|
 |
|
|
Abstract
Although anaphylactic shock during the perioperative period is rare, it can be lethal due to severe cardiovascular and respiratory collapse. Midazolam is generally used as premedication for relieving anxiety about the operation, and the danger of anaphylactic shock after intramuscular injection is not widely recognized. We report the first case of anaphylactic shock occurring during the perioperative period after intramuscular injection of midazolam. Since anaphylactic shock after intramuscular injection can be of slow onset, the operation should be delayed if an anaphylactic reaction is suspected, even if the symptoms are limited. In addition, anesthesiologists should be prepared for the occurrence of anaphylaxis at any time in the perioperative period.
Anaphylaxis is a systemic hypersensitivity reaction that develops when patients are exposed to a specific antigen to which they have been previously sensitized [1]. In spite of the rarity of anaphylaxis during the perioperative period, with rates of 1 : 2,800 to 1 : 20,000, it can be fatal and contributes up to 3% of anesthesia-related deaths caused by severe cardiovascular and respiratory collapse [2,3,4,5].
Although midazolam is widely used as premedication for relieving anxiety during operations, rare hypersensitivity reactions have only been reported when it was injected intravenously [6,7,8]. We present the first case of anaphylactic shock caused by midazolam administered intramuscularly as premedication.
A 59-year-old male, height 170 cm and weight 68 kg, was admitted for open reduction and internal fixation of ankle fracture. The patient underwent an open reduction and internal fixation of ankle fracture under spinal anesthesia with bupivacaine 2 years ago. During the surgery, he was sedated by intravenously administered midazolam without premedication and the anesthesia- or surgery-related complications were not reported. He has not experienced sedation management with the exception of the surgery. There was no known history of atopy, asthma, or allergies. Preoperative evaluations such as laboratory tests, electrocardiography, and chest radiography were normal.
For the preoperative treatment, atropine 0.5 mg and midazolam 3.0 mg were intramuscularly administered 30 minutes before the operation. His blood pressure was 122/75 mmHg with a heart rate of 85 beats/min, and a respiratory rate of 16 breaths/min before he transferred to the operation room. After his arrival in the operating room, the patient's vital signs were monitored by noninvasive blood pressure measurement, electrocardiography, and pulse oxymetry. Before induction of general anesthesia, he complained generalized pruritus. Urticarial skin rashes of upper and lower extremity and an angioedema of face and tongue were observed.
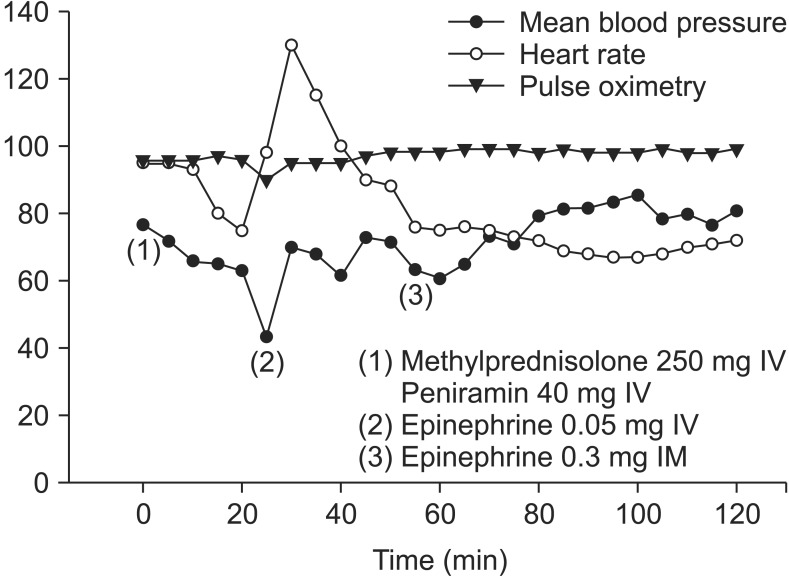
Because an anaphylactic reaction was suspected judging from these generalized skin symptoms, the operation was delayed and he was moved to the recovery room. Although 250 mg of methylprednisolone and 40 mg peniramin were injected intravenously to treat the anaphylactic reaction, his blood pressure decreased to 70/40 mmHg with heart rate of 98 beats/min and pulse oxygen saturation on pulse oximetry (SpO2) of 90% 25 minutes after skin symptoms (Fig. 1). Although he began to experience mild dyspnea, the sound of wheezing was not auscultated. The patient was given 100% oxygen via face mask. Simultaneously, epinephrine 50 ┬Ąg and hydrocortisone 100 mg were injected intravenously with plasmalyte solution (Plasma Solution-A, CJ Pharma, Seoul, Korea) hydration. And then, the serum tryptase level and the total serum immunoglobulin E (IgE) level were measured for confirming anaphylaxis. After transient tachycardia of heart rate of 130 beats/min, heart rate decreased to 100 beats/min within 10 minutes and the blood pressure increased to 90/60 mmHg, SpO2 to 95%. Except the tachycardia, other specific changes of electrocardiography were not observed. During the rest of the period, vital status was stably maintained without medication other than intramuscular epinephrine 0.3 mg. The elevated tryptase level to 29.9 ┬Ąg/L (normal range: 0ŌĆō11 ┬Ąg/L) was detected from laboratory test. Except that systemic erythematous rash and swelling of the skin around the eyes could still be seen, there were no other abnormal findings. The skin symptoms resolved after 2 hours in the recovery room. Arterial blood gas analysis (ABGA) gave pH 7.32, pCO2 44 mmHg, pO2 155 mmHg, and BE 0.5 mM at fraction of inspired oxygen 0.3. Since the results of ABGA and chest radiography were normal and there were no further specific clinical presentations, the patient was transferred to a general ward.
