|
 |
| Korean J Anesthesiol > Volume 73(5); 2020 > Article |
|
Abstract
Background
Regional nerve blocks are an integral part of multimodal analgesia and should be chosen based on their efficacy, convenience, and minimal side effects. Here, we compare the use of pectoral (PEC II) and serratus-intercostal fascial plane (SIFP) blocks in breast carcinoma cases undergoing modified radical mastectomy in terms of the postoperative analgesic efficacy and shoulder mobility.
Methods
The primary outcome of this prospective controlled study was to compare the postoperative static and dynamic pain scores, and the secondary outcome was to assess the shoulder pain, range of shoulder joint motion, and hemodynamic parameters. Sixty patients were randomly allocated to three groups and given general anesthesia. All patients received paracetamol, diclofenac, and rescue doses of tramadol based on the institute's acute pain service policy. No block was performed in group C (control), whereas groups P and S received PEC II and SIFP blocks, respectively, before surgical incision.
Results
The groups were comparable in terms of age, weight, height, and body mass index distribution. Dynamic pain relief was significantly better 12 and 24 h postoperatively in groups P (P = 0.034, P = 0.040 respectively) and S (P = 0.012 and P = 0.017, respectively) compared to group C. Shoulder pain relief and shoulder mobility were better in group S, while the hemodynamic parameters were more stable in group P.
The focus of modern medical technology is to provide quality health care services to patients. Anesthesia services have also extended to the entire perioperative period to ensure better postoperative recovery. Most patients undergoing breast and axillary surgery complain of acute postoperative pain in the chest, arm, shoulder, and axilla. If untreated, this leads to chronic pain and restriction of shoulder movements, thus decreasing the quality of life of breast cancer survivors [1]. The incidence of severe pain in the immediate postoperative period is 60% in patients that undergo mastectomy with reconstruction. However, chronic pain develops most often in cases that undergo axillary dissection of lymph nodes with mastectomy known as modified radical mastectomy (MRM) [2,3]. Preventive analgesia through multimodal approaches ensures control of continuous neuronal firing, which eventually decreases the incidence of chronic pain, morbidity, and mortality [4]. This includes giving regional analgesia along with parenteral analgesics during the early postoperative period and gradually stepping down to oral medications [5]. It is important to understand the area of pain distribution before we plan a regional nerve block for MRM. The skin overlying the chest and lateral thorax is supplied by anterior and lateral divisions of the intercostal nerves. The axilla is supplied by T1 and T2 dermatome, which includes the intercostobrachial nerve, whereas the pectoral muscles are supplied by lateral and medial pectoral nerves (branches from the lateral and medial cord of the brachial plexus) [6]. While there are various regional analgesic techniques used for MRM, including epidurals, intercostal nerve block, and paravertebral block, the fascial plane blocks are relatively new. Fascial plane blocks are often preferred because they avoid interfering with the epidural space, give a comparatively longer duration of pain relief with a single shot, and can be performed in patients with deranged coagulation without adverse side effects [7]. Pectoral (PEC) I block anesthetizes the lateral and medial pectoral nerves. In contrast, PEC II anesthetizes the medial and lateral pectoral nerves, the anterolateral branch of the intercostal nerve from T2-T8/9, and the nerve to the serratus anterior, also known as the long thoracic nerve. A serratus plane block (SPB), if performed above the serratus anterior muscle between the serratus anterior and latissimus dorsi (LD), anesthetizes the anterolateral branch of the intercostal nerves T2-T8/9, the nerve to the serratus anterior and thoracodorsal nerve. A SPB below the serratus anterior muscle, also known as a serratus-intercostal fascial plane (SIFP) block, anesthetizes the lateral and anterior cutaneous branches of the intercostal nerves T2-T8/9 [8ŌĆō10]. Information on analgesic duration and efficacy, postoperative dynamic mobilization, ease of performance, and side effects are the main concerns when deciding which analgesic block technique to use for MRM. Hence, this study aimed to compare PEC II and SIFP blocks in cases undergoing MRM for postoperative analgesic efficacy and shoulder mobility.
