|
 |
|
|
Abstract
Background
Although supplemental fentanyl has been widely used to blunt the hemodynamic responses to laryngoscopic intubation, its residual vagotonic effect may increase the risk of reflex bradycardia. We compared the incidence and severity of significant reflex bradycardia after a bolus injection of equivalent doses of fentanyl and remifentanil (control drug).
Methods
In this prospective, randomized, double-blind study, 220 adult patients undergoing major abdominal surgery were randomly assigned to receive fentanyl (1.5 µg/kg) or remifentanil (1.5 µg/kg). No anticholinergic prophylaxis was administered. Symptomatic reflex bradycardia was defined as a sudden decrease in heart rate to < 50 beats per minute (bpm) or to 50-59 bpm associated with a systolic arterial pressure < 70 mmHg in connection with surgical maneuvers. If bradycardia or hypotension developed, atropine or ephedrine was administered following a predefined treatment protocol.
Results
In total, 188 subjects (remifentanil, 95; fentanyl, 93) were included. The proportion of subjects with symptomatic reflex bradycardia in the fentanyl group was similar to that in the remifentanil group (30.1% vs. 28.4%, respectively). Atropine and/or ephedrine were needed similarly in both groups. The differences between the group of 55 patients who presented with symptomatic reflex bradycardia were not statistically significant with respect to the lowest heart rate, anesthetic depth-related data (bispectral index and end-tidal sevoflurane concentration), or the proportion of causative surgical maneuvers.
Bradycardia and bradyarrhythmia in connection with certain surgical maneuvers, such as abdominal wall retraction or manipulation of the bowel or mesentery, are commonly encountered during major abdominal surgery. In most cases, these are short lived and rapidly respond to relief of surgical traction or anticholinergic administration [1,2]. However, several studies [1,3-5] have reported critical bradycardia or asystole due to intense surgical vagal stimulation. Although detailed evidence for the pathway involved is still lacking, it is commonly assumed to be caused by reflex vagal activity [1,2].
Supplemental intravenous (IV) fentanyl has been widely used to blunt the hemodynamic response to laryngoscopy and tracheal intubation [6,7]. However, when considering the centrally mediated vagotonic and sympatholytic action of fentanyl [8], such therapy is useful during anesthetic induction but may increase the potential risk of reflex bradycardia, particularly during surgical procedures capable of invoking high vagal stimulation. This vagotonic effect of fentanyl persists for several hours after a single bolus injection [8,9]. However, to date, no study has described the residual effect of fentanyl administered intravenously during the induction of anesthesia on the incidence and severity of reflex bradycardia during major abdominal surgery.
Therefore, in the present prospective, randomized, double-blind study, we evaluated whether IV fentanyl administered during induction of anesthesia increased the incidence or severity of clinically significant reflex bradycardia during major abdominal surgery. These effects were compared to an equivalent IV dose of a control drug, remifentanil, which was selected because of its ultra-short duration of action.
This study protocol was approved by our Institutional Review Board, and written informed consent was obtained from all subjects before study enrollment. In total, 220 surgical patients, aged 18-75 years, classified as American Society of Anesthesiologists physical status I or II and who were scheduled to undergo major abdominal surgery under general anesthesia, were enrolled. Exclusion criteria were patients with known cardiovascular disease, those taking drugs with known cardiovascular activity, and those with electrocardiogram (ECG) abnormalities including sinus bradycardia or tachycardia. An independent investigator evaluated all inclusion/exclusion criteria and randomized the subjects into two groups of 110 subjects each using a computer-generated random number table. These patients received either 1.5 µg/kg remifentanil (group R) or 1.5 µg/kg fentanyl (group F).
