The minimum effective concentration (MEC90) of bupivacaine for an ultrasound-guided suprainguinal fascia iliaca compartment block for analgesia in knee surgery: a dose-finding study
Article information
Abstract
Background
In recent years, the suprainguinal fascia iliaca compartment block (SFICB) has become more common in clinical practice. This assessor-blinded dose-finding study aimed to determine the minimum effective concentration (MEC90, MEC95) of bupivacaine for a single-injection SFICB in patients undergoing arthroscopic anterior cruciate ligament repair.
Methods
This prospective study was conducted at a tertiary hospital (postoperative recovery room and ward). The SFICB was performed as a postsurgical intervention after spinal anesthesia. Seventy patients were allocated using the biased-coin design up-and-down sequential method. The ultrasound-guided SFICB was performed using different bupivacaine concentrations, and standard multimodal analgesia was administered to all patients. Block success was defined as the absence of pain or presence of only tactile sensation during the pinprick test conducted on the anterior and lateral regions of the mid-thigh six hours postoperatively.
Results
According to isotonic regression and bootstrap CIs, the MEC90 value of bupivacaine for a successful SFICB was 0.123% (95% CI: 0.098–0.191) and the MEC95 value was 0.188% (95% CI: 0.113–0.223).
Conclusions
Our study showed that the MEC90 and MEC95 values for bupivacaine administered via an SFICB for analgesia were 0.123% and 0.188%, respectively. One advantage of using lower concentrations of bupivacaine is the associated reduction in quadriceps weakness.
Introduction
The fascia iliaca compartment (FIC) is a funnel-shaped adipose space between the epimysium of the iliopsoas muscle and the fascia iliaca, which covers the muscle but is not attached to it [1]. The FIC contains the femoral (FN) and lateral femoral cutaneous nerves (LFCN) but not the obturator nerve (ON) [2].
In 1989, Dalens et al. were the first to describe a conventional infrainguinal fascia iliaca compartment block (FICB) for lower-limb orthopedic surgery [3]. Despite earlier publications (3:1 block), this approach has been shown to be incapable of blocking all nerves of the lumbar plexus [4–6].
Stevens et al. first described the suprainguinal fascia iliaca compartment block (SFICB) approach in 2007 [7] and in 2011, Hebbard et al. were the first to define the longitudinal ultrasound (US)-guided SFICB in cadavers [8]. Using this suprainguinal longitudinal approach, studies have shown that the ON, FN, and LFCN can all be anesthetized when a large volume is injected underneath the fascia iliaca [9]. This approach has also proven to be superior to the classic infrainguinal approach [4,10]. The SFICB may provide analgesia comparable to a lumbar plexus block in patients undergoing hip, femur, and upper thigh surgery, which explains why the authors refer to the SFICB as an “anterior lumbar plexus block” [8,9,11].
Some studies on the minimal effective volume (MEV) for the SFICB have also been performed, but none are available on the minimum effective concentration (MEC) of various local anesthetic agents [2,12,13]. For patient safety, particularly in patients who are elderly and frail, determining the lowest clinically significant concentration that blocks the major nerves targeted by the SFICB is critical and may provide an advantage by minimizing block-induced complications such as muscle weakness, local anesthetic systemic toxicity, and myotoxicity.
In this assessor-blinded dose-finding study, we aimed to detect the MEC90 and MEC95 of bupivacaine for a single-injection SFICB in patients undergoing arthroscopic anterior cruciate (ACL) ligament repair.
Materials and Methods
Study design
In this study, we investigated the MEC of bupivacaine at a fixed volume of 40 ml that achieved 90% and 95% blockade success in a population of patients undergoing arthroscopic knee surgery.
To achieve study homogeneity, all surgeries were performed by the same surgeon (LK) and the same anesthetist performed all blocks (ST). A designated participant prepared the predetermined concentration of bupivacaine. The concentration was concealed from the patient, anesthesiologist, surgeon, data collector, and evaluator (CG).
This study was approved by the local ethics committee (OMUKAE:2021/630) and the Ministry of Health (TITCK:22-AKD-08). Following registration at clinicaltrials.gov (NCT05408585), the study was conducted between June 7, 2022 and February 27, 2023 in accordance with the principles of the Declaration of Helsinki (2013), and all patients involved in the study provided written informed consent.