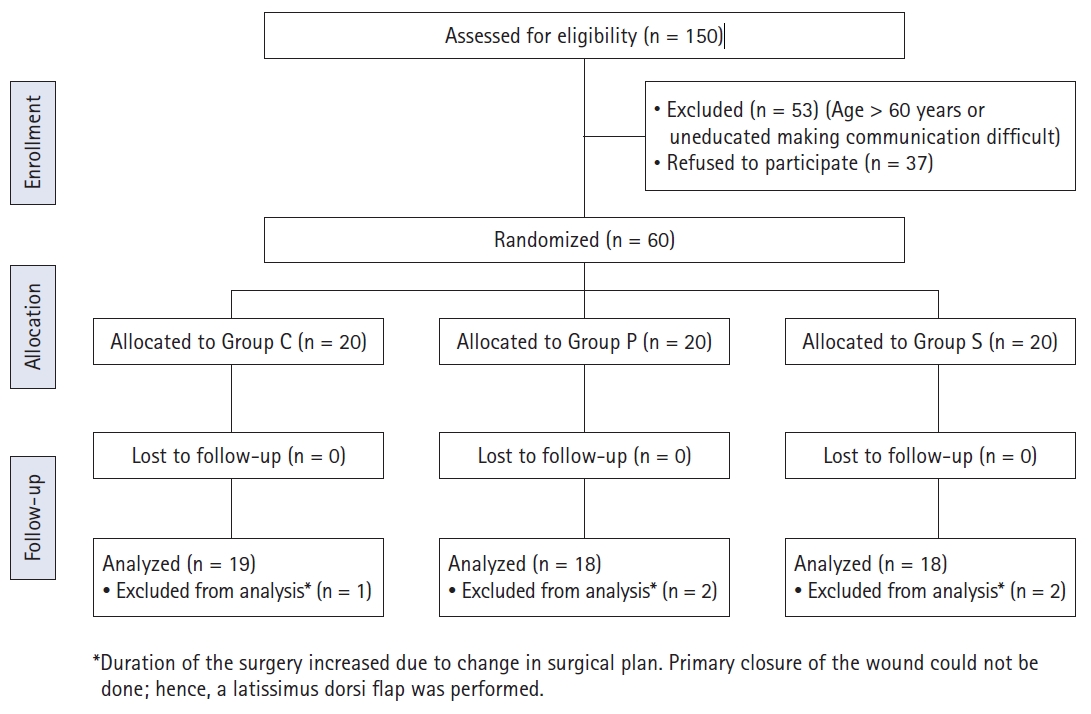
This was a prospective randomized, controlled, parallel-group, interventional trial, following the criterion of the Consolidated Standards of Reporting Trials. Bias was rigorously eliminated using the double-blinding technique. After approval from the institute ethics committee (IEC no: 2016-10-IP-89), the study was performed using patients that underwent MRM surgery between December 2016 and December 2018. The study is enrolled in the clinical trial registry (CTRI/2017/10/009965) and, the clinical research was done following the ethical principles for medical research involving human subjects in accordance with the Helsinki Declaration 2013. The primary objective of the study was to compare and assess the static and dynamic pain scores among the various groups. The secondary objectives were to compare shoulder pain, range of motion, postoperative nausea and vomiting (PONV), and hemodynamic alterations before and after surgical incision among the groups.
Cases were enrolled after they had consented for the procedure but were blinded to the group allocation. Randomization of cases was done when they were planned for the surgical procedure using the chit method. This was done by a surgical resident who was part of our project using 60 chits in a box. In the operation theater, the performer was informed about the group allocation, and the block was performed accordingly. Follow-up of the cases in the postoperative period was done by a resident who was not the performer and hence was blinded to the group allocation.
Patients who underwent MRM and were aged between 18 and 60 years, female, American Society of Anestheiologists grade I/II, with body mass index (BMI) < 40 kg/m2 were included. Pregnant patients, patients allergic to local anesthetics, planned for an additional simultaneous breast reconstruction using either autologous tissue or prosthesis, duration of surgery exceeding 3 h, patients who could not understand the clinical research and the questionnaire of study were excluded.
After a comprehensive pre-anesthetic evaluation was performed and informed consent was sought, the patients were advised about the numerical rating scale (NRS) with 0 as "no pain," 1ŌĆō3 as "mild pain," 4ŌĆō6 as "moderate pain," and 7ŌĆō10 as "severe pain." Patients were kept nil per mouth for 6 h, given lorazepam 0.5 mg the night before and ranitidine 150 mg early on the morning of surgery.
In the operation theater, intravascular access was obtained on the forearm contralateral to the operative breast. Standard monitoring with an electrocardiogram, non-invasive blood pressure, oxygen saturation levels was applied. All of the cases were induced using 0.01 mg/kg midazolam, 2 ╬╝g/kg fentanyl, 1ŌĆō2 mg/kg propofol and 0.1 mg/kg vecuronium. The cases were maintained using inhalational agent sevoflurane, oxygen, and air. After induction, the block was performed under full aseptic precautions before surgical incision, as it was presumed that there could be disruption of the fascial planes post-surgery. The arm on the same side as the operation was positioned at 90┬░ abduction, and the area of the block was painted using betadine with alcohol. The ultrasonography (USG) machine was prepared using a linear transducer (6ŌĆō13 Hz), and the probe was covered with a sterile sheet. Intraoperatively, all patients received paracetamol 1 gm intravenous (i.v) before the start of the surgical incision and an injection of fentanyl 1 ╬╝g/kg/h till completion of surgery. As a part of the institute's acute pain services protocol, all cases also received paracetamol 1 gm i.v every 6 h and diclofenac 75 mg i.v every 8 h from the intra-operative period till two days after surgery. Injection tramadol 1 mg/kg (maximum to three times a day) was used for rescue analgesia.
The patients were divided into three groups:
Group C (control): received no block.
Group P (PEC II block): Patients were given a PEC block (PEC II) single shot using a USG machine with local anesthetic 0.2% ropivacaine 30 ml (10 ml between the pectoralis major and minor +20 ml between the pectoralis minor and serratus anterior).
Group S (SIFP): Patients were given a block with 0.2% ropivacaine 0.4 ml/kg single shot.