Because the optimal timing of a bolus injection of fentanyl and remifentanil to blunt cardiovascular intubation responses differs [7,10], two syringes containing the study drugs and normal saline (NS) were prepared for every subject by the same investigator. The indicated dose of fentanyl or remifentanil was drawn into a syringe and then filled with NS to 5 ml (depending on the subject's weight and group assignment), whereas the other syringe was filled with 5 ml of NS alone. The allocation sequences were contained in a set of sealed envelopes, and the other investigator and patients involved in the study were blinded to the identity of the study drugs.
No premedication was administered to any patient. Heart rate (HR), arterial oxygen saturation, ECG, and bispectral index (BIS) were monitored continuously. Anesthesia was induced with 1.0 mg/kg propofol followed by an additional 10 mg every 5 s until loss of verbal contact. After loss of consciousness, the lungs were manually ventilated with 2% sevoflurane and 100% oxygen, and then 0.1 mg/kg vecuronium and the first syringe (either 1.5 µg/kg fentanyl or NS of an equal volume) were injected intravenously. Three minutes after administering the contents of the first syringe, the second syringe (1.5 µg/kg remifentanil or NS of an equal volume) was injected. Each syringe was injected over a 5-s period. Tracheal intubation was performed 2 min after injection of the second syringe. An arterial catheter for continuous monitoring of arterial blood pressure was inserted immediately after induction of anesthesia. The patient's lungs were then mechanically ventilated to maintain a partial pressure of end-tidal carbon dioxide of 30-40 mmHg.
Anesthesia was maintained with sevoflurane in combination with 67% air in oxygen. Depth of anesthesia was controlled by altering the inhaled sevoflurane concentration, based on the hemodynamic response to surgery and BIS value (target value, 40-60) at the discretion of the attending anesthesiologist. Neuromuscular block was monitored intraoperatively using a peripheral nerve stimulator at the ulnar nerve of the wrist. Muscle relaxation was supplemented with vecuronium to maintain a train-of-four count < 2 during the study period.
The study period was predefined as the first 2 h after the start of anesthetic induction, because the vagotonic effect of fentanyl almost disappears after that time [9]. A single attending anesthesiologist evaluated all observation variables in this study. Reflex bradycardia was defined as a sudden decrease in HR to < 60 beats per minute (bpm) in association with surgical manipulation of abdominal contents. Reflex bradycardia severity was graded as mild (50-59 bpm), moderate (40-49 bpm), or severe (< 40 bpm). Symptomatic reflex bradycardia, a major outcome of this study, was defined as a HR < 50 bpm or mild bradycardia associated with a systolic arterial pressure < 70 mmHg (Fig. 1). Hypotension was defined as systolic arterial pressure < 80 mmHg. Hypotension severity was graded as mild (70-79 mmHg), moderate (60-69 mmHg), or severe (< 60 mmHg).
Syringes containing atropine and ephedrine were always available in all cases. Atropine (0.5 mg) or ephedrine (4 mg) was administered if spontaneous return to the normal HR range was not observed after 10 s, depending on severity. This treatment protocol is described in Fig. 1. During every reflex bradycardia episode, the lowest HR and systolic arterial pressure were recorded and used in the statistical analysis. The BIS value and end-tidal sevoflurane concentration at the moment of reflex bradycardia, and the onset time of reflex bradycardia were also recorded. The time taken from the start of anesthetic induction to the occurrence of reflex bradycardia was defined as the onset time of reflex bradycardia. Additionally, the attending anesthesiologist classified the causative surgical maneuvers into three categories of manual retraction of the wound edge, set up of the self-retaining retractor, and manipulation of the bowel or mesentery. Appropriate cardiovascular interventions were applied when any episode of cardiac instability developed in the opinion of the attending anesthesiologist irrespective of surgical vagal stimulation and warranted the administration of cardiovascular drugs during the study period; such patients were subsequently excluded from the study.