This study was prospectively conducted at a tertiary hospital (Samsun University Education and Research Hospital) and included American Society of Anesthesiology (ASA) Class I-II patients (aged 18–65 years) with a body mass index (BMI) between 19 and 30 kg/m2 scheduled for ACL repair under spinal anesthesia. Patients with contraindications for regional anesthesia (e.g., bleeding-coagulation disorders, infection at the injection site) were excluded, as were patients with neurological or psychiatric diseases that would impair pain assessment, orientation-cooperation issues, or a history of allergy to local anesthetics. To homogenize the samples, cases with a surgical duration < 30 min or > 60 min were excluded from the analysis.
Only the researcher responsible for randomization and drug preparation had access to the block success status of the patients during service follow-ups. The biased-coin design up-down sequential method (BCD-UDSM) was used, with the previous patient’s block success determining the bupivacaine concentration to be administered to the next patient. Based on previous studies, the initial concentration of bupivacaine was determined to be 0.25% and the up-down concentration was 0.025% [14,15,16]. Randomization was performed using the closed-envelope technique: one hundred sealed envelopes were placed in a pouch, 89 of which indicated that the bupivacaine concentration be maintained (89%) and 11 indicated the concentration be reduced (11%). Following a successful block, an envelope was drawn from the bag randomly to determine the following patient’s bupivacaine concentration, with an 11% probability (b = 0.11) it would be decreased by 0.025% and an 89% probability (1-b = 0.89) it would be maintained at the same concentration. If the block failed, the next patient was administered a 0.025% higher concentration of bupivacaine. The same probability was maintained for each patient, as the envelope that was drawn was returned to the bag after each drawing.
Anesthesia and analgesia management
All patients received standard monitoring (non-invasive arterial blood pressure, electrocardiography, and pulse oximetry) and none received any premedications. All patients received spinal anesthesia with 12–15 mg of heavy bupivacaine (2.4–3.0 ml) without an adjuvant via a midline approach at the L4–L5 interval. All arthroscopic ACL repair surgeries were performed by a single expert surgeon using the same surgical approach.
All patients underwent standard preoperative, perioperative, and postoperative analgesia protocols. At the conclusion of surgery, each patient received 20 mg intravenous (IV) tenoxicam and 1 g IV acetaminophen. The postoperative analgesia regimen consisted of IV tenoxicam every 12 h and IV paracetamol every 8 h.
Block performance
Since we excluded long- and short-term surgeries (< 30 min, > 60 min) and could not predict the complications that may develop during the operation, the block procedures were performed postoperatively under standard ASA monitoring in the post-anesthesia care unit (PACU). All blocks were performed by an experienced anesthesiologist who had successfully completed the procedure (SFICB) at least 100 times previously.
The patients were placed in the supine position after skin disinfection and draping, and a high-frequency linear US transducer (10–18 MHz, Esoate MyLabTM30Gold) was placed longitudinally at the level of the anterior superior iliac spine. The iliacus muscle and the overlying hyperechoic fascia iliaca were visualized by shifting the US probe in the caudal and medial directions. The US transducer was positioned slightly obliquely superomedially and inferolaterally and the internal oblique (cephalad) and the sartorius (caudad) muscles overlying the iliacus muscle were visualized, yielding a “bow-tie sign” image. The deep circumflex iliac artery, which is an important ultrasonographic landmark, was identified. In this position, an 80-mm block needle (Vygon Echoplex, 85 mm, 21 G) was advanced from the transducer’s caudal side using the in-plane technique, and 0.5 ml of the prepared local anesthetic solution was administered between the fascia iliaca and iliacus muscle with real-time US guidance (Fig. 1). The injection site was confirmed on US imaging. All patients then received a 40-ml local anesthetic injection at a predetermined concentration. Patients were monitored in the PACU for 20 min after the block and then transferred to the ward.

A–B. Relevant sonoanatomy for ultrasound-guided suprainguinal fascia iliaca compartment block (SFICB). (A) Natural ultrasound image of the SFICB. (B) Sonoanatomy of the muscles and fascia during an SFICB. The dashed white line represents the fascia iliaca, and the black arrow indicates the deep circumflex iliac artery. SC: subcutaneous tissue, SM: sartorius muscle, IOM: internal oblique muscle, IM: iliacus muscle, IB: iliac bone, LA: local anesthetic.
