Introduction
The use of simulations in medical practice is an exciting new sub-specialty that is being widely used for teaching, especially to simulate rare events and prepare students for real-life emergency scenarios. Simulations have been used effectively for procedural training, with mannequins replacing real patients, to avoid exposing novice students to patients for learning [
1,
2]. The use of simulations in research is still limited to statistical models and a few experimental studies, although there are many unexplored potential applications [
2,
3].
With the availability of human patient simulator (HPS) machines that can precisely simulate various physiological and pathological conditions through input from computer-based software, the role of simulators in anesthesia has expanded to an advanced level [
4]. Newer types of HPSs are based on physio-pharmacological models, which include real-time monitoring and feedback while interventions are performed on the simulator. HPSs are therefore ideal for complex critical scenarios and, thus, an appropriate tool for clinical research based on such scenarios.
Preoxygenation and apnea oxygenation are established techniques for prolonging the duration of apnea during intubation attempts in operation rooms and intensive care units [
5–
8], but most studies have been performed only on certain oxygen (O
2) saturation levels because of ethical issues related to exposing patients to apnea and potential hypoxia [
9–
11]. Recent literature has documented the efficacy of high-flow O
2 administered through a nasal cannula to provide longer periods of apnea in operation rooms and to maintain oxygenation in patients with pulmonary pathologies [
12–
17].
Through this experimental study, we aimed to investigate the effect of O2 therapy on prolonged apnea using a high-fidelity HPS. This allowed for longer periods of apnea to be studied and analyzed without risking patient safety. The primary objective of our study was to assess the utility of an HPS for apnea simulation and O2 therapy research, and the validity of extrapolating the data to human patients. The secondary objective was to compare the efficacy of different oxygenation strategies using a simple nasal cannula during apnea in terms of the time to desaturation.
Materials and Methods
Setup
After approval from the Institutional Review Board (IRB no. 261/IEC/IM/NF/2019), an experimental simulation study using an HPS was planned over four months. The HPS (CAE Healthcare™, USA) is equipped to detect delivered O2 and is used along with an anesthesia workstation and monitor with hemodynamic and oxygenation parameters.
Functioning of the HPS
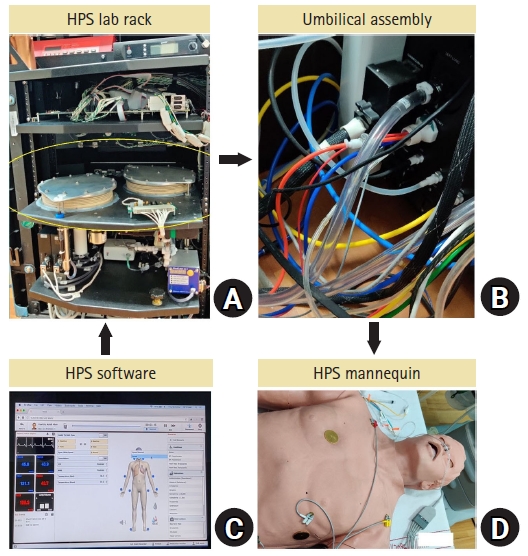
The study object was a high-fidelity HPS. The basic components of this HPS are a mannequin attached to a central control unit or lab rack (
Fig. 1A) through an umbilical assembly (
Fig. 1B). The lab rack is driven by various gases, such as O
2, nitrogen (N
2), carbon dioxide (CO
2), and compressed air. A specific software (Müse, CAE Healthcare™, USA) is used to control the functioning of the HPS (
Fig. 1C). Circulation and respiration are simulated through the lab rack. Two bellows inside the lab rack serve the function of the lungs (
Fig. 1A). Gas monitoring sensors are present at the level of the lab rack even though sampling is performed on the mannequin (
Fig. 1D). Simulated hemodynamic measurements, such as non-invasive blood pressure, pulse oximetry for O
2 saturation (SpO
2), and electrocardiography, can also be obtained from the mannequin using actual monitors.