The surgical incision was started 10 min after the block to give adequate time for the block to act. An i.v injection of ondansetron 4 mg and dexamethasone 8 mg was given to all patients to address PONV. Cases were extubated after surgery and were assessed immediately postoperatively, as well as 12, 24, and 48 h for static and dynamic pain, shoulder pain, rescue analgesic requirements, or any side effects. Complete shoulder abduction was not allowed prior to 6 h post-surgery because of wound and drain issues.
For the PEC II block: The USG probe was initially kept longitudinally over the clavicle in the midclavicular line, behind which is the first rib. Moving the probe downwards in the third intercostal space showed the thoracoacromial artery. Here, between the pectoralis major and pectoralis minor, we deposited 10 ml of 0.2% ropivacaine (PEC I block). This block anesthetizes the medial and lateral pectoral nerves. Then, the probe was rotated toward the axilla by 45┬░. Moving the probe to the anterior axillary line, the attachment of the serratus anterior muscle just above the rib could be seen. Here, in the third intercostal space, we deposited 20 ml of 0.2% ropivacaine between the pectoralis minor and serratus anterior muscle.
SIFP block (SPB below the serratus anterior muscle): The USG probe was moved beyond the PEC II position to the fourth intercostal space, midaxillary line, and again rotated into a longitudinal position to visualize the thick belly of serratus anterior and intercostal muscles below it. The needle was inserted in-plane under real-time visualization to hit the fourth/fifth rib, and 0.4 ml/kg of 0.2% ropivacaine was deposited.
The normality of continuous variables was assessed using the Shapiro-Wilk test. Normally distributed continuous variables are presented as the mean ┬▒ SD, whereas ordinal variables (NRS score) are presented as the mean ┬▒ SD (median). Means were also used to describe the ordinal data along with median. A one-way analysis of variance was used to compare the means among the three independent groups. The Kruskal Wallis test was used followed by multiple comparisons (Bonferroni test) to compare the distribution of the NRS pain scores among the three study groups. A paired sample t-test was used to test the change in means between the pre to post observations. Fisher's exact test was used to compare the proportions between the groups. A two-sided P value of < 0.05 was considered statistically significant. Statistical Package for Social Sciences, version 23 (SPSS-23, IBM, USA) was used for data analysis.
To compare the detected means of the differences (pre-post observations, i.e., between the immediate postoperative and 24 h time points) of the NRS pain score in the three study groups, with a minimum two-sided 95% CI and 90% power, with an assumed effect size between the treatment and control of 0.5, the estimated sample size for each of the three study groups came out to be 18 (total 54). Similarly, with a minimum two-sided 95% CI and 90% power, we required at least 18 individuals to detect the effect size of 0.82 between paired observations. The sample size was estimated using G Power, version 3.1.9.2 (D├╝sseldorf University, Germany).
Out of the 150 patients evaluated for participation in this study, 60 eligible patients were enrolled and randomized into three groups, C, S, and P, with 20 in each group (Fig. 1). One patient in group C, as well as two each in groups S and P, were excluded during the process of data collection because their surgical plan was changed intraoperatively (primary closure of the wound was not achievable; hence a LD flap was done for wound cover). Therefore, data were analyzed for 19 patients in the control group C and 18 patients in each of the intervention groups S and P.
The mean age, weight, height, and BMI of the patients were comparable among the three groups (P = 0.382, 0.921, 0.411, and 0.861, respectively; Table 1).
The NRS scores for static and dynamic pain were both significantly less 12 and 24 h postoperatively in groups P and S, as compared to group C. However, they were comparable with each other (Table 2).
The patients in group S complained of significantly less shoulder pain 12 and 24 h after the procedure as compared to groups C and P (Table 2).
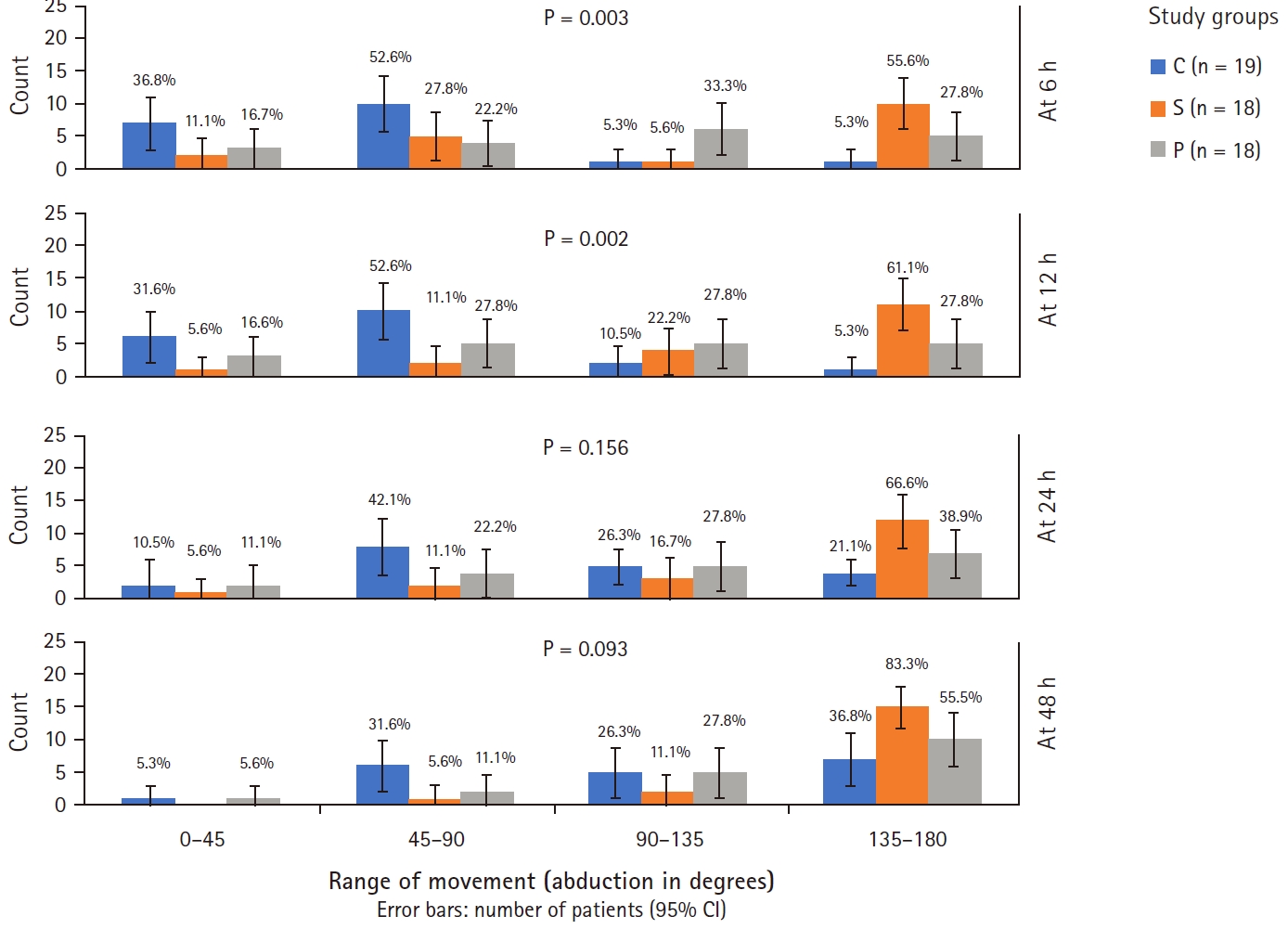