Patient follow-up
A blinded anesthetist conducted patient follow-ups in the ward. This anesthetist was not involved in any aspect of determining the bupivacaine concentration for the block, the preparation or administration of the block, or the surgical procedure. The exclusive role of the anesthetist was limited to conducting postoperative service follow-ups at 6, 12, 18, and 24 h postoperatively. Similar to the study conducted by Aliste et al. [14], our first patient follow-up was planned to be performed 6 h postoperatively to ensure that spinal anesthesia had worn off and to evaluate the patient for quadriceps weakness, which can develop due to the block. Moreover, the cutaneous and motor functions of the contralateral thigh were examined and compared 6 h postoperatively to confirm that quadriceps weakness was not due to spinal anesthesia. During patient visits at 6, 12, 18, and 24 h postoperatively, pain was assessed using the Numeric Rating Scale (NRS). Additionally, nurses assessed pain during the hourly patient follow-ups in the ward. At these intervals, block success was evaluated using the pinprick score, pain intensity using the NRS, and quadriceps muscle weakness using the Bromage score. Patients with an NRS score ≥ 4 received 50 mg of IV tramadol as a rescue analgesic.
To examine sensory block, the pinprick test was conducted using a 27-gauge hypodermic needle (0 = no sensory block; 1 = tactile sense present, no pain; 2 = no tactile sense, no pain). The mid-thigh was assessed separately in three quadrants: the anterior, lateral, and medial. Blockage of the FN, LFCN, and ON was recorded separately. The pinprick test conducted 6 h postoperatively was used to determine block success: the block was considered “successful” if the pinprick score was 1 or 2 in the anterior and lateral parts of the mid-thigh and unsuccessful or “failed” if the pinprick score was 0 in the anterior or lateral parts of the mid-thigh. The medial aspect of the thigh was evaluated and the results were documented; however, these findings were not used to determine block success. The Bromage score was used to assess the motor weakness of the quadriceps muscle (0 = full flexion of feet and knees, 1 = only able to move knees, 2 = only able to move feet, and 3 = unable to move feet or knees). A Bromage score of 2–3 at 6 h postoperatively was classified as “quadriceps muscle weakness.”
Outcome measurements
The primary outcome of the study was block success as this was used to determine the bupivacaine concentration administered to the subsequent patient (BCD-UDSM). The secondary outcomes included quadriceps muscle weakness, NRS scores, total number of analgesics required in 24 h, opioid-related side effects (nausea/vomiting), and block-related complications (vascular puncture, hematoma, local anesthetic toxicity, etc.).
Statistical analysis
This dose-finding study aimed to estimate the MEC90 and MEC95 for the US-guided SFICB. Based on the up-and-down sequential method described by Dixon [15,16], the first patient received bupivacaine at a concentration of 0.25%. The sample size for our study was dynamically determined using the BCD-UDSM, targeting a minimum of 48 successful blocks based on the dose-block success ratio observed in our previous cases. To accommodate for potential losses and setbacks during follow-up, we increased the sample size by 30%, aiming for 63 successful blocks. This approach aligns with recommendations in the literature and our clinical experience, ensuring a robust study while meeting the ethical requirements for maximum patient enrollment [15]. In comparable studies using the biased-coin model, sample size ranges between 50 and 70 were used [17–20].
Analyses were performed using R version 4.2.2 (R Foundation for Statistical Computing). Patient characteristics and results were reported using descriptive tests. The MEC90 and MEC95 were calculated using isotonic regression (upndown package, version 0.1.0). To calculate the 95% CI, the data frame was bootstrapped 2000 times (boot package, version 1.3-28.1). A P value < 0.05 was considered statistically significant.
Results
Seventy participants were recruited and assigned to the study groups (Fig. 2). The demographic characteristics of the patients are delineated in Table 1.
According to isotonic regression and bootstrap CIs, the MEC90 and MEC95 values of bupivacaine for a successful SFICB were 0.123 (95% CI: 0.098–0.191) and 0.188 (95% CI: 0.113–0.223), respectively. Of the total 70 blocks performed, 63 were successful and seven were unsuccessful. Additionally, sensory loss was observed in 36 patients, demonstrating the efficacy of the ON block. Table 2 shows the proportion of patients that exhibited successful blocks along with observed ON block efficacy according to the bupivacaine concentration. The number of failed blocks according to bupivacaine concentration was as follows: one patient at 0.225%, one patient at 0.15%, one patient at 0.125%, one patient at 0.1%, and three patients at 0.075%. Five of the failed blocks were conducted on males and two were conducted on females. Fig. 3 depicts all the blocks applied in the study and their success statuses.