Software
The HPS software, allows for two modes of functioning (
Fig. 1C). In the first mode, the HPS response is controlled by an operator at the computer interface, whereas with the second mode, responses are modeled based on normal adult physiology. Several parameters are set for normal adult physiology, such as the shunt fraction (set at 5% for this study), O
2 consumption, lung volume, and respiratory quotient, among others.
Table 1 shows a list of all the parameters that were set using the default values [
4]. The clinical hemodynamic and blood gas parameters, such as partial pressure of alveolar (PA), arterial (Pa), and venous (Pv) O
2 and CO
2, are visible on a separate screen, which can either be a fixed expected response based on the experiment, a setting that is applied in the software, or a modeled response after synchronizing the monitors with the HPS mannequin. Once in the modeled mode, the HPS behaves like an independent unit from the fixed software and responds physiologically to real-time interventions, such as external O
2 or anesthetic agents. For this particular study, we were specifically interested in the functioning of the respiratory system, simulation of lung function, and gas-sensing mechanisms. For gas monitoring, alveolar O
2 and CO
2 values were sensed at the level of the lab rack, and for all other arterial parameters, including pulse oximetry, the values were determined based on set physiological parameters.
Fig. 1 shows a schematic of the components and functions of the HPS system.
Experiments
Apnea settings were applied from baseline values, and changes in hemodynamic and blood gas parameters (PAO2, PaO2, PACO2, PaCO2, SpO2, pH) were recorded until the end of the experiment, which was defined as the attainment of 50% SpO2. The following four experiments were conducted with apnea: 1) no oxygenation (NO): only apnea was applied, 2) only apnea oxygenation (AO): 15 L/min O2 was provided via simple nasal cannula after apnea was applied, 3) only preoxygenation (PO): O2 was provided until the PAO2 reached 400 mmHg, at which time apnea was applied with no O2 during apnea; and 4) para-oxygenation (PAO): preoxygenation was provided until the PAO2 reached 400 mmHg, at which time apnea was applied and 15 L/min O2 was administered via simple nasal cannula during apnea. Alveolar and arterial gas levels were noted every 1 min from the beginning of the apnea period till 50% SpO2 was reached. Time to desaturation was noted for 92%, 75%, and 50% SpO2.
Settings and analysis
Initial O2 consumption was set at 250 ml/min. Each apnea experiment was repeated three times (at different sittings), and the mean values of the three recordings were used for the comparative analysis. Each set of four experiments was performed on different days to allow the machine to restart and equilibrate to baseline physiological values. This allowed us to maintain a quality check on the data and avoid erroneous readings. All experiments were then performed again at O2 consumption rates of 500 and 750 ml/min. The apnea trends were compared among all four experiments (NO, AO, PO, and PAO). The time to desaturation was compared between the experiments and between the different O2 consumption rates.
