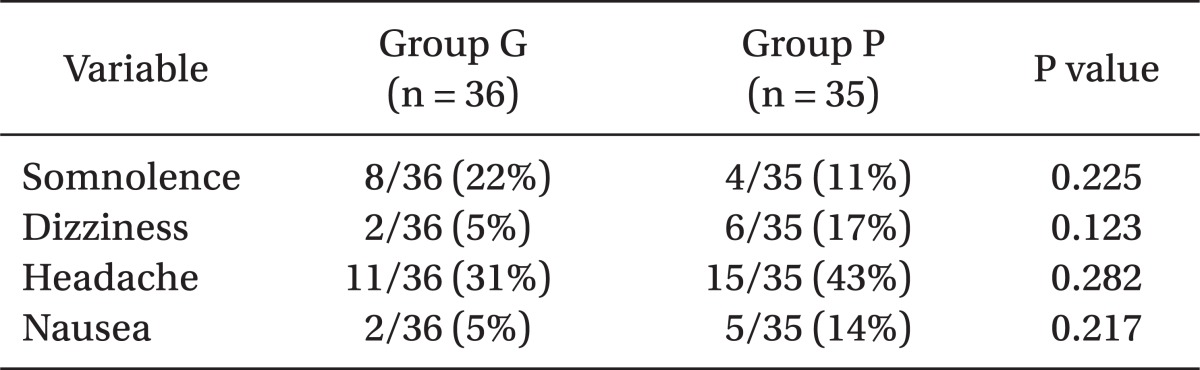|
 |
|
|
Abstract
Background
Postoperative sore throat (POST) is considered a usual complication after tracheal intubation, especially, thyroid surgery. Gabapentin is a widely studied multimodal perioperative drug, which can be used to treat acute postoperative pain. The primary endpoints of this study was a reduction of the incidence of POST at rest and during the swallowing movements after thyroid surgery. And the second endpoints was a reduction of the intensity of the POST after thyroid surgery.
Methods
Seventy-one patients that underwent elective thyroid surgery received either gabapentin (Neurontin™ 600 mg) or placebo, orally, one hour before anesthesia. The VAS scores and incidences of POST and adverse effects were determined at 1 hr, 6 hr, 12 hr, and 24 hr after surgery at rest and during swallowing movement.
Results
The gabapentin group (N = 36) showed a lower incidence of POST than the placebo group (N = 35) (47% vs. 78%, P = 0.038), and had significant lower VAS score at 6 and 24 hours after surgery in the resting state. However, during the movement, no intergroup differences were found in terms of the incidence of POST (83% vs. 91%, P = 0.305) or VAS score. Furthermore, no significant difference was observed between the two groups, in adverse effects.
Conclusions
Gabapentin (Neurontin™ 600 mg) administered 1 hr before anesthesia reduced the intensity and incidence of POST during the resting state without a significant adverse event, during the 24 hr after thyroid surgery. However, gabapentin did not reduce the intensity and incidence of POST during the swallowing movement.
Postoperative sore throat (POST) is not an uncommon complaint, and has been described as one of the most undesirable outcomes during the postoperative period, because it adversely affects the patient satisfaction and activities after discharge from hospital [1,2]. Furthermore, POST is considered to be more common and more serious after thyroid surgery [3,4], and it was assumed that the movement of the endotracheal tube was during the positioning and manipulation of the thyroid mass.
Gabapentin was introduced in 1993 as an adjuvant anticonvulsant for the treatment of refractory partial seizure. Recently, gabapentin is widely used as a multimodal perioperative drug. Many clinical trials have evaluated the potential roles for gabapentin, such as, postoperative analgesia, preoperative anxiolysis, the prevention of chronic post surgical pain, the attenuation of hemodynamic response to direct laryngoscopy and intubation, and the prevention of postoperative nausea, vomiting and delirium [5-10]. These studies address the important areas in the anesthetic field, and adequately demonstrated the multimodal effects of gabapentin.