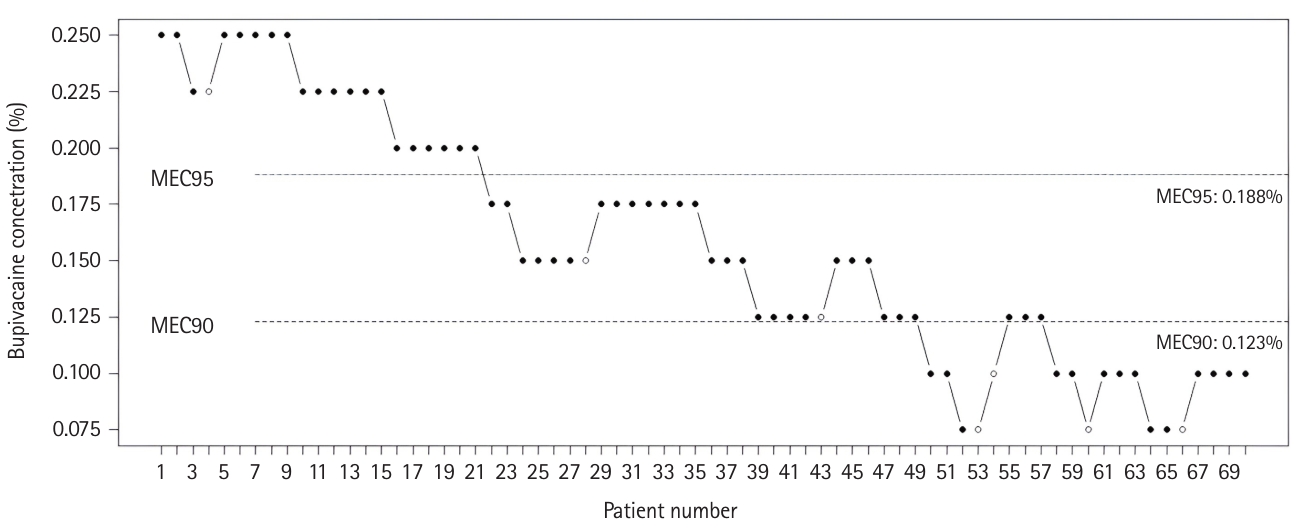
The up-and-down sequence. The black dots represent successful blocks and the white dots represent unsuccessful blocks with varying concentrations of bupivacaine. The horizontal lines represent the calculated minimum effective bupivacaine concentrations. MEC: minimum effective concentration.
Motor weakness was noted in 48 patients at 6 h postoperatively. In 33 of these patients, motor weakness resolved in subsequent follow-ups but persisted for up to 24 h in the remaining 15 patients. In the first 9 h of follow-up, all patients reported NRS scores < 4. After 9 h postoperatively, 9 patients reported an NRS score of 4. After the administration of rescue analgesics, the NRS score decreased to < 4 in all patients. NRS scores according to the bupivacaine concentration are presented in Table 3. No patient received more than one rescue analgesic during the first 24 h postoperatively, and all patients who required rescue analgesics did so at 9 h postoperatively or later. Table 2 shows the number of patients who required rescue analgesia according to the bupivacaine concentration. None of the patients experienced nausea or vomiting. No complications related to the spinal anesthesia or SFICB occurred in the PACU or in the ward up to 24 h postoperatively. Table 2 shows the number of successful and unsuccessful blocks; observed response rate; and the number of patients with successful ON blocks, quadriceps weakness, and requiring rescue analgesics according to the bupivacaine concentration.
Discussion
Our study revealed that the MEC90 and the MEC95 values of bupivacaine for the SFICB in arthroscopic knee surgeries was 0.123% and 0.188%, respectively. The incidence of SFICB-related motor weakness appears to be higher at relatively high bupivacaine concentrations.
Postoperative pain varies in intensity and duration based on factors such as surgical type, individual pain tolerance, and efficacy of pain management. Adequate perioperative pain management is crucial because poorly managed pain can prolong recovery, increase complications, and impact the overall patient experience. Consequently, a comprehensive approach to postoperative pain management should be adopted that incorporates both pharmacological and non-pharmacological interventions. In this study, a MEC analysis of the SFICB was conducted.