The first evaluation of complete shoulder abduction was assessed for each patient after 6 h due to wound and drain issues, followed by 12, 24, and 48 h (Fig. 2). The patients were compared between the study groups in terms of their range of abduction. The results showed that the study groups were significantly associated with a range of movements at 6 h (P = 0.003) and 12 h (P = 0.002). In group C, the highest number of patients belonged to the 45ŌĆō90┬░ range of abduction at all time points. In groups S and P, the highest number of patients belonged to the 135ŌĆō180┬░ range of abduction at all time points. The proportion of patients that achieved shoulder abduction between 135ŌĆō180┬░ was significantly higher in group S at 6 h (P = 0.027) and 12 h (P = 0.018) postoperatively as compared to group C. However, the range of shoulder abduction was statistically the same among the three groups at all other time points.
The hemodynamic response to incision, as elicited by a rise in heart rate and blood pressure 1 min after incision as compared to 1 min before incision, was insignificant in group P, while significant in the other two groups (Table 3).
Rescue analgesia (an injection of tramadol) was required in three patients in group C and two in group S. Three patients of group C and two of group S reported postoperative nausea without vomiting. Out of the former three, two received tramadol as rescue analgesia. No other side effects, such as sedation or hypotension, were observed in any patient.
Our results demonstrate that patients given PEC II/SPB below the muscle (SIFP) had a significant and comparable decrease in static and dynamic pain in the postoperative period (intergroup P > 0.05 between groups P and S). However, pectoral pain was controlled better in group P (PEC II), and shoulder pain was significantly less in group S (intergroup P < 0.05 at 12 h). Static pain is described as pain at rest, whereas dynamic pain is defined as pain on deep breathing, coughing or movement [11].
The analgesic effects of SIFP or PEC blocks have been studied previously [9,10,12ŌĆō15]. No study has directly compared the efficacy of these two blocks in terms of pectoral pain, shoulder or axillary pain, and shoulder mobility after MRM, which is important to promote early physiotherapy to avoid long-term complications like lymphedema, axillary web syndrome (AWS), or shoulder immobility [16]. There was a study protocol that proposed to include shoulder mobility as a parameter in their study, including chest blocks, but to date, no data has been made available [17].
The static and dynamic NRS in both groups P and S were significantly less (P < 0.05) at 12 and 24 h compared to group C. The duration of the SPB below the serratus anterior muscle as proposed by Blanco et al. was approximately 360 min. The longer duration seen in our study can be explained by the use of dexamethasone, which was given to relieve PONV [18,19].
The evaluation of shoulder pain included pain in and around the shoulder joint, including the muscles, joint, axilla (which forms the inferior part of the shoulder joint), anterior, and posterior axillary fold. During the early postoperative period, shoulder pain is caused by surgical positioning, muscular spasms, and axillary retraction or manipulation. Our study emphasizes better coverage of shoulder pain in the SIFP group. A cadaveric study by Daga et al. [20] showed the cranial spread of dye up to T2/3 when the drugs were deposited in the plane between the serratus anterior and intercostal muscles at the fourth/fifth intercostal space. Further studies on this are required because out of the total 30 ml of saline that was used for the plane separation, the dye was mixed only in the last 10 ml. The fascia between the serratus anterior and intercostal muscles is continuous from T1-T8, so it is unclear what stops the drug from reaching the T1 dermatome, which, along with T2, covers the axilla, anterior and posterior axillary fold, and upper third of the arm. Blanco also found better axillary analgesia after SPB below the serratus anterior muscle because the drug is deposited directly in the midaxillary line over the exit of the lateral cutaneous nerve, compared to the PEC II group where the drug gradually seeps and reaches the target. Hence, less pain around the shoulder in group S could be an indirect representation of better coverage of T1 and T2 dermatomes. This is important because it would ensure pain-free shoulder mobility leading to early and effective physiotherapy, which is a major concern in cases undergoing axillary lymph node clearance to limit joint mobility due to lymphedema, rotator cuff tendinitis, or disuse muscular contracture [21].
