Regional analgesia techniques for video-assisted thoracic surgery: a frequentist network meta-analysis
Article information
Abstract
Background
Various regional analgesia techniques are used to reduce postoperative pain in patients undergoing video-assisted thoracic surgery (VATS). This study aimed to determine the relative efficacy of regional analgesic interventions for VATS using a network meta-analysis.
Methods
We searched the Medline, EMBASE, Cochrane Controlled Trial Register, Web of Science, and Google Scholar databases to identify all randomized controlled trials (RCTs) that compared the analgesic effects of the following interventions: control, thoracic paravertebral block (TPVB), erector spinae plane block (ESPB), serratus plane block (SPB), and intercostal nerve block (INB). The primary outcome was opioid consumption during the first 24 h postoperative period. Pain scores were also collected during three different postoperative periods: the early (0–6 h), middle (6–18 h), and late (18–24 h) periods.
Results
A total of 21 RCTs (1,391 patients) were included. TPVB showed the greatest effect on opioid consumption compared with the control (mean difference [MD]: −13.2 mg, 95% CI [−16.2, −10.1]). In terms of pain scores in the early period, ESPB had the greatest effect compared to control (MD: −1.6, 95% CI [−2.3, −0.9]). In the middle and late periods, pain scores showed that TPVB, ESPB and INB had superior analgesic effects compared to controls, while SPB did not.
Conclusions
TPVB had the best analgesic efficacy following VATS, though the analgesic efficacy of ESPBs was comparable. However, further studies are needed to determine the optimal regional analgesia technique to improve postoperative pain control following VATS.
Introduction
The use of video-assisted thoracic surgery (VATS), a minimally invasive alternative to open thoracotomy, has increased over the years, which has led to a significant reduction in postoperative pain and shorter hospital stays [1,2]. However, some patients continue to suffer from moderate-to-severe pain after VATS and postoperative pain control remains challenging [3,4]. Although thoracic epidural analgesia (TEA) has been regarded as the gold standard for postoperative pain management in thoracic surgery [5–8], complications such as epidural hemorrhage, hypotension, and postoperative urinary retention can be fatal in high-risk patients [9–11]. Considering the risks and benefits, it is necessary to use an appropriate regional analgesia technique suitable for minimally invasive thoracic surgery.
Thoracic paravertebral block (TPVB) provides unilateral thoracic analgesia comparable to TEA. Additionally, not only is it less invasive than TEA, but can also maintain hemodynamic stability and carries lower risk of complications due to anticoagulation therapy associated with anticoagulation [9,12]. According to the Enhanced Recovery After Surgery (ERAS) guidelines and the Procedure-specific postoperative pain Management (PROSPECT) group, TPVB is recommended as the primary method of regional analgesia for thoracic surgery [13,14].
Recently, however, various regional analgesia techniques, such as the erector spinae plane block (ESPB) and the serratus plane block (SPB), have superseded the traditional TPVB through their comparable analgesic effect along with reduced associated complications [15,16]. Although many studies have reported the efficacy of each of these regional analgesia techniques and have compared their effectiveness in VATS, the relative efficacy of these techniques has not been compared using network meta-analysis (NMA).
Therefore, we identified and reviewed all the articles that investigated the effects of various techniques used for postoperative analgesia for VATS, and performed an NMA to the rank order of the regional analgesia in terms of effectiveness for VATS. Our primary outcome was opioid consumption during the first 24 h postoperative period, and we also evaluated pain severity during three different postoperative periods (the early, middle, and late periods).
Materials and Methods
This study was conducted in accordance with the recommended Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines [17] and was registered with the International Prospective Register of Systematic Reviews (PROSPERO, CRD42021252062).
Data source and search strategy
The literature search was conducted to identify eligible studies for this systematic review and meta-analysis. Two researchers (S.P. and B.H.) independently searched the following electronic databases: Medline, EMBASE, Cochrane Controlled Trial Register, Web of Science, and Google Scholar for relevant studies published in English. Articles published between September 2005 and December 2020 in peer-reviewed journals were included. The primary search was conducted on January 28, 2021; however, an additional search was conducted on February 28, 2021 to include more recent studies. In addition, the studies referenced in the selected articles were searched manually.