Studies on the MEC and MEV in relation to regional anesthesia techniques have focused primarily on peripheral nerve blocks [21,22]. As the number of fascial plane blocks performed in clinical practice has increased in recent years, various studies on the MEC and MEV have also been conducted. Similar to other plane and sheath blocks, the volume and concentration of the injected local anesthetic are crucial for the success of the SFICB, for which the local anesthetic is injected between the fascia iliaca and iliacus muscle. Nevertheless, when a high volume of local anesthetic is required, caution is advised to avoid local anesthetic systemic toxicity, particularly in elderly and frail patients.
Bupivacaine is a potent local anesthetic commonly used for regional anesthesia and pain management. It belongs to the amide class of local anesthetics and is known for its long duration of action, making it suitable for various medical procedures. However, excessive systemic absorption or accidental intravascular injections can lead to bupivacaine toxicity, affecting the central nervous and cardiovascular systems. Symptoms include dizziness, confusion, seizures, and, in severe cases, cardiac arrhythmias or cardiac arrest.
Comparative studies evaluate the effects of different predetermined volumes and concentrations. Gül et al. [23] compared three different volumes of bupivacaine (0.3 ml/kg, 0.4 ml/kg, 0.5 ml/kg) with a fixed 0.25% bupivacaine concentration with the fascia iliaca compartment block and found that 0.5 ml/kg provides more effective analgesia.
The literature includes MEV studies on cadavers and patients undergoing the SFICB [2,12,13,20]. For example, Zhang et al. [20] determined that the MEV95 of ropivacaine was 34.6 ml for the SFICB. In a cadaveric study, Kantakam et al. [12] found that the MEV90 value required for the diffusion of the dye to the FN, LFCN, and ON was 62.5 ml. In a similar study, Vermeylen et al. [2] reported that the volume of local anesthetic that could reach the FN, LFCN, and ON was 40 ml. The different values of MEV90 reported in the cadaveric study can be attributed to the lack of passive muscle movements in cadavers, the freshness of the cadavers, and the effects of these factors on the spread of the dye [24]. This is an important limitation of human cadaver studies. To our knowledge, this is the first MEC investigation of bupivacaine for the SFICB.
While the local anesthetic is likely to reach the FN and LFCN with the SFICB, involvement of the ON remains unclear. Although involvement of the ON is possible, numerous hypotheses currently exist regarding its “mechanism of action.” The most widely accepted mechanism is the involvement of the ON through cranial spread of the local anesthetic [9]. Another mechanism could be the migration of the local anesthetic from the fascia iliaca to the retroperitoneum, close to the iliac vessels and ON [12]. The possible effect of biodynamics due to the passive muscle mobilization on the distribution of local anesthetics itself might also be important.
The most significant disadvantage of most regional anesthesia techniques used in lower extremity surgical procedures is that they may lead to motor block [25]. Carella et al. [26] performed the SFICB on patients undergoing hip surgery and evaluated their walking performance on the first and second postoperative days. In this study, the SIFCB had positive results regarding postoperative functional recovery, both in terms of walking performance and incidence of orthostatic intolerance, as well as opioid-sparing effects and reduced opioid-related side effects. The effectiveness of the SFICB has also been investigated in different age groups and has been reported to provide an opioid-sparing effect in hip surgery in elderly and fragile patients [27].
The motor-sparing effect is one of the most influential factors in current studies on patients undergoing knee surgery [28]. Quadriceps muscle weakness may develop even after a single-shot adductor canal blockade if the local anesthetic dose is increased [29]. Therefore, researchers are continually searching for new techniques that preserve motor function while providing effective postoperative analgesia. The 4-in-1 block, modified 4-in-1 block, infiltration between the popliteal artery and capsule of the knee block, and adductor canal block could be included in the newer motor-sparing peripheral nerve block techniques for analgesia after total knee arthroplasty [28,30,31]. In our study, we found that the motor blockage was lower with a lower concentration of bupivacaine, and therefore, the SFICB could be used as an alternative to motor-sparing analgesia in patients undergoing knee surgery.