Hemodynamic parameters 1 min after the incision were better controlled in group P (PEC II). This could be explained by the direct and faster coverage of the anterior cutaneous branches of the intercostal nerves by the PEC II block as compared to the SIFP block. Although, Fajardo et al. [12], in his study on SIFP, reported controlled hemodynamic parameters on the surgical incision that did not require rescue opioid dose despite less time between the block and surgery. In the literature, dexamethasone has been shown to delay the onset of block action [18]. Therefore, if we had waited longer before the surgical incision, we potentially could have controlled the hemodynamic parameters in group S (SIFP) as well. Postoperatively, both groups had comparable pain relief. This could be explained by the gradual spread of drugs to the anterior hemithorax in group S [8,13]. This is due to the anatomical continuity of the serratus-intercostal plane up to the mid-clavicular line anteriorly and the seeping of drugs from the external intercostals to the more intimate layers.
We checked shoulder mobility by asking the patient to abduct their arm once at 6, 12, 24, and 48 h postoperatively. Maximum abduction was observed in patients of group S followed by group P and later by group C. However, this was statistically significant only at 6 and 12 h (P = 0.003 and P = 0.002, respectively). This could be correlated to less axillary and shoulder pain in patients that received the SIFP block. Better shoulder mobility in patients that received the PEC II block compared to controls has also been found by Khemka et al. [22]. AWS is a condition that develops two weeks after axillary dissection in about 48.3% of patients, the main etiology of which is the discontinuity of the lympho-venous channels and myofascial trigger points [23]. To avoid the incidence of such trigger zones, it is necessary to maintain good arm mobilization postoperatively. It is debated as to whether physiotherapy should be started early or late; however, pain-free arm movements help in the performance of exercises.
SIFP and PEC II blocks have their own set of advantages and disadvantages. For example, while the duration of analgesia is comparable between the procedures, SIFP is easier to perform due to the endpoint of the needle and the drugs being deposited in one place. In contrast, PEC II blocks require precise placement of the needle tip first between the pectoralis major and minor and then between the pectoralis minor and serratus anterior. Thoracic nerves are selectively more targeted in the SIFP block, and vascular injury is more common while performing the PEC II block due to its proximity to the thoracoacromial artery [8]. The USG machine will always be a limiting factor in performing the PEC II block, unlike the SIFP, which can be performed blindly [14]. This can be achieved through surface markings using the midaxillary line, fourth/fifth intercostal space, and depositing the drug above the rib. We chose to use the midaxillary line rather than the posterior axillary line because the serratus anterior muscle belly is the thickest there, and it would be challenging for the drug to reach the anterior cutaneous nerve of the intercostal muscle from the posterior axillary line [20]. Also, the lateral branch of the intercostal pierces and branches into the anterior and posterior division in the midaxillary line to lie between the serratus and external intercostal muscles, hence ensuring its good coverage at this point.
The benefits of blocking the nerve to the serratus anterior and thoracodorsal nerves, which are primarily motor nerves, using SPB above the serratus anterior/PEC II block, are unclear. Anesthetizing the nerve to the LD will be beneficial if reconstruction is done using LD flap, but otherwise, in a regular MRM with primary closure, it does not appear to be overly beneficial. This could be due to relief of the muscular spasms induced by surgical manipulation, which might decrease axillary pain postoperatively. The concerns regarding motor nerve paresis have been raised previously with SPB above the serratus anterior muscle. This block was abandoned at some centres as it was found to disrupt the axillary fascia and hinder the surgical performance of the surgeons, who faced difficulty in identifying the long thoracic and thoracodorsal nerves by nerve stimulation as they were anesthetized [13]. As such, there were concerns raised regarding the potential to damage these nerves.