We hypothesized that prophylactic administration of gabapentin would be effective in reducing POST. The aim of this prospective, randomized, double-blind study was to evaluate the prophylactic effect of gabapentin on reducing POST in patients during 24 hr after thyroid surgery.
This was a single-center, prospective, randomized, double-blinded study, in which the prophylactic effect of gabapentin on POST was investigated in patients scheduled for total or hemi-total thyroidectomy. The Institutional Review Board approved the study protocol and written informed consent was obtained from all participants.
The patients (aged 20-65 years), were of American Society of Anesthesiologists physical status of I or II, and were scheduled to receive total or hemi-total thyroidectomy for thyroid cancer. Patients meeting any of the following conditions were excluded; preexisting sore throat and hoarseness, corticosteroid use, or respiratory disease. Patients were randomly assigned, using a computer-generated random table, either to orally receive a gabapentin 600 mg (Neurontin™, Pfizer, Goedecke, Germany) 1 hour before the induction of anesthesia (Group G, N = 40) or placebo (Group C, N = 40). The participating anesthesiologist and the investigators that collected the postoperative data were unware of randomization process and the identities of study drug. After applying the exclusion criteria, mentioned below, nine of the 80 patients were excluded from this study; four from the gabapentin group and five for the placebo group.
Patients were not premedicated on the day of surgery. Anesthesia was induced with propofol (Pofol®, Jeil Pharmacy, Korea; 1-1.5 mg/kg) and remifentanil (Ultiva®, GSK, UK; 1 µg/kg), and tracheal intubation was facilitated with rocuronium (Esmeron®, Organon, the Netherlands; 0.6 mg/kg). After the loss of all four twitches from train-of-four stimulation of the ulnar nerve, direct laryngoscopy was performed, using either a Machintosh 3 or 4 laryngoscope blade, and an endotracheal tube was inserted. Female patients received a 7.0 mm diameter tube, while male patients received an 8.0 mm diameter tube. The tube positions were confirmed by auscultation, and the intra-cuff pressures of endotracheal tubes were measured using a noninvasive manometer (VBM, Sulz, Germany), every 30 min and maintained less than 20 cm H2O during procedures. Anesthesia was maintained with 50% oxygen in air, at a total flow of 2 L/min, containing sevoflurane (Abbot, UK), and with remifentanil infusion (0.1-0.5 µg/min/kg), and rocuronium as needed. Tidal volumes were 8-10 ml/kg and respiratory rates were 8-12/min, and end-tidal CO2 was maintained at 35-40 mmHg. At the end of surgery, a