The primary outcome was the difference in the proportion of patients with symptomatic reflex bradycardia between groups R and F. In an unpublished preliminary study, in which the protocol was identical to the present study except for no use of opioids during anesthetic induction, we observed that symptomatic reflex bradycardia occurred in six of the 30 patients who underwent major abdominal surgery. Thus, the incidence of symptomatic reflex bradycardia was estimated to be 20% in group R. Because critical reflex bradycardia that does not respond to prompt cardiovascular drug therapy is rare in a clinical situation, we considered at least a 100% increase of this incidence in group F to be clinically significant. Based on these figures, and using α = 0.05 and β = 0.2 for the clinical study design incorporating two groups of equal size, we calculated that a sample size of 91 patients per group would be required. Therefore, we enrolled 110 patients per group to compensate for possible dropouts.
Categorical variables were compared using Pearson χ2-tests with a continuity correction or Fisher's exact test as applicable. Continuous variables were tested for normality using the Kolmogorov-Smirnov test. Values for normally distributed variables are presented as mean ± standard deviation and compared using an unpaired two-tailed t-test. BIS values and end-tidal sevoflurane concentrations at the time of symptomatic reflex bradycardia, and at the onset time of symptomatic reflex bradycardia are reported as medians (interquartile range) and compared using an unpaired two-tailed t-test. SPSS ver. 13.0 (SPSS Inc., Chicago, IL, USA) was used for the statistical analysis. P values < 0.05 were considered statistically significant.
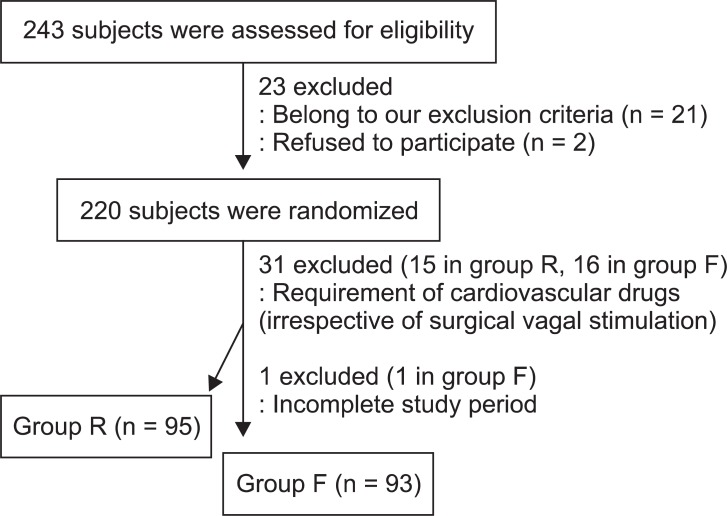
Of the 220 subjects, 32 did not complete the study. Fifteen patients in group R and 16 in group F required cardiovascular drugs because of cardiac instability, which developed irrespective of surgical vagal stimulation during the study period. One subject in group F was additionally excluded because of an incomplete study period, during which surgery was aborted after discovering advanced carcinoma. Finally, 95 subjects in group R and 93 subjects in group F were included in the analysis (Fig. 2).
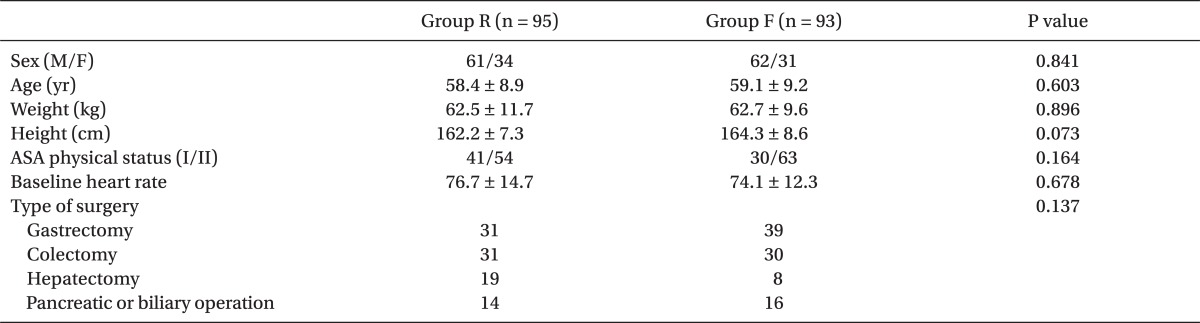
Both groups were similar in terms of demographic characteristics, baseline HR, and type of surgery (Table 1). The proportion of patients with symptomatic reflex bradycardia in group F was similar to that in group R (30.1% vs. 28.4%, respectively; Table 2). The incidence of severe bradycardia (HR < 40 bpm) did not differ between the groups, with five patients in group R and six patients in group F. Atropine and/or ephedrine were needed similarly in both groups (Table 2). Of the 49 total cases requiring atropine or ephedrine, all but five patients responded well to the treatment (two in group R and three in group F); initial non-responders were given repeat doses of atropine or ephedrine. Dysrhythmia occurred similarly in both groups, ranging from junctional rhythm to atrio-ventricular block, but these episodes lasted only a few seconds.
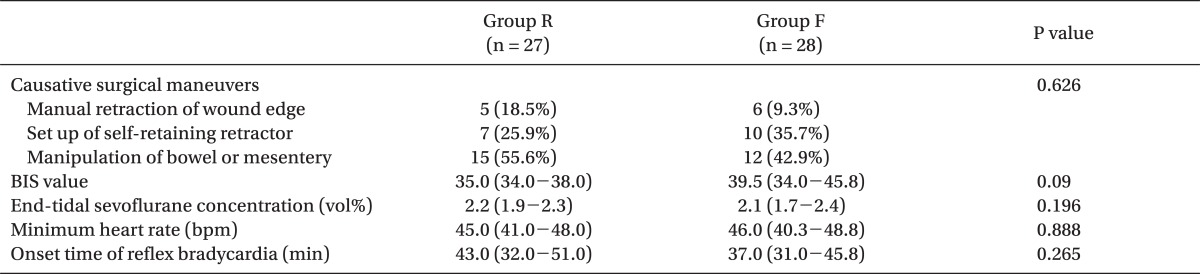
In 55 patients presenting with symptomatic reflex bradycardia, the differences between the groups were not significant with respect to the lowest HR, anesthetic depth-related data (BIS value and end-tidal sevoflurane concentration), or the proportion of causative surgical maneuvers (Table 3). Manipulation of the bowel or mesentery was the leading causative surgical maneuver (55.6% in group R and 42.9% in group F), and set up of the self-retaining retractor and manual retraction of the wound edge were the second and third most common causes. Symptomatic reflex bradycardia was observed similarly in both groups during the early part of surgery. The medians (interquartile range) for the onset times of symptomatic reflex bradycardia were 43.0 min (range, 32.0-51.0 min) in group R and 37.0 min (range, 31.0-45.8 min) in group F (Table 3).
The main finding of our study was that 1.5 µg/kg fentanyl by bolus injection during anesthetic induction did not increase the incidence or severity of clinically significant reflex bradycardia during major abdominal surgery compared to that of an equivalent IV dose of remifentanil.
Although the incidence of reflex bradycardia has not yet been separately evaluated, the overall incidence of intraoperative bradycardia varies from 20 to 42% in the absence of anticholinergic prophylaxis [11-13]. In this study, the incidences of symptomatic reflex bradycardia were 30.1% in group F and 28.4% in group R during the first 2 h of anesthesia. The incidence of reflex bradycardia can be highly variable, depending on the type of surgery, anesthetic drugs used, and cut-off value for the definition of bradycardia. We adopted symptomatic bradycardia as a major outcome, and the cut-off value was predefined differently depending on blood pressure. If systolic arterial pressure was < 70 mmHg, HR < 60 bpm was chosen as the cut-off value. We defined symptomatic bradycardia irrespective of arterial pressure if HR was < 50 bpm. It is not always clear what degree of bradycardia is tolerable. Although a HR of < 60 bpm is a conservative definition, bradycardia may be clinically significant in cases accompanied by hypotension. A sudden decrease in HR to < 50 bpm, even without associated hypotension, is generally grounds for immediate treatment.