Results
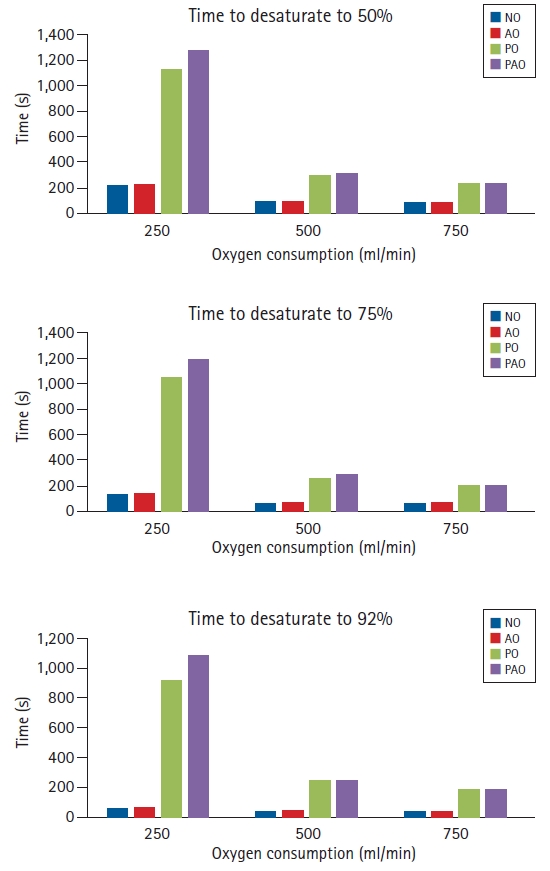
Fig. 2 shows a comparison of the desaturation time to 50%, 75%, and 92% SpO
2 among the four experimental settings at O
2 consumptions of 250, 500, and 750 ml/min. At 250 ml/min, NO (222.5 s) and AO (239 s) had a similar desaturation time to 50% SpO
2. PAO (1274.5 s) had a longer desaturation time to 50% SpO
2 than PO (1121 s), which was a 400% increase compared to NO. At higher O
2 consumption rates, the desaturation times were shorter. At 500 ml/min, the desaturation time to 50% SpO
2 for NO and AO was 97 s and 98 s, respectively. Compared with NO, the desaturation time to 50% SpO
2 for PO and PAO was 300 s and 316 s, respectively, which was an increase of approximately 200%. Similar comparative trends were observed for the desaturation time to 92% and 75% SpO
2 (
Fig. 2).
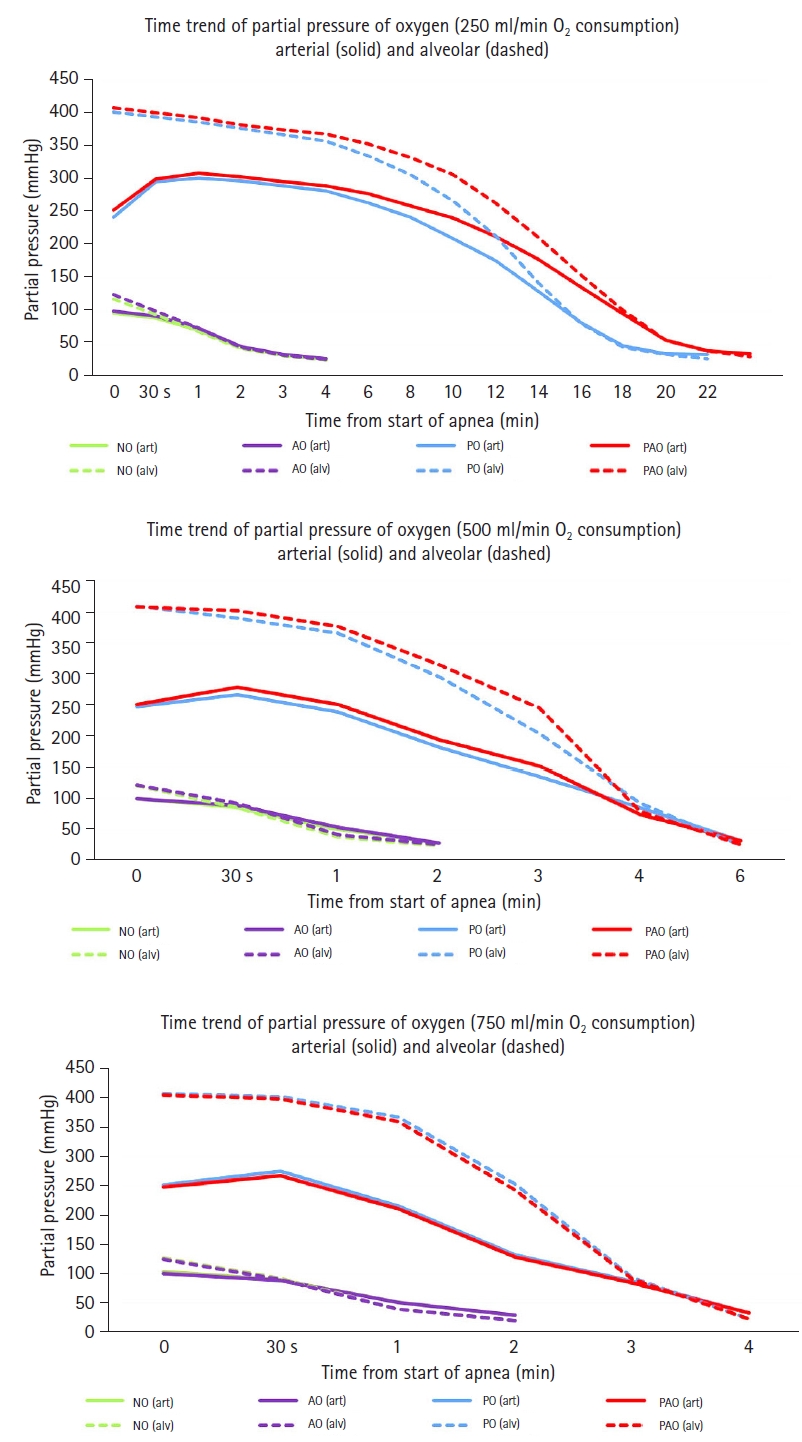
Fig. 3 shows a graphical trend of the partial pressure of O
2 in the alveoli (dashed line) and arterial blood (solid line) until the end of the each of the four experiments (i.e., once 50% SpO
2 was reached). The results are shown for all three O
2 consumption rates 250, 500, and 750 ml/min.
Table 2 shows the “end of experiment” values of PaCO
2 and pH for all four experiments and different O
2 consumption rates. Because of the shorter time to 50% SpO
2 at 250 ml/min O
2 consumption, the maximum PaCO
2 achieved was lower with NO (52.13 mmHg) and AO (52.9 mmHg) than with PO (72.5 mmHg) and PAO (75.1 mmHg), which allowed for longer apnea times and higher PaCO
2 values (
Table 2). Thus, lower pH values were observed with PO (7.2) and PAO (7.18), primarily due to respiratory acidosis. Similarly, experiments at higher O
2 consumption rates also had lower PaCO
2 and higher pH values than the 250 ml/min experiments due to the shorter times to desaturation (
Table 2).
Discussion