The search strategy was as follows: (“Video assisted thoracoscopy surgery” or VATS) and [(“Thoracic paravertebral block” or TPVB) or (“paravertebral block” or PVB) or (“Serratus plane block” or “Serratus anterior plane block” or “Serratus interfascial plane block” or SPB) or (“Erector spinae plane block” or ESPB) or (“Intercostal nerve block” or INB)].
Inclusion and exclusion criteria
Studies were considered eligible if they were randomized controlled trials (RCTs) published in English that reported postoperative pain scores or total postoperative opioid consumption in both the experimental and control groups as outcomes. Non-RCTs (quasi-experimental designs), abstracts, conference proceedings, unpublished gray literature, and review studies were excluded. Among the regional analgesia techniques, continuous blocks via catheterization were also excluded.
Review procedure
We performed six steps to select the studies. First, two researchers (S.P. and B.H.) imported the titles and abstracts of the articles identified in the searches into reference management software (EndNote 20, ClarivateTM) and performed a preliminary review. Second, duplicate papers were identified and eliminated using the reference management software. Third, two researchers (S.P. and B.H.) independently reviewed the imported studies. We excluded all of the imported studies that did not clearly meet the inclusion criteria (due to the study design, participants, types of intervention, and comparison groups). Fourth, they also independently screened the titles, abstracts, and methodology sections of the studies that appeared to meet the inclusion criteria. Fifth, we retrieved the full texts of the papers that met all the inclusion criteria for data extraction and linked multiple reports of the same study. Finally, the studies included in the final selection were confirmed and coded for analysis by two researchers (B.H. and Y.J.). These coding sheets were independently checked for accuracy by researchers who were not involved in the review process. If there were any differences between the codes provided by the two reviewers, the discrepancies were resolved by consulting a third independent reviewer (C.O.).
Data extraction
The information from the included articles was independently extracted by two reviewers (B.H. and Y.J.), and each selected article was reviewed twice by both reviewers. To determine the outcomes of individual studies, pain scores and opioid consumption were determined for each group and recorded as the means and standard deviations (SDs). Medians and interquartile ranges (IQRs), as approximations of the mean and SDs, were determined using the estimation method proposed by Wan et al. [18]. When outcome data were available only as graph, a virtual ruler was used to extract the value by matching the interval between the basic unit of the plot and the ruler. The effect sizes and standard errors were calculated. Additional data, including the location, sample size, characteristics of individual study populations, and the intervention design, were extracted using a predetermined data extraction table.
Outcome definitions
The primary outcome was cumulative opioid consumption during the first 24 h postoperative period. All opioids were converted to equianalgesic intravenous (IV) morphine doses (IV morphine 1 mg = IV fentanyl 10 µg = IV sufentanil 2 µg = IV tramadol 10 mg). The secondary outcome was pain scores assessed during three different periods in the first 24 h, namely, the early (0–6 h), middle (6–18 h), and late (18–24 h) periods. For studies that included several time points within each time period, pain scores close to 1 h for early, close to 12 h for middle, and close to 24 h for late were used. In the one study that timetable was expressed as an interval (ex. 6 a.m. to 2 p.m.), a similar period expected to include the interval was used. Pain scores that were assessed using visual analogue scales (VAS) were converted to a 0–10 analogue scale to allow for statistical evaluation.