In this study, eligible surgical procedures were limited to major abdominal surgery consisting of gastrectomy, colectomy, pacreatectomy, and major hepatobiliary surgery, which are surgical procedures with known potential for high vagal stimulation [4,13]. Reflex bradycardia can occur during abdominal surgery with varied magnitude and duration such as immediately after displacement of the liver, during insertion of the hand into the peritoneal cavity, traction on the abdominal mesentery, placement of packing, or retraction of wound edges [1,2].
The mechanism of the oculocardiac reflex observed during strabismus surgery has been well established in many clinical studies and neuroanatomical investigations. However, detailed evidence for the pathway involved in reflex bradycardia during abdominal surgery is still lacking. Similar to the oculocardiac reflex observed during strabismus surgery, reflex bradycardia is commonly assumed to be caused by increased vagal tone. Mechanical manipulations of the viscera, mesentery, or abdominal wall stimulate the hepatic plexus, celiac plexus, splanchnic nerve, or vagus nerve. In turn, these afferent stimuli cause increased vagal activity in the heart, thereby leading to bradycardia and/or hypotension as an efferent response [1,2].
Thus, we hypothesized that residual excessive dominance of vagotonic activity caused by fentanyl administered during induction could increase the incidence and severity of reflex bradycardia during major abdominal surgery. Contrary to our hypothesis, a single dose (1.5 µg/kg) of IV fentanyl did not augment reflex bradycardia. This result was in contrast to the finding that the intraoperative use of opioids is associated with a higher incidence of oculocardiac reflex during pediatric strabismus surgery [14,15]. Although the afferent pathway is quite different from that of reflex bradycardia during abdominal surgery, the oculocardiac reflex is also a vagal-mediated reflex bradycardia (i.e., trigemino-vagal reflex). The most likely explanation for this difference might be that the residual vagotonic effect of a single dose (1.5 µg/kg) of IV fentanyl was not sufficient to increase the risk of reflex bradycardia during abdominal surgery. According to a simulated pharmacokinetic and pharmacodynamic experiment, the plasma concentration of fentanyl is about 2-3% of its initial plasma concentration during the first 90 min after bolus administration [9]. In oculocardiac reflex studies with an opposite result to ours, the timing of opioid administration was different from that in our study, as remifentanil was continuously infused throughout the surgery [14], whereas IV opioids were injected 5 min before the extraocular muscle manipulation [15].
The usual recommended IV dose of fentanyl for attenuating hemodynamic responses to laryngoscopic intubation is 1.5-3.0 µg/kg [6,7]. As a higher dose of fentanyl carries an increased risk for reflex bradycardia during surgical procedures with high vagal stimulation, further study will be necessary to verify this issue.
No use of opioids during anesthetic induction was reasonable in the control group to clarify the influence of IV fentanyl administered during anesthetic induction on vagal-mediated reflex bradycardia during surgical procedures. However, no use of opioids during anesthetic induction did not blunt the hemodynamic responses of laryngoscopic intubation effectively in our preliminary study. Therefore, in this study, we chose remifentanil (1.5 µg/kg) as a control drug for comparison with fentanyl (1.5 µg/kg) because it has an ultra-short duration of action lasting an average of about 8 min [10,16]. Remifentanil by bolus injection is effective and safe for preventing an untoward hemodynamic response to laryngoscopy and intubation [10,16]. Dose selection of remifentanil in this study was based on the study of Lang et al. [17], which reported that the potency ratio for fentanyl to remifentanil is nearly 1 : 1, using a 50% minimum alveolar concentration reduction of isoflurane. Thus, we believe that bolus injection of remifentanil during anesthetic induction might have exerted no residual vagotonic effect during surgical procedures in group R. Actually, the incidence of symptomatic reflex bradycardia in group R of this study was similar to that in the control group (no use of opioids during anesthetic induction) of our preliminary study (28.4% vs. 20%, respectively; P = 0.478).
