The effects of etomidate and midazolam on adipose tissue-derived mesenchymal stem cell proliferation
Article information
Abstract
Background
Stem cell therapy using adipose tissue-derived mesenchymal stem cells (ADSCs), which are capable of multipotent differentiation, is currently being investigated in the field of tissue regeneration and the treatment of patients in intensive care units. It is known that type-A γ-aminobutyric acid (GABAA) receptor activity has an influence on stem cell proliferation. Thus, we investigated the effects of the clinically available GABAA receptor agonists, etomidate and midazolam, on ADSC proliferation measured by the cell counting kit-8 assay.
Methods
ADSCs cultured in control medium or adipogenic differentiation medium for 15 days were divided into 5 treatment groups: non-medicated (Control) and 4 groups including treatment with etomidate or midazolam at 1 and 50 µM (n = 3 per group). The cell counting kit-8 assay was performed for determining the cell proliferation in both medium groups at day 0, 3, 6, 9, 12, and 15 in culture. The absorbance values at 450 nm were then measured by enzyme-linked immunosorbent assay reader and statistically compared among groups.
Results
There was no significant difference in cell proliferation profiles among the 5 groups at any time point in both control and adipogenic differentiation media.
Conclusions
Etomidate and midazolam did not influence ADSC proliferation under both media when compared to the non-medicated group and there was no dose-dependent effect of etomidate and midazolam on ADSC viability.
Introduction
Mesenchymal stem cells (MSCs), a type of adult stem cells, are distributed in various tissues and are capable of multipotent differentiation into osteogenic, chondrogenic, and adipogenic cell lineages [1]. It is acknowledged that a population of MSCs is isolated from adult human adipose tissues, which can be harvested in large amounts by liposuction [2]. In recent years, clinical trials with adipose tissue-derived MSCs (ADSCs) are being attempted. To achieve improved fat-graft survival, ADSC based-tissue engineering and regeneration are recently co-introduced in the surgical fields where autologous fat grafts alone have been in charge [3456]. Recent studies have also investigated whether stem cell therapy with ADSCs can be used for improving the outcome of patients in intensive care units, including the acute urogenital organ damage observed in sepsis [7] and toxic shock syndrome [8]. Several factors should be considered to achieve successful stem cell therapy [9]. Among these, maintaining the applied stem cells at an active proliferation state, or the maintenance of cell viability, is essential to achieve stem cell therapy objectives.
Previous studies raise the possibility that the type-A γ-aminobutyric acid (GABAA) receptor is involved in modulating stem cell proliferation [1011]. Currently, well-known intravenous GABAA receptor agonists, etomidate, and midazolam, are used for sedation of patients in surgery or in intensive care units. They act through the enhancement of GABA mediated inhibition via the synaptic GABAA receptor [1213].
However, the effect of clinically available GABAergic agents on ADSC proliferation is not known. Thus, our study investigated the effects of the clinically available GABAergic agents, etomidate and midazolam, on ADSC proliferation both in control medium and under induction of adipogenic differentiation using the Cell Counting Kit-8 (CCK-8) assay.
Materials and Methods
Isolation and preparation of ADSCs
Ethical approval for this study protocol was obtained from the Institutional Review Board of Kangbuk Samsung Hospital (IRB File No: 2011-01-031). The ADSCs used in this study were obtained from the lipoaspirates of donors after receiving informed consent, using a previously reported method [1415]. In brief, the lipoaspirates were washed extensively with sterile Dulbecco's phosphate buffered saline (DPBS) (Biowest, Rue de la Caille, Nuaillé, France) to remove the contaminating debris and blood cells. The rinsed aspirates were treated with 0.075% collagenase type I in DPBS for 60 min at 37℃ under gentle agitation and then inactivated with an equal volume of Dulbecco's modified Eagle's medium (DMEM) (Gibco, Grand Island, NY, USA)/ 10% fetal bovine serum (FBS) (Gibco, Grand Island, NY, USA). The cell pellet collected by low speed centrifugation for 10 min was resuspended in DMEM containing 10% FBS and strained through a 100-µm mesh filter to remove debris. After centrifugation as detailed above, the filtrate was plated onto conventional tissue culture plates in control medium (DMEM, 10% FBS, 1% antibiotic/antimycotic solution [Gibco, Grand Island, NY, USA]) and incubated at 37℃ in a CO2 incubator. The control medium was replaced with fresh medium after every 3 days. During the study period, ADSCs were maintained at a density of 8 × 104 cells per well in a 6-well plate in the control medium. The ADSCs for this study were from passages 3–5.