There are several limitations to our study. Only a small number of cases were included in the current study, and the hemodynamic monitoring was only conducted for a brief period. Furthermore, the patients were not followed up in the long-term to assess the incidence of surgical or chronic pain and the range of shoulder movement. However, to our knowledge, this is the first study to compare the SIFP and PEC II blocks in terms of the postoperative analgesic efficacy and range of shoulder movement. This study also questions the need to block the nerve to the serratus anterior and thoracodorsal nerve using SPB above the serratus anterior/PEC II block when equal analgesia can be achieved using SIFP (SPB below the serratus anterior) block. Further studies should be conducted to compare the efficacy of PEC I + SPB, PEC II, SPB and modified PEC II to evaluate the incision response, intra- and postoperative pectoral and shoulder pain with a longer follow-up period to see the effect on post-mastectomy syndrome and shoulder mobility. Moreover, studies with additives or a continuous catheter to increase the duration of analgesia could be performed, along with studies that examine the impact of starting physiotherapy earlier or later.
In conclusion, both SIFP and PEC II blocks were found to provide comparable dynamic and static pain relief. Shoulder pain and, in particular, axillary pain was found to be better managed with SIFP. A greater range of shoulder movement was possible after SIFP block compared to PEC II.
Acknowledgments
We would like to thank Dr. Abdul MM Ansari for his assistance with the data analysis.
NOTES
Author Contributions
Ushkiran Kaur (Formal analysis; Writing ŌĆō original draft)
Chetna Shamshery (Conceptualization; Data curation; Investigation; Methodology; Visualization; Writing ŌĆō original draft)
Anil Agarwal (Conceptualization; Project administration; Supervision; Writing ŌĆō review & editing)
Neel Prakash (Writing ŌĆō review & editing)
Ramya Chakrapani Valiveru (Investigation; Methodology; Writing ŌĆō review & editing)
Prabhaker Mishra (Formal analysis; Software; Validation)
Fig.┬Ā2.
Shoulder abduction in different study groups at different post-operative time intervals (6, 12, 24 and 48 h) are presented. The graph depicts the number of patients (in percentage) and its 95% CI. FisherŌĆÖs Exact test has been used to compare the percentage. Result showed that the study groups were significantly associated with range of movements at 6 h (P = 0.003) and 12 h (P = 0.002). In 135-180┬░, there was significant difference in Group S at 6 h (P = 0.027) and 12 h (P = 0.018) post-operatively as compared to Group C.
Table┬Ā1.
Demographic Variables and Duration of Surgery among the Three Groups
Table┬Ā2.
Comparison of the Numerical Rating Scale for Static, Dynamic, and Shoulder Pain among the Three Groups Postoperatively
| Time points | Group C (n = 19) | Group S (n = 18) | Group P (n = 18) | P value |
|---|---|---|---|---|
| Static pain | ||||
| ŌĆāImmediate | 1.4 ┬▒ 1.6 (2.0) | 1.4 ┬▒ 1.6 (2.0) | 1.3 ┬▒ 1.5 (0.5) | 0.851 |
| ŌĆā12 h*,ŌĆĀ | 3.3 ┬▒ 2.9 (2.0) | 1.9 ┬▒ 1.1 (2.0) | 1.7 ┬▒ 1.1 (2.0) | 0.047 |
| ŌĆā24 h*,ŌĆĀ | 2.7 ┬▒ 2.8 (2.0) | 1.0 ┬▒ 0.9 (1.0) | 1.7 ┬▒ 0.9 (1.0) | 0.022 |
| ŌĆā48 h | 1.0 ┬▒ 0.9 (0) | 0.5 ┬▒ 0.7 (0) | 0.8 ┬▒ 1.3 (0) | 0.098 |
| Dynamic pain | ||||
| ŌĆāImmediate | 4.3 ┬▒ 2.3 (4.0) | 3.1 ┬▒ 2.9 (2.8) | 2.9 ┬▒ 2.7 (2.8) | 0.124 |
| ŌĆā12 h*,ŌĆĀ | 4.5 ┬▒ 2.4 (4.5) | 2.9 ┬▒ 1.4 (3.0) | 2.5 ┬▒ 1.4 (3.0) | 0.037 |
| ŌĆā24 h*,ŌĆĀ | 3.8 ┬▒ 2.1 (4.0) | 1.9 ┬▒ 1.2 (2.0) | 1.8 ┬▒ 1.3 (2.0) | 0.018 |
| ŌĆā48 h | 1.4 ┬▒ 1.4 (1.5) | 1.3 ┬▒ 0.9 (1.0) | 1.4 ┬▒ 1.8 (1.0) | 0.877 |
| Shoulder pain | ||||
| ŌĆāImmediate | 1.0 ┬▒ 2.0 (0) | 0.9 ┬▒ 1.3 (0) | 0.9 ┬▒ 1.5 (0) | 0.775 |
| ŌĆā12 h* | 2.9 ┬▒ 2.7 (1) | 1.4 ┬▒ 1.5 (1.0) | 2.5 ┬▒ 2.5 (2.5) | 0.018 |
| ŌĆā24 h*,ŌĆĪ | 2.0 ┬▒ 2.4 [0] | 0.8 ┬▒ 0.7 [1.0] | 2.1 ┬▒ 2.4 [2.0] | 0.028 |
| ŌĆā48 h | 0.2 ┬▒ 0.4 [0] | 0.1 ┬▒ 0.3 [0] | 0.7 ┬▒ 1.5 [0] | 0.154 |
Table┬Ā3.
Change in the Hemodynamic Values between Pre- and Post-incision
| Study groups | Heart rate | Systolic blood pressure | ||||
|---|---|---|---|---|---|---|
| Before incision | After incision | P value* | Before incision | After incision | P value* | |
| Group C (n = 19) | 85.1 ┬▒ 14.2 | 91.3 ┬▒ 15.8 | < 0.001 | 114.3 ┬▒ 14.9 | 119.9 ┬▒ 15.6 | 0.029 |
| Group S (n = 18) | 77.4 ┬▒ 12.6 | 88.1 ┬▒ 17.4 | < 0.001 | 112.0 ┬▒ 20.4 | 125.38 ┬▒ 23.0 | < 0.001 |
| Group P (n = 18) | 77.9 ┬▒ 18.1 | 81.9 ┬▒ 20.7 | 0.060 | 118.9 ┬▒ 18.5 | 118.5 ┬▒ 15.8 | 0.851 |
| ŌĆĀP value | 0.091 | 0.132 | 0.291 | 0.292 | ||
References
1. Mejdahl MK, Andersen KG, G├żrtner R, Kroman N, Kehlet H. Persistent pain and sensory disturbances after treatment for breast cancer: Six year nationwide follow-up study. BMJ 2013; 346: f1865.