When we designed this study, we wanted to deliver fentanyl or remifentanil during anesthetic induction in such a way that their peak site effects occurred at the same time; thus, most effectively attenuating the cardiovascular intubation response. The time required to reach peak effect-site concentration after a single bolus injection is 1-2 min for remifentanil and 5 min for fentanyl [7,9,10,16]. Thus, fentanyl and remifentanil were injected 5 and 2 min before intubation, respectively.
In this study, all episodes of symptomatic reflex bradycardia developed within 60 min after anesthetic induction (i.e., during the early part of the surgical period). The medians (interquartile range) of bradycardia onset times were 43.0 min (range, 32.0-51.0 min) in group R and 37.0 min (range 31.0-45.8 min) in group F. A possible explanation for this result is that repeated mechanical manipulations may lead to a reduced or fatigued reflex after the first manipulation. This phenomenon has also been reported for the oculocardiac reflex during strabismus surgery [18].
Some potential criticisms of our study exist. First, it may be suggested that routine use of anticholinergic premedication is sufficient for protecting the patient against excessive vagal tone mediated by surgical procedures. Although not consistently reliable, anticholinergic prophylaxis definitely decreases the incidence of reflex bradycardia [1,11,13]. However, this practice remains controversial. Tachycardia can be as harmful as bradycardia in some patients with cardiovascular instability. Additionally, IV atropine can induce premature ventricular beats, ectopic beats, or tachycardia, and these arrhythmias are more consistent than reflex bradycardia [14]. Thus, routine use of anticholinergic premedication should be weighed against the possible risk of serious intraoperative reflex bradycardia.
The other major concern may be whether the depth of sevoflurane in both groups was comparable during surgical vagal stimulation. However, as the duration of surgical vagal stimulation was uncertain, and the strength of surgical stimuli is widely different in various surgical procedures, it was unethical to apply equally predetermined doses of sevoflurane during the study period. Previous studies [19,20] have suggested that inappropriate anesthetic depth increases the incidence of the oculocardiac reflex during pediatric strabismus surgery. In this regard, we compared the sevoflurane concentrations and BIS values in both groups at the moment of significant reflex bradycardia, and these were comparable with respect to anesthetic depth.
Last, we excluded patients with known cardiovascular disease or those taking drugs with known cardiovascular activity from this study. Cardiac instability episodes occur frequently in these populations of patients during induction or maintenance of anesthesia, irrespective of surgical vagal stimulation. As a result, many patients would have been dropped out by our exclusion criteria (administration of cardiovascular drugs during the study period); thus, it may have been difficult to acquire a sufficient number of subjects. As vagally mediated reflex bradycardia can pose more risk to these populations [21], it may be additionally difficult to solve this ethical problem in the study design.
In conclusion, compared to an equivalent IV dose of remifentanil, fentanyl (1.5 µg/kg) administered intravenously during anesthetic induction did not increase the incidence or severity of significant reflex bradycardia in patients undergoing major abdominal surgery. Therefore, this anesthetic technique is unlikely to increase the risk of reflex bradycardia, even during major abdominal surgery capable of invoking high vagal stimulation. However, as clinically significant reflex bradycardia occurs with a relatively high frequency (in up to 30% of patients undergoing major abdominal surgery), HR should be carefully monitored and appropriate treatments considered if significant bradycardia is observed.
References
1. Doyle DJ, Mark PW. Reflex bradycardia during surgery. Can J Anaesth 1990; 37: 219-222. PMID: 2088315.

