Use of LMAⓇ GastroTM in esophagogastroduodenoscopy and endoscopy
Article information
With increasing complexity and duration of minimally invasive upper gastrointestinal (GI) endoscopy, the need for deep sedation or general anesthesia (GA) during these procedures has grown rapidly. Therefore, the ability to secure the airway while allowing an easy endoscopic access has also become more relevant. Sedation during esophagogastroduodenoscopy (EGD) is not risk-free. Deep sedation can occasionally turn into GA, particularly when propofol is used [1]. Sometimes, deeply sedated patients may also have patient state index levels associated with GA. Deep sedation can potentially risk airway and respiratory compromise [1]. In a retrospective analysis of 73,029 GI endoscopies, 72% of the peri-procedural cardiac arrests were associated with airway management [2].
The laryngeal mask airway (LMA) LMAⓇ GastroTM Airway (Teleflex Medical, Ireland) is a cuffed peri-laryngeal supra-glottic airway (SGA) with an endoscopic channel, having a maximum outer diameter of 14 mm, which suits all standard endoscopes. Its design features include a channel for esophageal intubation, a separate channel with a terminal cuff for lung ventilation, and an integrated bite block and cuff pressure indicator. It comes in three available sizes: #3 (30–50 kg), #4 (50–70 kg), and #5 (70–100 kg). In a recent prospective observational study, Terblanche et al. showed that LMAⓇ GastroTM had an airway insertion success rate of 99% and a first-attempt endoscopy success rate of 93% in 292 patients with low risk of pulmonary aspiration. The median lowest intraoperative oxygen saturation was 98% [3].
We describe our experiences with the use of LMAⓇ GastroTM Airway in two patients undergoing upper GI endoscopy. Both patients provided written informed consent for the publication of this study. In both cases, a suitably sized LMAⓇ GastroTM was inserted after the standard routine general anesthesia protocol, and GA was maintained using volatile agents.
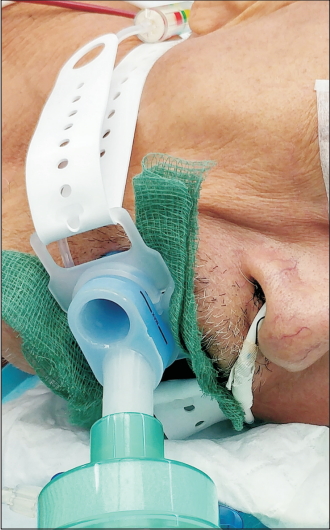
Case 1 was that of a 65-year-old Chinese man (height, 175 cm; weight, 58.9 kg; body mass index, 19.23 kg/m2) with a history of hypertension and recurrent esophageal cancer with previous Ivor Lewis esophagogastrectomy. He underwent EGD and stenting of the recurrent esophageal cancer under GA using LMAⓇ GastroTM #4 inserted successfully in the first attempt after induction by a senior anesthesiologist (Fig. 1). Subsequently, the cuff was inflated with air until the integrated cuff pressure indicator was at the green level, which corresponded to a pressure of 60 cmH2O on the manometer. SGA positioning was satisfactory with a grade 1 equivalent laryngeal view when checked with a #5.5 flexible bronchoscope. The LMA cuff fit well around the glottis. The oropharyngeal leak pressure (OPLP) was 38 cm-H2O. A standard 12-mm EGD scope was inserted through the LMA’s endoscopic channel, and the endoscope was seen passing directly into the esophagus. However, the surgeon was unable to pass the stent delivery system through the esophageal stricture; therefore, a pediatric scope was inserted, and balloon dilatation with stent insertion was performed. The procedure took one and half hour.
Case 2 was of a 19-year-old Malay man (height, 178 cm; weight, 58.2 kg; body mass index, 18.37 kg/m2) who presented with persistent globus sensation for EGD evaluation. LMAⓇ GastroTM #4 was inserted successfully in the supine position under GA, and the cuff was inflated once again to a corresponding pressure of 60 cmH2O on the manometer. The LMA cuff fit well around the glottis with an OPLP of 37 cmH2O. An Olympus GIF-H260 video gastroscope with a distal end diameter of 9.8 mm was inserted smoothly without any issue. The whole procedure took 45 minutes.
There were no unexpected intraoperative or postoperative complications in either case. Neither patient reported any oropharyngeal discomfort, such as sore throat or dysphonia, and there was no difficulty in introducing the gastroscope through the LMAⓇ GastroTM lumen. LMAⓇ GastroTM can also act as a longer version of the oral bite block, which is usually used to aid the insertion of the gastroscope and may help facilitate the passage of the gastroscope to direct it straight to the upper esophageal sphincter for an easier route.
To date, there have been no randomized trials comparing LMAⓇ GastroTM to the usual endoscopic procedures. Currently, in Singapore, LMAⓇ GastroTM is the only supraglottic airway device available for use in gastric endoscopy [4].
Endoscopic access in the airway can potentially help prevent complications of sedation, such as loss of airway, hypoventilation, hypoxemia, and pulmonary aspiration, while avoiding hemodynamic swings and paralysis associated with tracheal intubation. This is especially important in patients in whom sedation is more challenging because of pre-existing medical conditions, such as morbid obesity or severe sleep apnoea. It also helps shorten the extubation and recovery time compared to tracheal intubation. Moreover, LMAⓇ GastroTM can serve as a useful rescue device when the patient's airway is required to be secured during EGD in the lateral position compared to tracheal intubation, which can be challenging in such a position, thereby providing an airway that is less invasive than a tracheal tube but more secure than an oropharyngeal airway.
In conclusion, we report our experiences with the new LMAⓇ GastroTM Airway in upper GI endoscopy. We hope that this report may encourage more dedicated research on the advanced use of LMAⓇ GastroTM in various clinical settings, such as emergency situations and high-risk patients with an American Society of Anesthesiologists physical status of III or IV.
Notes
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Authors’ contribution
Wan-Ling Alyssa Chiew (Writing–original draft; Writing–review & editing)
Qingyan Chen (Writing–original draft; Writing–review & editing)
Leng Zoo Tan (Supervision; Writing–original draft; Writing–review & editing)