COVID-19 pandemic: ethical and legal aspects of inadequate quantity and quality of personal protective equipment for resuscitation
Article information
The coronavirus disease 2019 (COVID-19) pandemic has implications for cardiopulmonary resuscitation (CPR). Chest compression is an aerosol generating procedure that is associated with a high risk of disease transmission to healthcare workers (HCWs) [1]. Before commencing CPR, guidelines recommend usage of a minimum respiratory personal protective equipment (PPE), “an FFP3 mask (FFP2 or N95 if FFP3 is not available)” [1].
Adherence to guidelines may have been challenged in two scenarios. First, there was an “inadequate quantity” (supply) of N95 masks during this pandemic. This rendered the HCWs unable to physically adhere to the guidelines. Second, masks may have been or may have been perceived to be of “inadequate quality” for several reasons [1]. The N95 mask has a lower filtration performance than the first-choice FFP3 mask, i.e., 95% vs. 99% [1], although the clinical significance of this is unknown. Additionally, counterfeit and poor quality N95 masks are being sold. Since chest compression involves vigorous movements, it may lead to poor mask seal, decreased protection rates, and mask failure including strap slipping [2]. Finally, N95 masks undergoing extended use and reuse are associated with disease transmission and decreased functionality [3].
Ethical dilemmas and confusions arise in both scenarios. In the “inadequate quality” scenario, the HCW is expected to perform a duty (CPR) adhering to the guidelines (wearing a N95 mask). Hence, it is uncertain whether the HCW should proceed with an actual or perceived “inadequate quality” PPE. Moreover, existing PPE guidelines are not specific to CPR, and some guidelines recommend powered air-purifying respirators (PAPRs) during CPR in patients with COVID-19 [4]. As the risk of disease transmission during CPR is uncertain, it is unclear whether we should aim for the “maximum” level protection, or be satisfied with “adequate.” However, refusal to treat may result in disciplinary or legal action against the HCW.
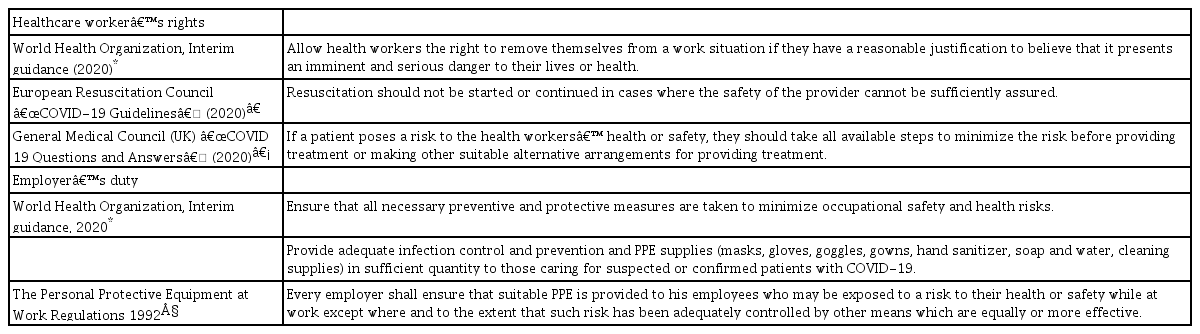
Various statements and considerations pertaining to a doctor’s duty to treat (or not) are shown in Table 1. The duty to treat is guided by the ethical principles of beneficence, non-maleficence, autonomy, and justice [5,6]. As these principles apply to the patient, HCWs and society, there may be conflicting priorities. HCWs have an obligation to prevent self-infection and onward transmission of infections to other patients, their colleagues and relatives, and the wider community [1]. Further, the ethical principle of justice takes into account the right of not being killed by another human being [6] (with a serious infectious disease). Justice also requires hospitals to provide adequate PPE.
Moreover, there are the doctrines of expressed consent, implied consent, special training, reciprocity, and professional oaths and codes [5]. Expressed consent includes signing of a contract on the basis that adequate PPE would be provided [5]. Arguments against implied consent include those that the duty does not specifically involve potentially deadly infectious diseases, and that the HCW was not fully informed of the risks [5] (related to a new or evolving disease, e.g., COVID-19). Special training does not entail that the HCW should incur more than minimal risk [5]. Reciprocity implies that doctors may have benefited from their profession, however, this does not necessitate risking one’s life to aid another [5]. In return, employers must provide support, a safe working environment, adequate PPE, and information on acceptable risks to HCWs. Further, oaths are more symbolic and not all HCWs share them [5]. A medical code may not explicitly state the risk of harm, and its scope is open to interpretation.
If HCWs refuse to perform CPR due to “inadequate quantity and quality” of PPE, available options for self-protection include PAPRs. Since the safety of staff is paramount, it should be considered even if it may cause a brief delay in commencing CPR [1]. As alternatives are not always available, an early, proactive, system-level approach should be adopted. This avoids the “inadequate quantity and quality”-related scenarios, decreases the risk to HCWs, and minimizes ethical dilemmas. Measures should include assessing the adequacy of PPE, recording and escalating safety concerns, and requesting the organization to take action [7].
Legal requirements and guidance regarding PPE provision are shown in Table 1. The legal duty to ensure that “safety is reasonably practicable” [8] is open to interpretation. If employers breach their duty of providing adequate PPE (in appropriate quantity and quality), they may be liable to prosecution depending on local regulations [7]. Furthermore, negative publicity and legal risk may ensue [9]. On the other hand, disadvantages of non-disclosure include the inability to conduct investigations and provide treatment (e.g., counselling and isolation), risk of further transmission, and potential harm (e.g., COVID-19) [9]. To balance this, “disclosure should be the norm, even when the probability of harm is extremely low” [9].
“Inadequate” PPE causes many ethical dilemmas for the HCWs. Global supplies of quality-controlled PPE must be robust to cope with the surging demands of the pandemic. Further studies are needed to confirm the risk of disease transmission and the clinical significance of the various levels of PPE protection. If protection is inadequate, corresponding guidelines must evolve to better protect HCWs.
Notes
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Author Contributions
Patrick Wong (Conceptualization; Data curation; Supervision; Validation; Writing – original draft; Writing – review & editing)
Wan Yen Lim (Data curation; Writing – original draft; Writing – review & editing)
Huei Leng Chee (Writing – review & editing)
Rehana Iqbal (Validation; Writing – review & editing)