2. Kinsella SM, Tuckey JP. Perioperative bradycardia and asystole: relationship to vasovagal syncope and the Bezold-Jarisch reflex. Br J Anaesth 2001; 86: 859-868. PMID: 11573596.


3. Park JY, Park SJ, Kim JY, Shin HW, Lim HJ, Kim J. Cardiac arrest due to a vagal reflex potentiated by thoracic epidural analgesia. J Int Med Res 2006; 34: 433-436. PMID: 16989501.

4. John P, Raj S, Kartikeyan , Chandy TT. Asystole following profound vagal stimulation during hepatectomy. Indian J Anaesth 2008; 52: 81-82.
5. Croitor LB, Módolo NS, Braz JR, Cury Rojas A. Unexpected cardiac arrest during cholecystectomy. Rev Bras Anestesiol 2002; 52: 457-460. PMID: 19479111.
6. Dahlgren N, Messeter K. Treatment of stress response to laryngoscopy and intubation with fentanyl. Anaesthesia 1981; 36: 1022-1026. PMID: 7032347.

7. Kovac AL. Controlling the hemodynamic response to laryngoscopy and endotracheal intubation. J Clin Anesth 1996; 8: 63-79. PMID: 8695083.

8. Fukuda K. Edited by Miller RDIntravenous opioids anesthetics. Miller's anesthesia. 2005, 6th ed. : Philadelphia, Churchill Livingstone Inc. pp 379-437.
9. Shafer SL, Varvel JR. Pharmacokinetics, pharmacodynamics, and rational opioids selection. Anesthesiology 1991; 74: 53-63. PMID: 1824743.

10. Yang QY, Xue FS, Liao X, Liu HP, Luo MP, Xu YC, et al. Comparison of bolus remifentanil versus bolus fentanyl for blunting cardiovascular intubation responses in children: a randomized, double-blind study. Chin Med J (Engl) 2009; 122: 44-50. PMID: 19187616.
11. Cozanitis DA, Lindgren L, Rosenberg PH. Bradycardia in patients receiving atracurium or vecuronium in conditions of low vagal stimulation. Anaesthesia 1989; 44: 303-305. PMID: 2566284.