Data synthesis and statistical analysis
A random-effects NMA within a frequentist framework was performed using the R software version 4.0.3 (R Foundation for Statistical Computing, Austria) and the “netmeta” package for frequentist NMA [19,20]. A network plot was constructed to evaluate the direct and indirect comparisons of network structures, including studies. Heterogeneity was evaluated using I2 statistics. The Q statistic based on the full design-by-treatment interaction random-effects model was calculated to evaluate the global inconsistency [21]. We also evaluated the local inconsistencies between the direct and indirect effects using the net splitting technique. If the P value of the net splitting was < 0.05, we presumed there was a significant disagreement (inconsistency) between the direct and indirect estimates. We visualized the net split results using forest plots and direct evidence plots, which showed the percentage of direct and indirect evidence used for each estimated comparison. A mean path length > 2 indicated that the comparison estimate should be interpreted with caution. Additionally, a net heat plot was constructed to determine the importance of each comparison and the inconsistency of the design. Network league tables and forest plots were produced to show details of the results of the comparisons between the interventions. Outcomes are presented as mean differences with a 95% CI. To rank the analgesic interventions in order, we reported the P score, which measures the level of certainty that an intervention is better than the competing interventions [22]. In this study, the P score ranged from 0 to 1, with 1 indicating that the treatment option was statistically best and 0, the worst. Potential publication bias was assessed using comparison-adjusted funnel plots and Egger’s test. The confidence for every outcome was rated according to the grading of recommendations assessment, development, and evaluation (GRADE) system with the support of the CINeMA (Confidence in Network Meta-Analysis, https://cinema.ispm.unibe.ch/) web application (Institute of Social and Preventative Medicine, University of Bern, Switzerland) [23]. This is based on a methodological framework that considers six domains: within-study bias, reporting bias, indirectness, imprecision, heterogeneity, and incoherence [24]. The minimal clinically important difference was set at 1 out of 10 for postoperative pain and 10 mg for IV morphine-equivalent consumption.
Results
Baseline characteristics of the included studies
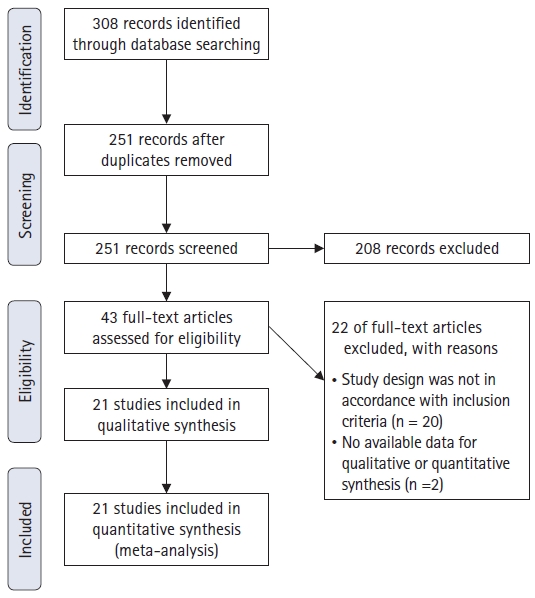
The literature screening process and results are shown in Fig. 1. The screening sequence of the PRISMA 2009 flow diagram identified 21 studies that compared the analgesic efficacy of TPVB, ESPB, SPB, intercostal nerve block (INB) and against control (no block) [25–45] with a total of 1,391 patients included. Table 1 shows the characteristics of the included studies. Table 2 shows the number of included studies and enrolled patients sorted by outcomes.
Methodological quality and risk of bias
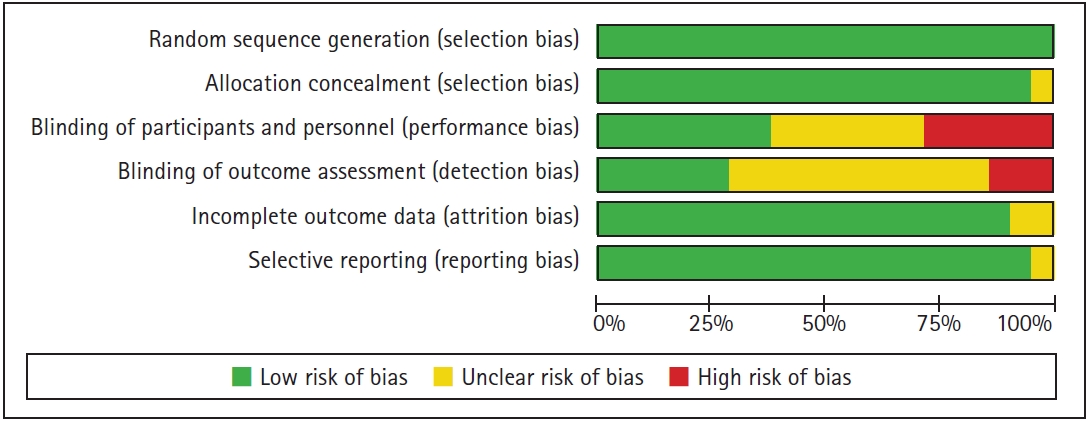
Individual studies were assessed using the Cochrane Collaboration’s Risk of Bias tool [46] and ranked according to a low/high/unclear grading scale (Fig. 2). The overall quality of the 21 included studies was moderate. Some of the studies showed possible patient selection bias and bias in methodology, with 70% showing an unclear or high risk of bias in performance concealment, 25% in blinding of participants and personnel, and 55% in blinding of the outcome assessment. A comparison-adjusted funnel plot showed evidence of a visually symmetric plot of opioid consumption and pain scores during the three time periods. The results of Egger’s regression test of outcomes also showed no significant publication bias (P > 0.05) (page 14 of each Supplementary Materials 1 to 4). The quality of evidence was rated as very-low-to-low in nature according to the GRADE system (Table 2), and the confidence rating of each comparison using CINeMA is described in the supplement file (Supplementary Material 5).