Recently, there has been growing interest in simulation/mannequin-related research. One advantage of such in vitro studies includes being able to perform research on complex clinical scenarios that are rarely encountered in practice or that would be a potential health risk to the patient [
2,
3]. One such scenario is apnea oxygenation. Also described as “apneic diffusion oxygenation,” this technique primarily involves drawing ambient O
2 “en masse” into the lungs by providing continuous flow through the airways [
18]. Devices such as nasal cannula have conventionally been used, though higher flow devices such as transnasal humidified rapid-insufflation ventilatory exchange (THRIVE), which allow for the toleration of longer apnea times in clinical situations, have been developed [
16].
Apnea oxygenation has been studied in humans since 1959. In one of the first studies, conducted by Frumin et al. [
18], patients undergoing elective surgeries under general anesthesia with neuromuscular blockades were subjected to prolonged apnea following a period of preoxygenation and de-nitrogenation. Although the apnea termination point was not fixed, periods of apnea ranged from 15 to 55 min, during which arterial samples were monitored for blood gas analysis and catecholamine levels. The lowest SpO
2 level reached was 98%, the lowest pH was 6.88, and the highest PaCO
2 was 250 mmHg. A similar study would be very difficult to perform in modern research settings due to the ethical issues of subjecting patients to such high levels of respiratory acidosis and CO
2. In the current study, a normal physiologically modeled HPS was used to study various oxygenation strategies during apnea to an extent that would not be feasible for real subjects (50% SpO
2). The lowest pH reached was 7.18 and the highest PaCO
2 value was 75 mmHg (250 ml/min O
2 consumption) at 50% SpO
2.