residual neuromuscular blockade was antagonized with glycopyrrolate (Tabinul®, Hana Pharmacy, Seoul; 0.2 mg IV) and pyridostigmine (Phygmin®, Hana Pharmacy, Seoul; 10 mg IV). In addition, to prevent postoperative nausea, all patients were administrated ramosetron 0.2 mg (Nasea®, Astellas, Japan). All patients were extubated without endotracheal suctioning and visible saliva around the oropharyngeal area was carefully suctioned to avoid tissue trauma. Patients received more than two time's attempts of intubation, and those with blood-tinged endotracheal tube, after extubation, were excluded in this trial. All of that anesthesia were carried out by one of the anesthesiologist, who was not aware of this trial.
No patients received PCA (patient controlled analgesia), and all were orally administered acetaminophen (Acetaminophen®, Samnom, Korea) 900 mg at 6 hours after surgery, and followed up on 8 hours interval. In addition, on patient's request, tramadol 50 mg (Tridol®, Youhan, Korea) was injected at 4 hours intervals.
At 1 hr, 6 hr, 12 hr, and 24 hr after extubation, patients were questioned about the intensity of the sore throat in the resting state and during the swallowing movement, respectively, by an anesthesiologist who was unware of this study detail. The incidence of throat pain was recorded and its intensity was measured using a 0-100 mm visual analogue scale (VAS), where a score of 0 denotes no pain and a score of 100 means the worst pain imaginable. And the incidence of POST was confined as non-zero VAS score through 24 hours after surgery. The incidences of any complications (somnolence, dizziness, headache, nausea) and the total number of rescue analgesics injected during 24 hours after surgery were recorded.
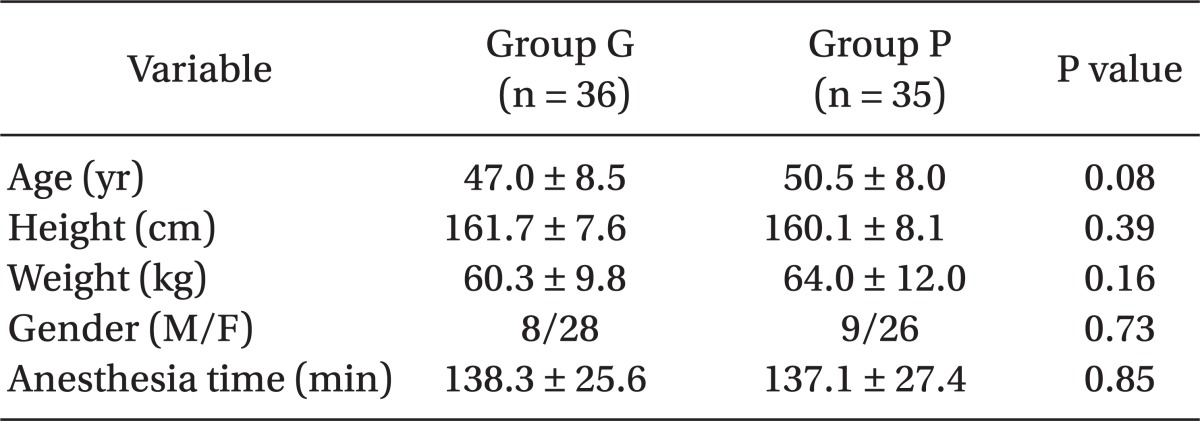
Based on our preliminary data, the incidence of POST after thyroid surgery was 80%, and, we assumed that 40% reduction of the incidence of POST, at rest, would clinically be relevant. The power analysis suggested that 32 patients in each group would be needed for a type I error (α) of 0.05 and a power (1-β) of 0.8. To compensate for the potential dropouts, we enrolled 40 patients in each group. The distribution of all continuous variables that followed a normal distribution, as assessed by the Kolmogorov-Smirnov Lilliefors goodness-of-fit test, such as, age, height, weight, and anesthesia time, are presented as the means ± SDs and were compared using the two sample Student's t-test. Non-normally-distributed continuous data, such as, VAS scores at rest and during movement, at specific times, were compared with the non-parametric Mann-Whitney U test with Bonferroni correction for multiple comparisons. And the number of times of rescue