Induction of Adipogenic differentiation in ADSCs
ADSCs were incubated at a density of 8 × 104 cells per well in a 6-well plate in complete medium for 24 h at 37℃ in a CO2 incubator. After confirming cell adhesion, ADSCs were cultured with adipogenic differentiation medium comprising 1 µM dexamethasone (Sigma, St. Louis, MO, USA), 1 µM indomethacin (Sigma, St. Louis, MO, USA), 500 µM 3-isobutyl-1-methylzanthine (Sigma, St. Louis, MO, USA), and 10 µg/ml insulin (Sigma, St. Louis, MO, USA) in complete medium [16]. Cell differentiation was induced for 15 days with media replacement once in every 3 days.
Cell treatment with GABAergic anesthetics
The ADSCs under control or adipogenic differentiation medium were divided into the non-medicated group and 4 experimental groups based on the type and amount of GABAergic agents applied to the cells (n = 3 for each group). ADSCs cultured in control or differentiation medium were simultaneously treated with GABAergic anesthetics. During medium changes, the prepared etomidate (Lipuro® 2% inj, B. Braun, Melsungen, Germany) or midazolam (Midazolam® 1% inj, Bukwang pharmaceutical. Co. Ltd., Ansan, Korea) was added to the cultured ADSCs to achieve final concentrations of 1 and 50 µM. The concentration of each agent was determined based on previous references; the optimal plasma concentrations of each agent for sedation were 1 µM for etomidate and 1 µM for midazolam [17], and the concentration of etomidate and midazolam resulting in declined cardiac performance was beyond 50 µM [18].
Determination of proliferative ability of ADSCs
The CCK-8 assay allows sensitive colorimetric determination of cell viability in cell proliferation and cytotoxicity assays. A highly water-soluble tetrazolium salt, WST-8 [2-(2-methoxy-4-nitrophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2H-tetrazolium, monosodium salt], is reduced by the dehydrogenase activity in cells to generate a yellow-colored formazan dye, which is soluble in tissue culture media. The amount of the formazan dye, generated by the dehydrogenase activity in cells, is directly proportional to the number of living cells. The assay was conducted in ADSCs at 0, 3, 6, 9, 12, and 15 days after induction of cell proliferation or adipogenic differentiation according to the manufacturer's instructions. In brief, ADSCs cultured in a 6-well plated were washed with PBS and then incubated in the dark with a mixture of control medium and the CCK-8 reagent at a ratio of 20 : 1. After 90 min at 37℃ in a 5% CO2 incubator, the cells were analyzed using an enzyme-linked immunosorbent assay (ELISA) reader with the wavelength set to 450 nm. The absorbance values from the ELISA reader were used to determine the degree of cellular proliferation.
Statistical analysis
To prove whether cell viability values measured by the CCK-8 assay at each time point showed normal distribution, the Kolmogorov-Smirnov test was performed. Since the data showed non-normal distribution at some measured time points, the Kruskal Wallis test was used to compare the data among 5 comparison groups.
Statistical significances were determined after Bonferroni's method to adjust type I error. The data were analyzed using PASW Statistics 18.0 (SPSS Inc., Chicago, IL, USA). Values of P < 0.05 were considered statistically significant.
Results
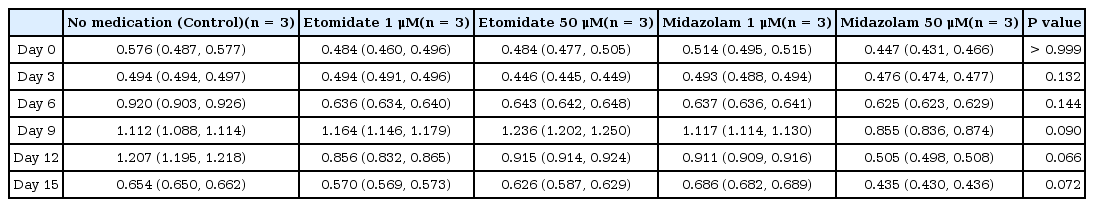
Values obtained from the CCK-8 assay are presented as median (Q1-Q3) (Tables 1 and 2). According to the Kolmogorov-Smirnov test, the results of the CCK-8 assay were not normally distributed at day 12 and 15 in control media (both P < 0.001), and at day 6 and 9 in adipogenic differentiation media (P < 0.001 and P = 0.001, respectively). Thus, because the data were not normally distributed, the statistical analyses were performed using Kruskal-Wallis test to investigate the differences in cell proliferation among groups.