A local anesthetic injected into the fascia iliaca can similarly reach the FN with the SFICB, resulting in motor block. We assessed quadriceps muscle strength at 6 h postoperatively using the Bromage score and discovered that the incidence of motor block decreased as the local anesthetic concentration decreased (Table 2). The incidence of quadriceps weakness was 23% on average, especially when a bupivacaine concentration < 0.15% was administered. Most studies have evaluated quadriceps muscle weakness after various regional anesthesia techniques, aiming to identify the most appropriate technique for providing adequate analgesia while preserving motor function. For instance, in a study conducted by Vamshi et al. [32] designed to compare quadriceps weakness in patients undergoing total hip arthroplasty, the pericapsular nerve group (PENG) block and the SFICB were each applied to 30 patients. Similar to our study, the authors assessed quadriceps weakness at 6 h postoperatively; however, they used a local anesthetic mixture consisting of 30 ml 0.25% bupivacaine with 1 µg/kg clonidine for the SFICB procedure. They found that five patients in the SFICB group and none in the PENG group had quadriceps weakness and thus reported that the PENG block resulted in less quadriceps weakness than the SFICB. In our study, varying bupivacaine concentrations were administered at a fixed volume of 40 ml. The relatively higher incidence of quadriceps muscle weakness observed in our study could thus be attributed to this higher volume. Consequently, if the SFICB is administered for perioperative analgesia, a lower bupivacaine concentration is likely advantageous, particularly if early rehabilitation is crucial. At bupivacaine concentrations ≤ 0.10%, quadriceps weakness was negligible or nearly nonexistent.
Our study underscores the vital role of postoperative mobilization in patient care, which is consistent with the existing literature, and emphasizes the potential benefits, including the mitigation of complications, such as deep vein thrombosis and pulmonary embolism. Furthermore, our findings highlight the positive impact of early ambulation on musculoskeletal factors, emphasizing its crucial role in preventing postoperative muscle atrophy and joint stiffness post-surgery [33]. The ongoing search for regional anesthesia techniques that enable early patient mobilization while ensuring effective perioperative pain management is critical. Research into the concentration and volume of relevant regional techniques are a notable focus of these investigations.
This study had several limitations. First, we were unable to record the onset time of analgesia because the patients received the block whilst under the effect of spinal anesthesia, and we were also unable to determine the block duration because we did not observe the patients for more than 24 h. Levente et al. [34] reported that the median duration of a conventional fascia iliaca block with 40 ml of 0.5% ropivacaine is 48 h and can be prolonged by increasing the volume and concentration of the local anesthetic. However, caution is advised to not exceed the limits for local anesthetic systemic toxicity when using high volumes and concentrations [35]. This is particularly crucial for the elderly and fragile patients. Second, we used the pinprick test to evaluate block success even though the use of the pinprick test for sensory testing is controversial. In similar studies using the pinprick test for cutaneous testing among patients and healthy volunteers for an erector spinae plane block, the authors reported highly variable results [36,37]. In addition, evaluating block success using the pinprick test after ON block application is controversial. Bouaziz et al. [38] performed a selective ON block with 7 ml of 0.75% ropivacaine in patients scheduled for knee surgery and stated that cutaneous examination alone was insufficient to determine block success and that the adductor muscle tension should also be evaluated. Third, to assess motor block, more objective and non-biased data can be obtained using techniques such as dynamometry to measure quadriceps muscle strength [39,40].
This study demonstrated that the MEC90 and MEC95 of bupivacaine for the SFICB in arthroscopic knee surgeries were 0.123% and 0.188%, respectively. Reduced motor blockade is an advantage of using a lower concentration of bupivacaine. Further research is needed to determine the efficacy of the SFICB using varying concentrations and various local anesthetic agents.
Notes
Funding
None.
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Data Availability
Author Contributions
Caner Genc (Data curation; Project administration; Visualization; Writing – original draft; Writing – review & editing)
Serkan Tulgar (Investigation; Methodology; Writing – original draft; Writing – review & editing)
Murat Unal (Investigation; Methodology)
Ahmet Serhat Genc (Investigation; Methodology)
Lokman Kehribar (Investigation; Methodology)
Basar Erdivanli (Software; Validation; Visualization; Writing – original draft)
Kris Vermeylen (Writing – original draft; Writing – review & editing)
Ersin Koksal (Data curation; Formal analysis; Investigation; Methodology; Writing – original draft; Writing – review & editing)