analgesics used are reported as the median and ranges, and were compared with the non-parametric Mann-Whitney U test. Categorical data for each group, such as, gender distribution, the incidence of sore throat, and complications, were compared using the chi-square test or the Fisher's exact test. For all statistical tests, two-tailed value of P < 0.05 were considered statistical significant. Statistical analysis was conducted using the SPSS package version 19.0 (SPSS Inc/IBM, Chicago, IL).
Eighty patients, aged 23 to 65 years were enrolled in this study, between March, 2011 and September, 2011. A total of four patients were excluded from the gabapentin group because of more than two intubation attempts in two patients and a blood-tinged endotracheal tube after extubation in the other two patients. Five patients were excluded from the placebo group; more than two intubation attempts in three and a blood-tinged endotracheal tube after extubation in the other two. Thus, a total of 71 patients (36 in the gabapentin group and 35 in the placebo group) completed this study.
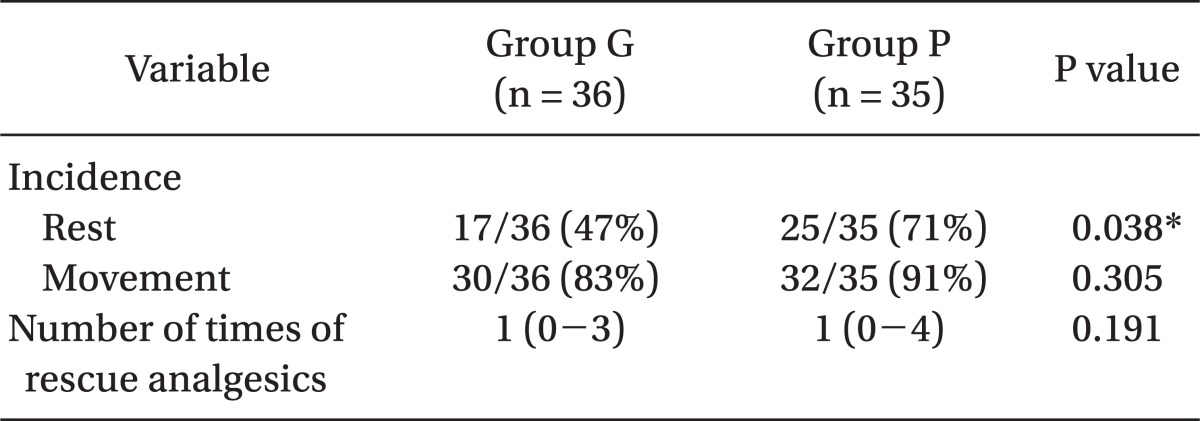
Demographics in the two study groups were similar (Table 1). Table 2 summarizes the incidences of POST during rest and movement. The incidence of POST during rest in the gabapentin group (47%, P = 0.038) was significantly lower than in the placebo group (71%). However, the incidences of POST during the swallowing movement were not significantly different in the two groups (83% vs. 91%, P = 0.305).
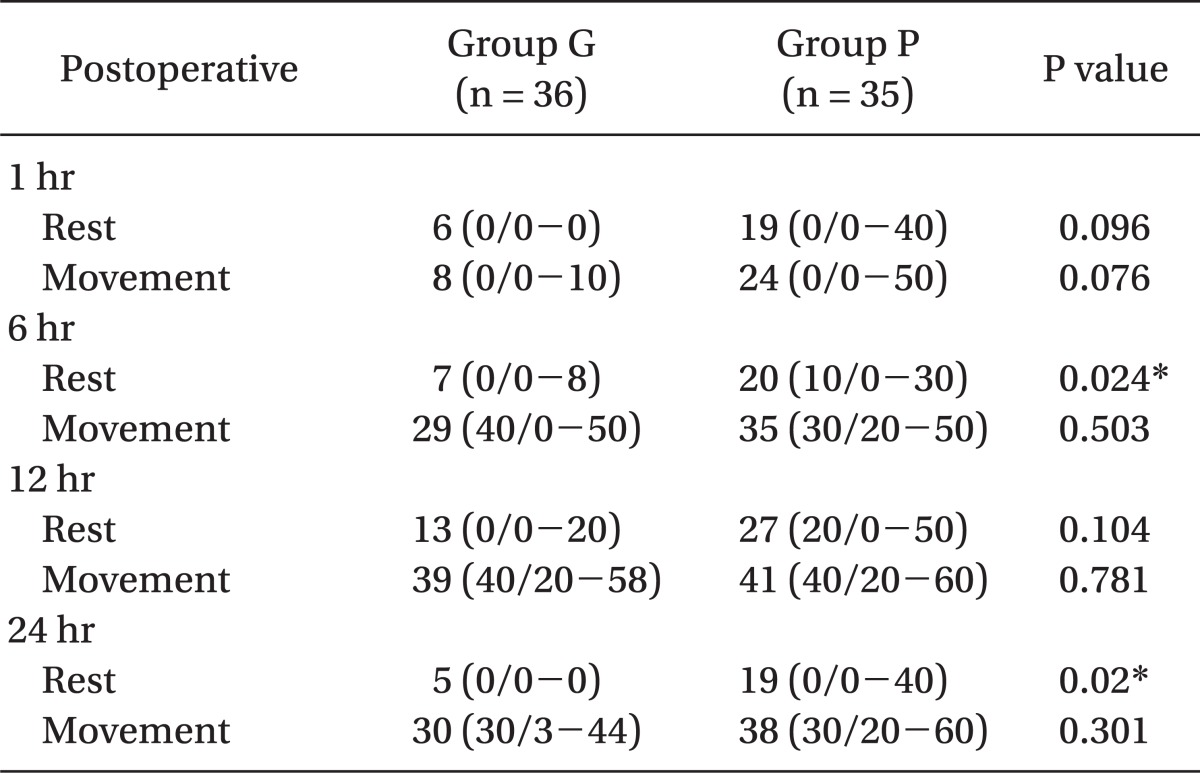
Table 3 shows VAS scores at specific times after surgery. At 6 hr and 24 hr postoperatively, the gabapentin group had a statistically lower mean VAS score at rest than the placebo group (P = 0.024, and 0.02, respectively). But during movement, VAS scores were not significantly different between both groups. In addition, the number of times of rescue analgesics during 24 hours after surgery was not different in the groups (Table 3).
In terms of the incidence of side effects, (summarized in Table 4), those of somnolence, dizziness, headache, and nausea, were not different in the two groups.
This study shows that preoperatively administrated gabapentin (Neurontin® 600 mg) reduced the incidence of POST in the resting state for 24 hours after thyroid surgery. The incidence of POST at rest (patients who recorded non-zero VAS score through 24 hours after surgery) in the placebo group was 71% (25/35), and the incidence of POST in gabapentin group was 47% (17/36), which is lower with statistical significance than observed in the the placebo group. In addition, VAS scores at rest was significantly lower in the gabapentin group during specific times (6 and 24 hours) after surgery. However, incidences of POST during movement, were similar in both groups (83% vs. 91%).