2. Fecho K, Miller NR, Merritt SA, Klauber-Demore N, Hultman CS, Blau WS. Acute and persistent postoperative pain after breast surgery. Pain Med 2009; 10: 708-15.



3. Legeby M, Segerdahl M, Sandelin K, Wickman M, Ostman K, Olofsson Ch. Immediate reconstruction in breast cancer surgery requires intensive post-operative pain treatment but the effects of axillary dissection may be more predictive of chronic pain. Breast 2002; 11: 156-62.


4. Huang CC, Sun WZ, Wong CS. Prevention of chronic postsurgical pain: the effect of prevention and multimodal analgesia. Asian J Anesthesiol 2018; 56: 74-82.

5. Rawal N. Acute pain service: Organization, function, & implementation. In: Textbook of Regional Anesthesia and Acute Pain Management. Edited by Hadzic A: New York, McGraw-Hill Medical. 2007, p 1126.
6. Blanco R, Barrington MJ. Pectoralis and serratus plane blocks. In: Hadzic's Textbook of Regional Anesthesia and Acute Pain Management. 2nd ed. Edited by Hadzic A: New York, McGraw-Hill. 2017, p 653.
7. Freise H, Van Aken HK. Risks and benefits of thoracic epidural anaesthesia. Br J Anaesth 2011; 107: 859-68.