12. Morton CP, Drummond GB. Bradycardia and vecuronium: comparison with alcuronium during cholecystectomy. Br J Anaesth 1992; 68: 619-620. PMID: 1351737.


13. Cozanitis DA, Pouttu J, Rosenburg PH. Bradycardia associated with the use of vecuronium. A comparative study with pancuronium with and without glycopyrronium. Anaesthesia 1987; 42: 192-194. PMID: 2881499.

14. Chung CJ, Lee JM, Choi SR, Lee SC, Lee JH. Effect of remifentanil on oculocardiac reflex in paediatric strabismus surgery. Acta Anaesthesiol Scand 2008; 52: 1273-1277. PMID: 18823468.

15. Arnold RW, Jensen PA, Kovtoun TA, Maurer SA, Schultz JA. The profound augmentation of the oculocardiac reflex by fast acting opioids. Binocul Vis Strabismus Q 2004; 19: 215-222. PMID: 15530138.
16. Thompson JP, Hall AP, Russell J, Cagney B, Rowbotham DJ. Effect of remifentanil on the haemodynamic response to orotracheal intubation. Br J Anaesth 1998; 80: 467-469. PMID: 9640152.


17. Lang E, Kapila A, Shlugman D, Hoke JF, Sebel PS, Glass PS. Reduction of isoflurane minimum alveolar concentration by remifentanil. Anesthesiology 1996; 85: 721-728. PMID: 8873541.

18. Blanc VF, Hardy JF, Milot J, Jacob JL. The oculocardiac reflex: a graphic and statistical analysis in infants and children. Can Anaesth Soc J 1983; 30: 360-369. PMID: 6871777.

19. Allison CE, De Lange JJ, Koole FD, Zuurmond WW, Ros HH, van Schagen NT. A comparison of the incidence of the oculocardiac and oculorespiratory reflexes during sevoflurane or halothane anesthesia for strabismus surgery in children. Anesth Analg 2000; 90: 306-310. PMID: 10648311.
20. Yi C, Jee D. Influence of the anaesthetic depth on the inhibition of oculocardiac reflex during sevoflurane anaesthesia for paediatric strabismus surgery. Br J Anaesth 2008; 101: 234-238. PMID: 18524784.


Fig. 1
Predefined treatment protocol for bradycardia and hypotension. Symptomatic reflex bradycardia was defined as a heart rate < 50 bpm or mild bradycardia (heart rate, 50-59 bpm) associated with a systolic arterial pressure < 70 mmHg, which corresponds to the shaded area.
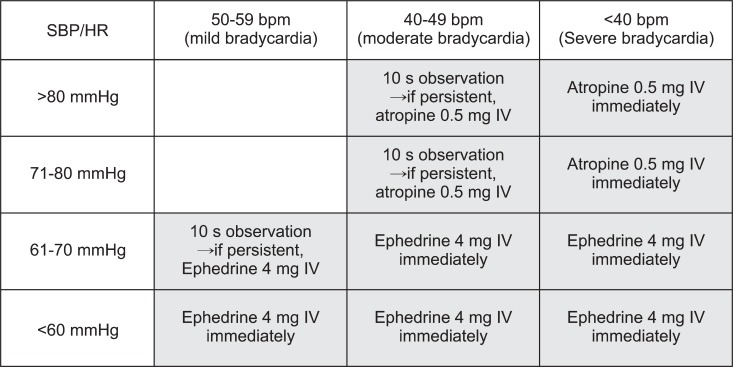
Table 2
Number (Proportion) of Patients with Reflex Bradycardia or Dysrhythmia Requiring Atropine and/or Ephedrine

Data are mean ± standard deviation (SD) or number (proportion). Bradycardia severity was graded as mild (50-59 bpm), moderate (40-49 bpm), or severe (< 40 bpm). Symptomatic reflex bradycardia was defined as a heart rate < 50 bpm or mildbradycardia associated with systolic arterial pressure < 70 mmHg.
- TOOLS