Heterogeneity and consistency test results
The results of the I2 and Q statistics (based on the full design-by-treatment interaction random-effects model) indicated that a random-effects model may be suitable for revealing any inconsistency or heterogeneity in our network model (Table 2). Additionally, according to the colored background of the net heat plot, the random-effects model appeared to be suitable for our data (pages 10 and 11 of each Supplementary Materials 1 to 4). A direct evidence plot (page 5 to 6 of each Supplementary Materials 1 to 4) and forest plot of the net splitting results (page 11 to 12 of each supplementary files, S1 to S4) were used to evaluate local inconsistency.
Efficacy outcomes (network meta-analysis)
Of the included studies, 17 [25–28,30,31,33,35,37–45] RCTs reported opioid consumption and 18 [25,26,28,30–36,38–45], 16 [25,26,28–35,38–44], and 17 [25–27,29,30,33–36,38–45] RCTs reported pain scores for each of the three postoperative time periods (early, middle, and late, respectively). The networks for the TPVB and control were greater than the networks for other blocks, followed by the ESPB and control. As shown in Fig. 3, TPVB had the best analgesic effect on opioid consumption compared with the control (mean difference [MD]: −13.2 mg, 95% CI [−16.2, −10.1]), followed by INB (MD: −9.55 mg, 95% CI [−13.2, −5.9]), ESPB (MD: −8.7 mg, 95% CI [−11.4, −6.1]), and SPB (MD: −5.9 mg, 95% CI [−9.4, −2.5]). In terms of pain scores in the early period, ESPB had the greatest effect compared with the control (MD: −1.6, 95% CI [−2.3, −0.9]), followed by TPVB, INB and SPB. In the middle and late periods, TPVB, ESPB and INB showed superior analgesic effects in reducing pain scores compared to the control, whereas SPB did not have a significant effect. The local inconsistency in ESPB and control was significant in the early and middle periods (Table 2). The two studies by Ciftci et al. [34,35] had effect sizes that tended to be higher than those measured in the other studies. Table 3 shows the network league table that displays the direct comparison and full model results separately.
