Clot-in-transit cardiopulmonary thrombosis during convalescent phase of COVID-19
Article information
Acute pulmonary thromboembolism (PTE) is a known complication in patients with respiratory symptoms of coronavirus disease 2019 (COVID-19) [1]. However, acute PTE is rarely reported as a late sequela in asymptomatic or minimally symptomatic COVID-19 [1–3]. We herein present, with consent for publication, a patient with asymptomatic nasopharyngeal reverse transcriptase–polymerase chain reaction (RT-PCR) swab-positive COVID-19 who developed massive acute PTE during the convalescent phase, nearly 4 weeks after two consecutive negative nasal swab results.
A 41-year-old man (weight: 99 kg, height: 177 cm), was diagnosed with asymptomatic COVID-19 disease based on a positive nasopharyngeal SARS-CoV-2 RT-PCR and household contact with a proven COVID-19 case. He had no prior comorbidities, except for deep vein thrombosis induced by lower limb cellulitis 5 years earlier, for which he received anticoagulation therapy for 6 months and had complete recovery. The COVID-19 was managed on home isolation for 16 days until two consecutive nasal swab tests, performed on day 11 and day 15 of the disease, were negative. During this period, he remained ambulatory and received low-dose enoxaparin prophylaxis for 4 weeks. Nearly 42 days following the disease onset, he presented with dyspnea, hypoxemia (room-air oxygen saturation of 91%), and hypotension. Serum D-dimer (> 35,200 ng/ml), C-reactive protein (94.8 mg/L), and troponin-T (1.196 µg/L) levels were elevated. A repeat RT-PCR swab test result was negative. Transthoracic echocardiography showed a large freely mobile right atrial thrombus measuring 5 cm × 0.8 cm attached to the posteroinferior wall, normal left ventricle, dilated right ventricle, severe pulmonary arterial hypertension (systolic pressure 60 mmHg), and moderate tricuspid regurgitation. CT chest images did not show pulmonary consolidation. CT pulmonary angiogram showed extensive right and left pulmonary artery thrombosis and an enlarged right atrium and right ventricle with filling defects. In the first 24 hours, he was treated with intravenous fluids, inotropes, and tissue plasminogen activator, resulting in an improved blood pressure of 110/75 mmHg, heart rate of 76 beats/min, and oxygen saturation of 96% on 2 L/min nasal oxygen. Subsequently, therapeutic-dose subcutaneous enoxaparin was initiated, and transesophageal echocardiography (TEE) was performed to delineate the extent of cardiac thrombosis. 2D and 3D TEE showed a large right atrial thrombus attached to the Chiari network, a strangulated thrombus measuring 1.1 cm × 0.3 cm crossing a patent foramen ovale, thrombosis of both main pulmonary artery branches, right ventricular dilatation with mild dysfunction, and minimal tricuspid regurgitation (Fig. 1). Later, due to significant hemodynamic instability, an emergency pulmonary thrombectomy under cardiopulmonary bypass was performed. Immediate post-bypass TEE showed complete resolution of pulmonary arterial thrombosis; however, severe right ventricular dysfunction was present. Nonetheless, he was weaned from mechanical ventilation the next day and inotropes after 3 days. Follow-up echocardiography a week later showed no residual thrombus and normal right ventricular function.
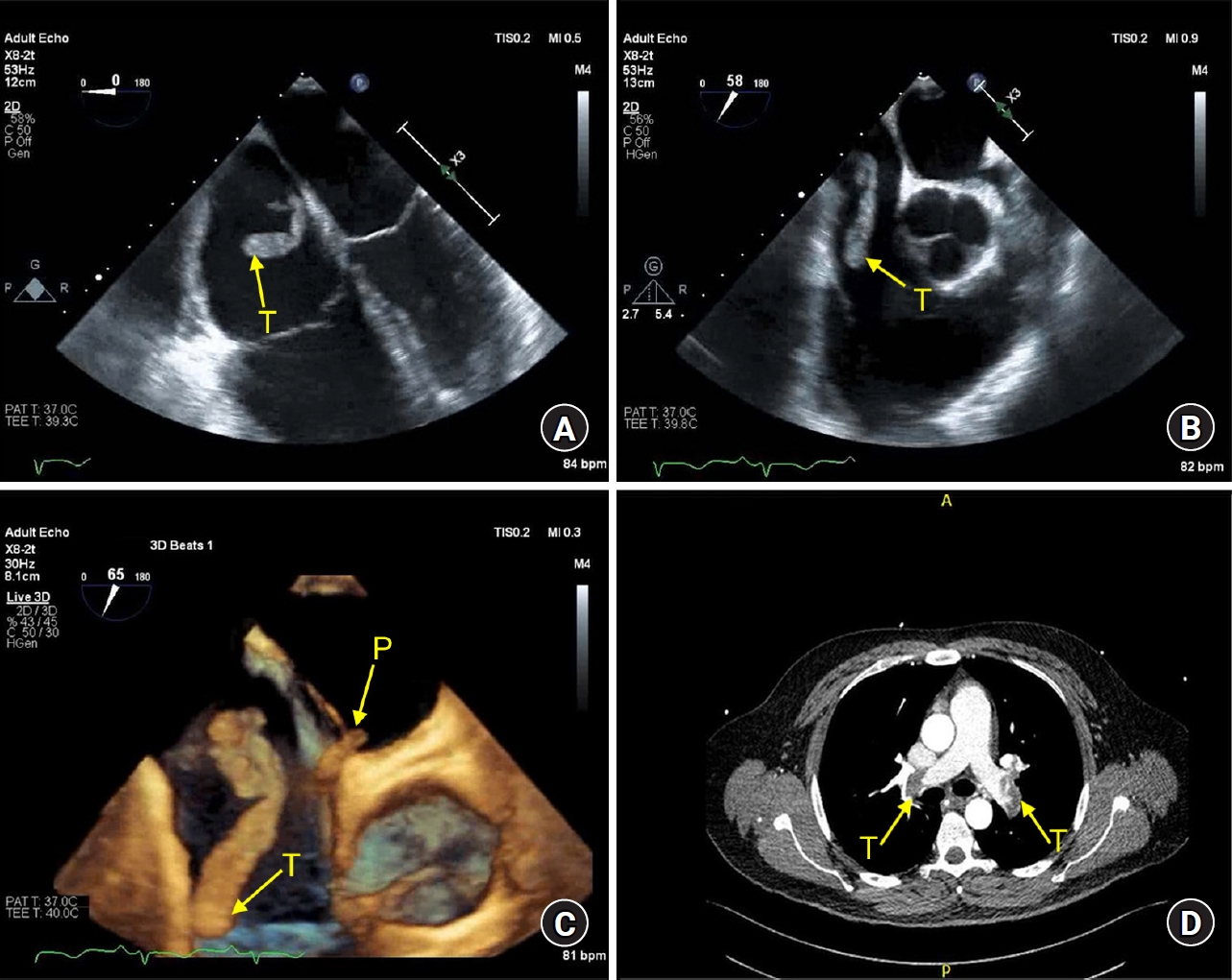
(A) 2D transesophageal echocardiogram images show a large right atrial thrombus (T) in 4-chamber view and (B) right ventricular inflow-outflow view. (C) 3D transesophageal echocardiogram image shows thrombus-in-transit through a patent foramen ovale (P). (D) CT pulmonary angiography shows bilateral pulmonary thrombi.