A recent study showed that binding of gabapentin to the alpha-2-delta subunit of N-type voltage-gated calcium channels in dorsal root ganglion neurons probably underlies its mode of action [6]. This binding is thought to reduce neurotransmitters, such as glutamate and substance P, from being released, and thus, suppress neuronal excitability, following nerve or tissue injury. Thus, our results suggest that neuronal excitement following mucosal injury, due to direct laryngoscope and endotracheal cuff pressure, is attenuated by gabapentin prophylaxis.
However, during movement, the effect of gabapentin was limited. McHardy and Chung [11] reported that the etiology of POST involves epithelial loss, glottis hematoma, glottis edema, submucosal tears, and contact ulcer granuloma. And Scuderi [12] suggested that POST is the result of mucosal injury and resulted in inflammation caused by the process of airway instrumentation or by the irritation of foreign objects; however, admitted that the actual cause of POST remains a mystery. Consequently, it appears that POST is probably caused by somatic and visceral tissues, and that it may be more pronounced and complicated than has been considered to date. Therefore. we suggested that 600 mg of gabapentin is somewhat small to decrease pain during movement. Thus, more studies are required to determine the optimal dose of gabapentin and proper times of administration of gabapentin to reduce POST during the movement.
In the present study, no significant intergroup differences in side effects were observed. Dizziness and somnolence have been demonstrated to be the most common adverse events of gabapentin in the previous controlled studies on chronic pain [13,14]. However, the situation in the postoperative period is quite different from that of the chronic pain state, and although gabapentin probably has a favorable side effects profile, the meta-analysis [15] showed that gabapentin is associated with greater sedation (OR 3.86, 95% CI 2.5-5.94).
In conclusion, gabapentin (Neurontin™ 600 mg) administrated 1 hour before the induction of anesthesia was found to reduce the incidence and intensity of postoperative sore throat at rest, without inducing significant adverse events at 6 and 24 hours after thyroid surgery. However, it did not reduce the incidence or intensity of postoperative sore throat during swallowing.
Acknowledgments
This article was supported by National Health Insurance Corporation Ilsan Hospital research fund.
References
1. Higgins PP, Chung F, Mezei G. Postoperative sore throat after ambulatory surgery. Br J Anaesth 2002; 88: 582-584. PMID: 12066737.