8. Blanco R, Parras T, McDonnell JG, Prats-Galino A. Serratus plane block: a novel ultrasound-guided thoracic wall nerve block. Anaesthesia 2013; 68: 1107-13.


9. Selvi O, Tulgar S, Senturk O, Serifsoy TE, Thomas DT, Deveci U, et al. Is a combination of the serratus intercostal plane block and rectus sheath block superior to the bilateral oblique subcostal transversus abdominis plane block in laparoscopic cholecystectomy? Eurasian J Med 2020; 52: 34-7.



10. L├│pez-Matamala B, Fajardo M, Est├®banez-Montiel B, Blancas R, Alfaro P, Chana M. A new thoracic interfascial plane block as anesthesia for difficult weaning due to ribcage pain in critically ill patients. Med Intensiva 2014; 38: 463-5.

11. Kumar R, Nath SS, Agarwal A. Intraperitoneal nebulization versus intraperitoneal instillation of ropivacaine for postoperative pain management following laparoscopic donor nephrectomy. Korean J Anesthesiol 2019; 72: 357-65.




12. Fajardo M, L├│pez S, Di├®guez P, Alfaro P, Garc├Ła FJ. A new ultrasound-guided cutaneous intercostal branches nerves block for analgesia after non-reconstructive breast surgery. Cir Mayor Ambulatoria 2013; 18: 3-6.
13. Abdallah FW, Cil T, MacLean D, Madjdpour C, Escallon J, Semple J, et al. Too deep or not too deep?: a propensity-matched comparison of the analgesic effects of a superficial versus deep serratus fascial plane block for ambulatory breast cancer surgery. Reg Anesth Pain Med 2018; 43: 480-7.


14. Yoon HS, Yu BW, Kim YM, Lee JH, Koh WU, Yang HS. Serratus anterior plane block combined with monitored anesthesia care for surgery of lateral side of breast -a case report. Korean J Anesthesiol 2019; 72: 500-3.




15. Bhoi D, Selvam V, Yadav P, Talawar P. Comparison of two different techniques of serratus anterior plane block: a clinical experience. J Anaesthesiol Clin Pharmacol 2018; 34: 251-3.



16. Stuiver MM, Ten Tusscher MR, Agasi-Idenburg CS, Lucas C, Aaronson NK, Bossuyt PM. Conservative interventions for preventing clinically detectable upper-limb lymphoedema in patients who are at risk of developing lymphoedema after breast cancer therapy. Cochrane Database Syst Rev 2015; (2): CD009765.

17. Langford R, Brown I, Vickery J, Mitchell K, Pritchard C, Creanor S. Study protocol for a double blind, randomised, placebo-controlled trial of continuous subpectoral local anaesthetic infusion for pain and shoulder function following mastectomy: SUB-pectoral Local anaesthetic Infusion following MastEctomy (SUBLIME) study. BMJ Open 2014; 4: e006318.



18. Mathew R, Radha KR, Hema VR. Effect of perineural and intravenous dexamethasone on duration of analgesia in supraclavicular brachial plexus block with bupivacaine: a comparative study. Anesth Essays Res 2019; 13: 280-3.



19. Knezevic NN, Anantamongkol U, Candido KD. Perineural dexamethasone added to local anesthesia for brachial plexus block improves pain but delays block onset and motor blockade recovery. Pain Physician 2015; 18: 1-14.

20. Daga V, Narayanan MK, Dedhia JD, Gaur P, Crick H, Gaur A. Cadaveric feasibility study on the use of ultrasound contrast to assess spread of injectate in the serratus anterior muscle plane. Saudi J Anaesth 2016; 10: 198-201.



21. Scaffidi M, Vulpiani MC, Vetrano M, Conforti F, Marchetti MR, Bonifacino A, et al. Early rehabilitation reduces the onset of complications in the upper limb following breast cancer surgery. Eur J Phys Rehabil Med 2012; 48: 601-11.