While thromboembolic complications occur with an incidence as high as 20% to 30% in patients with severe COVID-19 [1], only a few reports have described such complications following recovery from mild COVID-19 [1,2]. A single-center report of 163 patients without post-discharge thromboprophylaxis suggested a 2.5% cumulative incidence of thrombosis at 30 days following discharge, including segmental pulmonary embolism, intracardiac thrombus, thrombosed arteriovenous fistula, and ischemic stroke [4]. Tu et al. [5] recently reported 10 cases of acute ischemic stroke in adults aged ≤ 50 years in the convalescent phase of asymptomatic COVID-19. The median (Q1, Q3) time from a positive serological test result to acute ischemic stroke was 54.5 (0, 130) days. However, none of these patients had received thromboprophylaxis. Thus, the present case is unique, as the patient developed massive acute pulmonary embolism nearly 42 days post-asymptomatic COVID-19 despite thromboprophylaxis.
Further, this case highlights the possibility of prolonged COVID-19-associated coagulopathy (CAC) following asymptomatic SARS-CoV-2 infection resulting in massive cardiopulmonary thrombosis in the convalescent phase. Pow et al. [2] reported a similar case of submassive pulmonary embolism during the convalescent phase in a patient with minimally symptomatic SARS-CoV-2 infection. However, as their patient had a positive nasal swab result on readmission, it raised the possibility of reinfection resulting in a hypercoagulable state instead of prolonged CAC as a cause of submassive pulmonary embolism. Probable mechanisms of CAC and thrombosis in post-acute COVID-19 syndrome include dysregulated renin-angiotensin system activity, endothelial dysfunction, cytokine-induced inflammation, complement and platelet activation, platelet-leukocyte interaction, neutrophilic extracellular traps, lupus anticoagulant, and up-regulation of tissue factor, thrombin, and fibrin with down-regulation of fibrinolytic and anticoagulant mechanisms [1,2,4]. The risk of thrombotic complications in the post-acute COVID-19 phase is probably linked to the duration and severity of a hyperinflammatory state, although how long this persists is unknown [4].
Given the increase in thrombotic complications in post-acute COVID-19, Nalbandian et al. [4] argued that extended post-hospital follow-up (up to 6 weeks) and prolonged primary thromboprophylaxis (up to 45 days) in those managed as outpatients may have a more favorable risk–benefit ratio. However, individual patient-level considerations for risk versus benefit should dictate recommendations at this time. Elevated D-dimer levels, in addition to comorbidities such as cancer and immobility, may help to stratify patients at the highest risk of post-acute thrombosis [4]. Notably, increased D-dimer levels (> 500 ng/ml) were observed in 25.3% of patients up to 4 months post-SARS-CoV-2 infection [3]. Thus, elevated D-dimer levels and history of deep vein thrombosis (even though not associated with hypercoagulable disorders) might have placed our patient at high risk of thrombotic complications despite thromboprophylaxis and ambulation. Furthermore, as optimal therapy is not defined for COVID-19-associated thrombosis, thrombolytic therapy might have resulted in an unfavorable outcome in our patient.
In conclusion, we highlight the possibility of late-onset acute COVID-19-associated thrombosis even in asymptomatic infections, the lack of an established dose and duration of thromboprophylaxis, and the risk of failure of thrombolytic therapy. Close patient follow-up is warranted in the convalescent phase of COVID-19.
Notes
Funding
None.
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Author Contributions
John George Karippacheril (Conceptualization; Data curation; Formal analysis; Investigation; Resources; Writing – original draft; Writing – review & editing)
Amit Jain (Conceptualization; Writing – review & editing)