2. Macario A, Weinger M, Carney S, Kim A. Which clinical anesthesia outcomes are important to avoid? The perspective of patients. Anesth Analg 1999; 89: 652-658. PMID: 10475299.


3. Hisham AN, Roshilla H, Amri N, Aina EN. Post-thyroidectomy sore throat following endotracheal intubation. ANZ J Surg 2001; 71: 669-671. PMID: 11736830.


4. Christensen AM, Willemoes-Larsen H, Lundby L, Jakobsen KB. Postoperative throat complaints after tracheal intubation. Br J Anaesth 1994; 73: 786-787. PMID: 7880666.



5. Kinney MA, Mantilla CB, Carns PE, Passe MA, Brown MJ, Hooten WM, et al. Preoperative gabapentin for acute post-thoracotomy analgesia: a randomized, double-blinded, active placebo-controlled study. Pain Pract 2012; 12: 175-183. PMID: 21676165.


6. Gilron I. Gabapentin and pregabalin for chronic neuropathic and early postsurgical pain: current evidence and future directions. Curr Opin Anaesthesiol 2007; 20: 456-472. PMID: 17873599.


7. Fassoulaki A, Melemeni A, Paraskeva A, Petropoulos G. Gabapentin attenuates the pressor response to direct laryngoscopy and tracheal intubation. Br J Anaesth 2006; 96: 769-773. PMID: 16595614.



8. Menigaux C, Adam F, Guignard B, Sessler DI, Chauvin M. Preoperative gabapentin decreases anxiety and improves early functional recovery from knee surgery. Anesth Analg 2005; 100: 1394-1399. PMID: 15845693.



9. Pandey CK, Priye S, Ambesh SP, Singh S, Singh U, Singh PK. Prophylactic gabapentin for prevention of postoperative nausea and vomiting in patients undergoing laparoscopic cholecystectomy: a randomized, double-blind, placebo-controlled study. J Postgrad Med 2006; 52: 97-100. PMID: 16679671.

10. Leung JM, Sands LP, Rico M, Petersen KL, Rowbotham MC, Dahl JB, et al. Pilot clinical trial of gabapentin to decrease postoperative delirium in older patients. Neurology 2006; 67: 1251-1253. PMID: 16914695.


11. McHardy FE, Chung F. Postoperative sore throat: cause, prevention and treatment. Anaesthesia 1999; 54: 444-453. PMID: 10995141.


12. Scuderi PE. Postoperative sore throat: more answers than questions. Anesth Analg 2010; 111: 831-832. PMID: 20870978.


13. Backonja M, Beydoun A, Edwards KR, Schwartz SL, Fonseca V, Hes M, et al. Gabapentin for the symptomatic treatment of painful neuropathy in patients with diabetes mellitus: a randomized controlled trial. JAMA 1998; 280: 1831-1836. PMID: 9846777.


14. Rowbotham M, Harden N, Stacey B, Bernstein P, Magnus-Miller L. Gabapentin for the treatment of postherpetic neuralgia: a randomized controlled trial. JAMA 1998; 280: 1837-1842. PMID: 9846778.


15. Ho KY, Gan TJ, Habib AS. Gabapentin and postoperative pain--a systematic review of randomized controlled trials. Pain 2006; 126: 91-101. PMID: 16846695.


- TOOLS