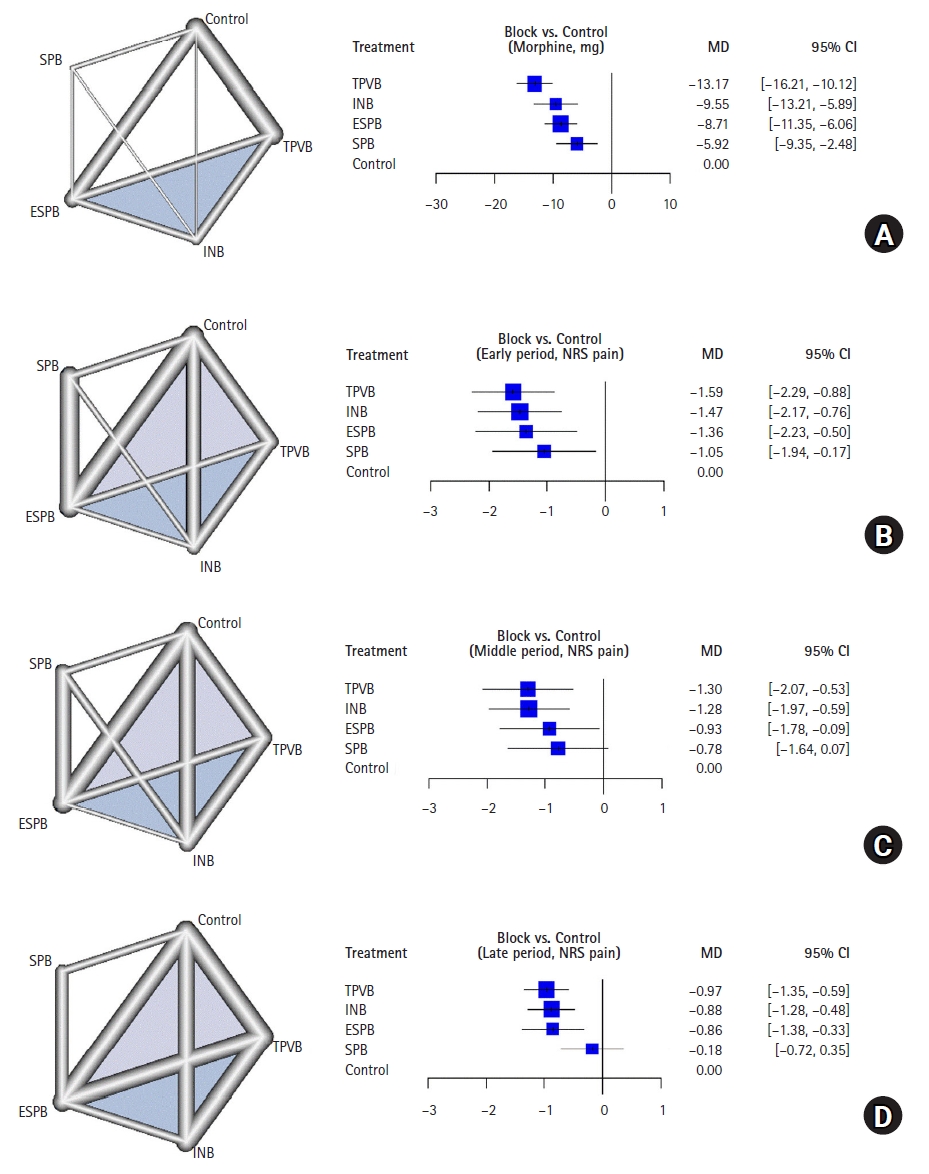
Network plots and forest plots for the network meta-analysis. (A) opioid consumption in the first 24 h post-operation, (B) early postoperative period (up to 6 h) pain scores, (C) middle postoperative period (6–18 h) pain scores, and (D) late postoperative period (18–24 h) pain scores. The mean difference (MD) and 95% CI are shown. ESPB: erector spinae plane block, INB: intercostal nerve block, SPB: serratus plane block, TPVB: thoracic paravertebral block.
Results of the ranking hierarchy
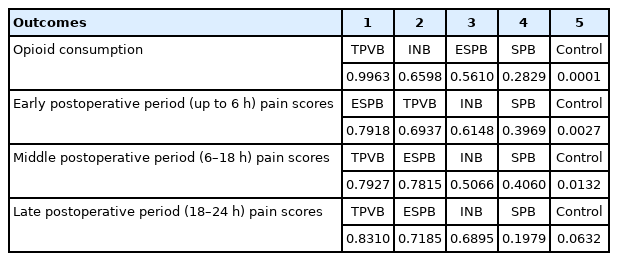
Table 4 shows the P-scores of analgesic efficacy and the ranking of the five groups. TPVB ranked highest for opioid consumption in the first 24 h (0.996) and for middle- and late-period pain scores (0.793 and 0.831, respectively). However, ESPB ranked first for pain scores in the early period (0.792). INB ranked second for opioid consumption and third for pain scores in all three periods. SPB ranked fourth for all the outcomes.