After 2 months, skin prick and intradermal tests were performed to establish the allergen. As a result, midazolam was identified as a cause of anaphylactic shock (Table 1) and the patient was informed to avoid this agent. The patient underwent an open reduction and internal fixation of ankle fracture under spinal anesthesia without sedation. After one week following surgery, the patient was discharged without any other problems.
Because shocks that develop during the perioperative period affect patients' safety, rapid identification of their cause and management are vital. Judging by the absence of abnormalities during the preoperative evaluations and the fact that no procedure except premedication was performed, the possibility of cardiogenic, hypovolemic or neurogenic shock was minimal in the present case. However the appearance of specific skin lesions in the form of urticarial skin rashes and angioedema made us suspect anaphylaxis.
The diagnosis of anaphylaxis during perioperative period can be quite difficult. The symptoms cardiovascular collapse of anaphylaxis can be mistaken as effects of the anesthetic agents and mechanical ventilation after application of a muscle relaxant may mask respiratory symptoms, which can also make it difficult to diagnose. Furthermore, even if anaphylaxis is identified, it is difficult to establish the cause because the variety of agents is exposed to the patients during the brief period of surgery.
The most important clues for diagnosing anaphylaxis are clinical presentations such as skin and mucosal involvement, respiratory symptoms, and cardiovascular dysfunction [5,9]. Information about antigen exposure can provide an indication of the cause of the anaphylaxis. Laboratory tests that detect an increase of serum tryptase are the most commonly used diagnostic methods for confirming anaphylaxis. Serum tryptase, a neutral serine protease released by mast cells, increases within 1ŌĆō2 hours after presentation of clinical symptoms of anaphylaxis, and its level returns to normal after about 24 hours [5,10]. Therefore, anesthesiologists should be aware that detecting elevated serum tryptase is an important indicator confirming the diagnosis when skin reactions are not obvious [11]. Lastly, the gold standard for identifying the cause of immunoglobulin-E mediated anaphylaxis are skin tests such as the skin prick test and intradermal test [9,12,13]. In order to avoid false negative results caused by depletion of specific immunoglobulin-E antibodies and mast cells, the skin test should be performed 4ŌĆō6 weeks after anaphylaxis [5,9].
Of the anaphylaxis occurring during the perioperative period, 55ŌĆō59% are caused by muscle relaxants, while latex (20ŌĆō22%) and antibiotics (13ŌĆō15%) are other major causes [2,10,12]. Reports of midazolam-associated hypersensitivity reactions have been rare and all such cases occurred after intravenous injection [6,7,8]. In this case, atropine and midazolam were administered intramuscularly for premedication. Fortunately, rapid detection of skin lesions deterred us from using other drugs, to induce anesthesia. Therefore, detecting the cause of the anaphylaxis was not difficult; the candidates were the two drugs injected, and the skin test pointed to midazolam. Midazolam is widely used intramuscularly for relieving anxiety before surgery, and anaphylactic shock presenting as severe cardiovascular collapse can occur when it is injected intramuscularly. As this present case has shown, anaphylactic shock can occur about 1 hour after intramuscular injection despite treatment. Therefore, if the skin lesions of anaphylaxis are observed, surgery must be postponed until the patient's safety is secured.
Cross-reactivity between benzodiazepines is a part that should not be overlooked. Cross-reactivity between neuromuscular blocking agents and antibiotics is known to be common due to structural similarity in these drugs [4,5,14]. In case of neuromuscular blocking agents, cross-reactivity occurs in up to 60% of the patients [4,5]. However, allergic reactions to benzodiazepines have been reported for individual agents and such reactions are rare. Except one study of anaphylaxis caused by tetrazepam which showed there was no cross-reactivity among benzodiazepine [15], cross-reactivity between drugs in this class is not as well known. Therefore, further studies regarding the cross-reactivity between benzodiazepines are needed.
In conclusion, although midazolam is generally used as premedication for relieving anxiety of surgery, the danger of anaphylactic shock after intramuscular injection of midazolam is not widely recognized. When it was injected intramuscularly, slow onset of severe anaphylactic shock can occur. Therefore, the surgery should be postponed when the anaphylactic reaction was suspected even though the symptoms were limited. Additionally, anesthesiologists should be prepared for the occurrence of anaphylaxis at any time in the perioperative period.
References
1. Brown SG, Mullins RJ, Gold MS. Anaphylaxis: diagnosis and management. Med J Aust 2006; 185: 283-289. PMID: 16948628.


