished. Because signs of cardiopulmonary instability were not found and SpO2 was maintained without an increase in FiO2, the insertion of a chest tube was not necessary and we safely extubated the endotracheal tube. The patient was transferred to the PACU and did not complain of any respiratory discomfort.
Discussion
Patients under general anesthesia may develop pneumothorax due to a variety of reasons, such as barotraumas, rupture of the lung bullae or needle insertion into the pleural cavity [3,5]. Capnothorax due to CO2 during laparoscopic surgery is rare, but, it has been described as a complication of almost every type of laparoscopic surgery [1-3,6-8]. The exact etiology of capnothorax still remains obscure. It has been speculated that an abnormal diaphragm caused by congenital failure of fusion (e.g., sternal, costal, and lumbar portions), weak point (e.g., around the aorta, vena cava, and esophagus), congenital defects or iatrogenic injuries by the instrument can lead to capnothorax [1-3,6]. Because the creation of pneumoperitoneum and positional changes can demonstrate several potential physiological alterations [9], the accurate diagnosis of pneumothorax is more difficult during laparoscopic surgery than other surgical procedure.
When Pneumothorax is suspected, it is traditionally diagnosed by portable chest radiography in the operating room. However, chest radiography may take a considerable amount of time and demonstrates a sensitivity of 31.8-75.5% for the diagnosis of pneumothorax [10-12]. In many different fields, lung ultrasonography is often used to diagnose pathological conditions of the lung. In particular, ultrasonography has received attention as a very effective method for confirming pneumothorax at the bedside in the operating room.
In numerous studies, ultrasonography has been reported as superior to conventional portable chest radiography for the detection of pneumothorax [4,10-13]. The presence or absence of characteristic ultrasonographic signs are key to a correct diagnosis, i.e., lung sliding, lung pulse, comet-tail artifact, barcode sign (stratosphere sign), seashore sign and lung point sign. Among these signs, lung sliding may be most widely known. It is shimmery horizontal movement of pleural line between two shadows of ribs [13]. However, the absence of lung sliding does not always indicate pneumothorax due to several mechanical factors (e.g., poor technique, inappropriate ultrasound probe frequency, the presence of dynamic noise filters) and the patient's affliction (e.g., main bronchial intubation, emphysema, atelectasis, pleural calcification, pleural adhesion, lung fibrosis) [13,14]. For these reasons, more specific ultrasonographic methods are needed to confirm pneumothorax.
In a particular area of the thorax, the ultrasonographic patterns of pneumothorax and the normal lung alternatively appear depending on the respiratory cycle. This is referred to as the "lung point" sign. This can be explained by a slight increase in pulmonary volume during inspiration and can be observed in two ultrasonographic modes: real-time (B-mode) and time-motion (M-mode) [4]. In B-mode, the lung point sign can be found by observing lung-sliding and absent of lung-sliding alternately at a same pleural line. Analogous patterns also can be found in M-mode. Seashore sign and barcode sign can be observed in turn, and these signs demonstrate the specificity of 100% for the diagnosis of pneumothorax [4]. Furthermore, by serially observing the location of the sign, the extension of pneumothorax may be identified. Despite this wide spread practice, there are few studies about the relationship between the lung point sign and the extension of pneumothorax. When it moves to the lower lateral chest wall, the volume of pneumothorax expands and the patient may need to have a chest tube immediately inserted. On the other hand, movement of the sign from the lower lateral chest to the anterior chest indicates an improvement in pneumothorax (Fig. 3). If capnothorax is highly suspected and cardiopulmonary instability is not severe, alteration of the "lung point" sign can be used to identify improvements in pneumothorax. In addition, for faster recovery of capnothorax, application of PEEP is supportive method to absorb CO2 more readily from the pleural cavity [15]. Therefore, a chest tube insertion, which can cause injury to other structures in the thorax, may not be necessary in these cases.
In conclusion, the lung point sign may predict the range and amount of pneumothorax during laparoscopic surgery. Ultrasonography can not only help to diagnose pneumothorax, but can also be used to decide treatment of pneumothorax. Most notably, in the case of capnothorax which resolves spontaneously as time goes on, the unnecessary insertion of a chest tube may be avoided by observing alterations in the lung point sign.