Results of the CCK-8 Assay for Adipose Derived Mesenchymal Stem Cells in Adipogenic Differentiation Medium
In the control medium, no statistical difference in cell viability was observed at 0, 3, 6, 9, 12, and 15 days among the control group and the midazolam and etomidate groups with a dose for sedation, at 1 µM, and a high dose for suppression of cardiac function, at 50 µM (Table 1). Even under adipogenic differentiation medium, the dehydrogenase activities of viable cells measured by the CCK-8 assay were not statistically different at any of the time points among the control and medication groups (Table 2).
Discussion
The present study revealed that the clinically available GABAA receptor agonists, etomidate and midazolam, did not influence the proliferation of ADSC when compared to that in the non-medicated group.
In the field of stem cell biology, it is known that GABAA receptor is expressed not only in neural stem cells [19], but also in different types of extraneural stem cells in the undifferentiated proliferative state [1020]. As observed with embryonic stem cells [10], the GABAA receptor, which is a binding site for etomidate or midazolam, is also expressed in human ADSCs [21]. However, the GABAA receptor subunit composition in ADSCs is not yet reported, even though Andang et al. [10] reported that embryonic stem cells express the GABAA receptor subunits α1, α3-5, β3, ε, π, and θ; they did not examine the ρ subunits.
Moreover, GABAA receptor is involved in modulating the cell cycle, especially DNA synthesis [1022], and the activity of the GABAA receptor on stem cells or on progenitor cells has an influence on cell proliferation [1011]. Additionally, GABA along with the activating GABAA receptor is known as a cell-cycle regulator [2223]. Previous studies like these raised the possibility that clinically available GABAA receptor agonists, like etomidate and midazolam, might affect stem cell proliferation.
The specific role of each subunit of the GABAA receptor in the cell cycle was recently demonstrated in studies on hepatocellular carcinoma cells [24].
Thus, we suppose that cell viability might differ between ADSCs treated with etomidate and midazolam because both GABAergic agents have different binding affinities on the specific subunit of the GABAA receptors [2526], which determines the potency and efficacy of each agent.
The etomidate binding sites are discovered near the α1 and β3 subunits of the GABAA receptor [27]. Etomidate is highly sensitive to the receptor containing the β2 or β3 subunit, but not the β1 subunit [25]. In animal studies, β2 and β3 subunits are shown to play different roles in the anesthetic action of etomidate [28]. The β3 subunit participates in the loss of consciousness whereas β2 is involved in the sedative properties of agent.
Midazolam is a kind of benzodiazepine, which produces various actions by binding to its specific site on the GABAA receptor that is composed of the respective α-subunit (α1, α2, α3, α5) and the γ2-subunit. Each subunit of the benzodiazepine site plays a role in the pharmacologic effects of various agents [29]. The anesthetic action of midazolam is obtunded in the absence of the β3 subunit [26]. The modulatory effect of midazolam on GABA activated currents via the GABAA receptor is negatively influenced by the co-expression of both α6 and β3 subunits [30].
However, our study revealed that the clinically available GABAA receptor agonists, etomidate and midazolam, did not modulate the proliferation of stem cells.
However, our study has the limitation that the sample size used was not sufficient to perform parametric statistics. Thus, a further study with a sufficient sample size is needed to confirm the effect of GABAergic anesthetics on ADSC proliferation.
In conclusion, the current study demonstrates that the clinically available GABAergic anesthetics, etomidate and midazolam, did not influence the proliferation ability of ADSCs in control medium. It is also suggested that these anesthetics do not influence the differentiation ability of these cells as observed by the absence of changes in cell proliferation after mixing anesthetics with stem cell differentiation inducers. Additionally, although it is known that etomidate and midazolam have different affinities for the GABAA receptor subunits, they showed similar effects on ADSC proliferation both in the control and adipogenic differentiation media.
Acknowledgments
This study was supported by a grant (No. 2011-0014224) from the National Research Foundation of Korea.