2. Mertes PM, Alla F, Tr├®chot P, Auroy Y, Jougla E. Anaphylaxis during anesthesia in France: an 8-year national survey. J Allergy Clin Immunol 2011; 128: 366-373. PMID: 21497888.


3. Lienhart A, Auroy Y, P├®quignot F, Benhamou D, Warszawski J, Bovet M, et al. Survey of anesthesia-related mortality in France. Anesthesiology 2006; 105: 1087-1097. PMID: 17122571.


4. Soetens FM, Smolders FJ, Meeuwis HC, Van der Donck AG, Van der Aa PH, De Vel MA, et al. Intradermal skin testing in the investigation of suspected anaphylactic reactions during anaesthesia--a retrospective survey. Acta Anaesthesiol Belg 2003; 54: 59-63. PMID: 12703347.

5. Ebo DG, Fisher MM, Hagendorens MM, Bridts CH, Stevens WJ. Anaphylaxis during anaesthesia: diagnostic approach. Allergy 2007; 62: 471-487. PMID: 17441788.


6. Fujita Y, Ishikawa H, Yokota K. Anaphylactoid reaction to midazolam. Anesth Analg 1994; 79: 811-812. PMID: 7943804.

7. Shin JG, Hwang JH, Lee BS, Park HJ, Lee SH, Lee JN, et al. A case of midazolam anaphylaxis. Clin Endosc 2014; 47: 262-265. PMID: 24944992.



8. Ayuse T, Kurata S, Ayuse T. Anaphylactoid-like reaction to midazolam during oral and maxillofacial surgery. Anesth Prog 2015; 62: 64-65. PMID: 26061575.



9. Dewachter P, Mouton-Faivre C, Emala CW. Anaphylaxis and anesthesia: controversies and new insights. Anesthesiology 2009; 111: 1141-1150. PMID: 19858877.


10. Krishna MT, York M, Chin T, Gnanakumaran G, Heslegrave J, Derbridge C, et al. Multi-centre retrospective analysis of anaphylaxis during general anaesthesia in the United Kingdom: aetiology and diagnostic performance of acute serum tryptase. Clin Exp Immunol 2014; 178: 399-404. PMID: 25070464.



11. Alonso D├Łaz de Durana MD, Fern├Īndez-Rivas M, Casas ML, Esteban E, Cuevas M, Tejedor MA. Anaphylaxis during negative penicillin skin prick testing confirmed by elevated serum tryptase. Allergy 2003; 58: 159PMID: 12622750.


12. Ewan PW, Dugu├® P, Mirakian R, Dixon TA, Harper JN, Nasser SM. BSACI guidelines for the investigation of suspected anaphylaxis during general anaesthesia. Clin Exp Allergy 2010; 40: 15-31. PMID: 20205694.


13. Muraro A, Roberts G, Worm M, Bil├▓ MB, Brockow K, Fern├Īndez Rivas M, et al. Anaphylaxis: guidelines from the European Academy of Allergy and Clinical Immunology. Allergy 2014; 69: 1026-1045. PMID: 24909803.


14. Terico AT, Gallagher JC. Beta-lactam hypersensitivity and cross-reactivity. J Pharm Pract 2014; 27: 530-544. PMID: 25124380.


15. Del Pozo MD, Blasco A, Lobera T. Tetrazepam allergy. Allergy 1999; 54: 1226-1227. PMID: 10604563.


Fig.┬Ā1
Graphs of vital signs in the recovery room. At 25 minutes after appearance of the skin lesions, blood pressure dropped to 70/40 mmHg with pulse rate 98 beats/min, and pulse oxygen saturation dropped to 90%. Epinephrine 50 ┬Ąg and hydrocortisone 100 mg were injected intravenously with hydration to treat the anaphylactic shock. Thereafter, after transient tachycardia, blood pressure increased to 100ŌĆō110/50ŌĆō60 mmHg, and pulse rate decreased to 70 beats/min.