Additional analysis after removal of retracted article
During proofreading, we noticed that the article which included our analysis had been retracted from its journal [37]. Thus, we performed an additional analysis after removing the retracted article. That retracted paper only had information on the 24 hours opioid consumption, and there was no significant change in effect estimate of opioid consumption, P-scores nor ranking of the Included blocks (Supplementary Material 6).
Discussion
Various regional analgesia techniques are used in clinical settings to improve postoperative pain management in VATS, and our NMA not only demonstrated the potential benefits of these but also ranked them based on efficacy. When compared with mere systemic analgesia, all four regional analgesic techniques significantly reduced cumulative opioid consumption during the first 24 h postoperative period. In particular, TPVB showed remarkable effectiveness in reducing opioid consumption. Additionally, ESPB ranked highest for lowering the pain score in the early postoperative period, while the effect size of TPVB was clinically similar to that of ESPBs. In the case of SPB, however, even though the statistical significance of opioid consumption was clear, the effect size was approximately half that of the other methods. Moreover, the pain scores measured in the middle and late periods were not significantly different compared to control.
Statistically significant differences are not always clinically significant— e.g., a difference of 10 mg or more in parenteral morphine [47], and a change of 10 mm in a 100 mm visual analog scale are regarded as clinically significant [48]. In our opinion, changes of about 1–2 points in the pain score for patients who had initially addressed moderate-to-severe pain should be considered a clinically significant difference. In addition, changes in the pain score from initial values of 4–5 points to values < 3 were considered to be a clinically significant difference. In fact, a score < 33 points on a 100-point VAS is accepted as a state of well-controlled pain in a clinical setting [48].
TPVB showed a reduction of 13.2 mg in opioid consumption along with a reduction of more than 1 point in the pain score, which were viewed as clinically significant. For ESPB, the reduction in opioid consumption was 8.71 mg, which was less than 10 mg, but the decrease in the pain score was 1.6, showing the best results in the early postoperative period. However, this result had direct and indirect inconsistencies; therefore, caution should be taken when interpreting these results. Two studies, which were performed by Ciftci et al. [34,35] and included in our NMA, compared ESPB to control with a very large effect size compared to the others, which may have led to this inconsistency.
ESPB is an emerging technique that has been widely applied in multiple fields. Importantly, it can be easily administered even by trainees [15]. Its analgesic effect has been verified in various studies [49-51]. However, its mechanism is not well understood. The most convincing hypothesis is that the local anesthetic physically spreads to the thoracic paravertebral space and the associated neural structures [52]. Penetration via diffusion into the paravertebral space through the intertransverse connective tissue complex may continue over a prolonged period. Therefore, if anterior spreading to the thoracic paravertebral space is sufficient, ESPB should provide an effect similar to that of TPVB. However, studies that have compared ESPB and TPVB have found a significant difference in analgesic effect between the two blocks [38,49]. Improvement in postoperative pain scores and a reduction in opioid consumption were found to be better with TPVB than with ESPB. Interestingly, in contrast to results with single ESPB, a comparison of continuous infusion through a catheter showed that ESPB was noninferior to TPVB [53]. In both groups, a continuous infusion of 8 ml/h following a 20 ml bolus injection was performed. In the early postoperative period, TPVBs presented favorable results with regard to pain scores compared to ESPBs, but in the long term, the effects of the two blocks were similar, and thus no difference in opioid consumption was observed. If the mechanism of action involves anterior spreading to the thoracic paravertebral space by gradual diffusion, continuous infusions may be more effective than a single injection. However, to reduce heterogeneity, we only included RCTs using the single-block technique. Continuous TPVB using a catheter is still recommended for thoracotomy by the PROSPECT group [14], but it is questionable whether continuous blocks are necessary in VATS, as it is a minimally invasive surgical technique. In most of the RCTs included in this NMA, pain scores during the middle and late periods were mild (NRS < 3) in the control group. Therefore, multimodal analgesia, including regular acetaminophen, NSAIDs, and adjuvants to prolong the blocks may be sufficient for VATS [54,55].