Another in vitro study conducted by Struys et al. [
3] compared the time course of inhaled anesthetic drug delivery between two types of anesthesia machine circuits using test lungs. Different combinations of settings (e.g., flow) were applied to observe various patterns of anesthetic delivery and end-tidal concentrations. Such studies can also be performed using an HPS and would even be more accurate than a test lung due to its more advanced real-time gas-sensing mechanisms once synchronized to the modeled mode. A substantial amount of anesthesia-related simulation research has been conducted in the field of airway management, with various studies involving airway techniques and devices [
2]. Although such studies are criticized for seldom following up with patient-based studies to validate the simulation data, apnea oxygenation scenarios can only be ethically conducted to a certain level of acceptable desaturation in actual patients. Such experimental simulation-based studies should offer insights into the mechanisms and a preliminary understanding of new interventions.
Patient studies involving apnea oxygenation have been performed primarily in the operative, emergency, and critical care settings [
7,
8,
10]. Outcome parameters range from time to desaturation, incidence of desaturation, lowest mean saturation reached during intubation, and mean saturation in respiratory failure, among others [
7,
9–
11,
13,
14,
17]. A study on apneic oxygenation comparing trans nasal humidified O
2 therapy versus conventional nasal oxygenation conducted by Rajan et al. [
16] found a longer time to 90% desaturation in the transnasal humidified group (796 s vs. 444 s). Similar time to desaturation (to 92%) was observed in the present simulation study at 250 ml/min O
2 consumption (915 s for PO, 1087 s for PAO) using a nasal cannula at 15 L/min. Other studies based on lower apnea oxygenation flows of 5–10 L/min had times to desaturation (92–95%) ranging from 408 s to 587 s [
19–
21]. Assuming that the O
2 consumption/pulmonary shunt fraction will be slightly more than ideal in real patients and considering the differences in flow rates used, the experimental HPS data correlated well with patient data from previous human apnea oxygenation studies.
In the present study, we compared different nasal oxygenation strategies on an HPS mannequin that varied from no oxygenation to apnea oxygenation alone to para-oxygenation, to assess the time to 92%, 75%, and 50% SpO2. Across all settings, no oxygenation and oxygenation only during apnea showed similar times to desaturation, and para-oxygenation produced marginally longer times to desaturation compared to preoxygenation alone, both of which were significantly longer than those in the earlier two settings. The above findings highlight that, without preoxygenation, apnea oxygenation alone does not provide any additional benefit, and that para-oxygenation can be used to maximize the duration of safe apnea.
The four experimental settings were also applied at O
2 consumption rates of 250, 500, and 750 ml/min to evaluate the performance of the HPS at different O
2 settings, which can be further extrapolated to varied O
2 demand situations. The comparative findings were similar across all consumption rates. In the current study, only the shunt fraction and O
2 consumption rate were manipulated; however, complex pulmonary pathologies may be simulated by adjusting various physiological parameters to estimate how an actual patient might behave under various conditions, such as anesthesia and surgery [
4]. For example, chest wall compliance and bronchial resistance factors can be used to simulate restrictive and obstructive lung pathologies, respectively. Different flow rates of nasal oxygenation could also be compared on the HPS, similar to the protocol proposed by Theiler et al. [
22].
The major and obvious limitation of the study is that the results obtained with any simulator experiment cannot with absolute certainty be compared to human data. Only its proximity to reality, as much as possible, can be ascertained, as was done in the present study. Another limitation is that the full spectrum of the software modifiable physiological factors could not be explored in the present study. This can be planned in future projects.
In conclusion, apnea trends in the HPS in the current study correlated well with similar prior human experiments, providing support for the use of HPS in similar future research and extrapolation of data to human patients. Through simulation experiments, we deduced that AO alone without preoxygenation provides no additional benefit to NO. High-flow O2 via nasal cannula prolonged the time to desaturation in the PAO scenario more than in the PO scenario. Complex, rare, and potentially dangerous scenarios, such as apnea oxygenation, can be easily performed using HPS to study the patterns of various new interventions without the ethical concerns of exposing real patients.