According to a recent Cochrane review, while TPVB was as effective as TEA for controlling acute pain, TPVB was associated with fewer complications, such as hypotension, urinary retention, nausea, and vomiting [56]. Owing to these advantages, TPVB has recently been preferred to TEA for thoracic surgery. For other surgeries (e.g., breast surgery), the excellent analgesic effect of TPVB is offset by concerns about the potential risk of pneumothorax [57]. However, concerns about pneumothorax are greatly reduced in VATS, which allows for the administration of TPVB without concern for this complication.
INB is a well-known traditional technique for pain management after thoracic surgery. INB can be performed easily using various techniques, such as ultrasonography or blind techniques. In addition, a thoracic surgeon can directly inject inside the thorax [30]. INB only result in a segmental somatic nerve blockade, and thus multiple injections are necessary for appropriate pain control. Therefore, one might expect that the effect size of INB would be similar to that of SPB. Although only two direct comparisons between INB and SPB were included in this NMA, no differences in analgesic effect was found.
SPB can be easily performed in the lateral decubitus position, which is the surgical position for thoracic surgery [58]. Although the analgesic effect of SPBs was comparable to that of TEA in a previous study [59], our NMA results showed only a limited effect in the early postoperative period. The reduction in opioid consumption was less than half that found with TPVB. Among the four block techniques, only SPB was adequate to block the long thoracic nerve, which controls pain derived from damage to the serratus muscle and strain on surrounding structures [60]. Blockades of the long thoracic nerve have been found to reduce postoperative pain after VATS [61]. In addition, there is growing evidence that motor nerves are also involved in afferent nociception via sensory innervation and connection with other nerves [62,63]. However, the clinical effects of long thoracic nerve blockade do not meet expectations and this is attributed to trivial muscle damage due to VATS, which does not significantly affect postoperative pain.
This study has several limitations. First, the included studies were highly heterogeneous. Although the present study included only RCTs in patients who underwent VATS, the concentrations of drugs and technical details were not consistent. In addition, various drugs were used for multimodal analgesia. Second, the time points at which pain scores were measured were not consistent between studies and were not always presented as accurate values. To reduce any bias, we divided the time period into three intervals and used the values corresponding to each interval as representative values. Third, the sample size was insufficient to draw definitive conclusions. Lastly, ESPB and SPB are currently developing techniques, which may lead to possible publication bias. In conclusion, in this study, NMA was conducted to compare regional analgesia techniques in terms of their efficacy at improving postoperative pain control after VATS. TPVB showed outstanding analgesic effects and ESPB led to the greatest reduction in pain scores during the early postoperative period. However, given the significant reduction in opioid consumption seen with all the four regional analgesic techniques evaluated, using any of these regional blocks after VATS seems reasonable. Further and more refined studies are needed to determine the optimal regional analgesia technique to improve postoperative pain control after VATS.
Notes
Funding
This work was supported by research funding from the National Research Foundation of Korea (NRF-2019R1G1A1099660).
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Author Contributions
Yumin Jo (Writing – original draft; Writing – review & editing)
Seyeon Park (Data curation; Formal analysis; Methodology; Visualization)
Chahyun Oh (Investigation; Software; Validation)
Yujin Pak (Methodology; Project administration; Resources)
Kuhee Jeong (Visualization; Writing – review & editing)
Sangwon Yun (Visualization; Writing – review & editing)
Chan Noh (Formal analysis; Resources; Validation)
Woosuk Chung (Conceptualization; Writing – review & editing)
Yoon-Hee Kim (Resources; Supervision; Writing – review & editing)
Young Kwon Ko (Conceptualization; Supervision; Writing – review & editing)
Boohwi Hong (Conceptualization; Data curation; Supervision; Writing – original draft; Writing – review & editing)
Supplementary Materials
24 hours opioid consumption.
Pain score at postoperative early (up to 6 hours) period.
Pain score at postoperative middle (6 to 18 hours) period.
Pain score at postoperative late (18 to 24 hours) period.
Confidence rating of each outcomes.
24 hours opioid consumption after removal of retracted article.