|
 |
|
|
Abstract
Coronary air embolism is a rare event. We report a case in which an acute myocardial infarction occurred in the region supplied by the right coronary artery after the removal of a double-lumen hemodialysis catheter. Emergent coronary angiography revealed air bubbles obstructing the mid-segment of the right coronary artery with slow flow phenomenon distally. The patient expired due to myocardial infarction.
Central venous catheterization (CVC) is an important practice in the intensive care unit. CVC is commonly used in critically ill patients for hemodynamic monitoring, administration of fluid to treat hypovolemia and shock, infusion of highly concentrated drugs and total parenteral nutrition, and for hemodialysis. Special care is usually taken during CVC insertion, with less attention given to CVC removal. Air embolism after CVC removal is a rare event, but when it occurs it leads to a variety of complications that can be fatal, which are related to the amount and speed of air into the bloodstream. Organs can experience the air lock phenomenon. We describe a case of a lethal coronary air embolism after removal of a double lumen hemodialysis catheter that was placed through the right internal jugular vein.
A 73-year-old man was admitted to our intensive care unit (ICU) via the emergency department with severe diarrhea, oliguria, and generalized edema. The patient's vital signs were blood pressure (BP) 85/50 mmHg, heart rate 120 beats/min, and drowsy mental status. His medical history included diabetes mellitus for 5 years with the operation of coil embolization in the right posterior communicating artery aneurysm 3 years previously.
On ICU admission, urinalysis revealed protein 4+ and serum albumin 2.1 g/dl. A chest radiograph showed a scant amount of pleural effusion. Generalized edema was evident on physical examination. To rule out nephrotic syndrome, he was scheduled for kidney biopsy. Also, he was diagnosed with prerenal acute kidney injury due to severe dehydration with laboratory test results of blood urea nitrogen 63 and creatinine (Cr) 3.0. Brain computed tomography (CT) showed no remarkable findings and mental status recovered with adequate fluid therapy. After the kidney biopsy, he was diagnosed with nephrotic syndrome with minimal change disease and prednisolone was administered. However, generalized edema did not get better and Cr remained high. The physician in charge of the patient judged that he had a relatively slight nephrotic syndrome with minimal change. Therefore, further evaluation, such as echocardiography was not considered as a treatment plan except for laboratory test and renal dialysis. He was determined to take dialysis and a 12 French, 2 lumen hemodialysis catheter (Niagara™, Slimcath® catheter; Bard, USA) was inserted in the right internal jugular vein. Generalized edema and serum Cr level improved for the 10 days of hemodialysis. Vital signs were stable and the patient was transferred to the general ward.
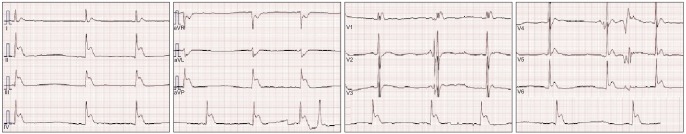
Because his renal function was improving, the physician decided to remove the hemodialysis catheter. With the patient in the supine position holding his breath, the catheter was removed slowly from the right internal jugular vein while the exit site was pressed with gauze for 5 minutes. After about 10 minutes, the spouse of another patient informed staff that the patient had suddenly slumped onto the floor of the ward. The attending physician and nurses entered the room and found the patient lying unconscious on the floor. He had no response to any stimulus. BP and pulse was not checked. Mental status was coma and Glasgow Coma Scale score was 3. Cardiopulmonary resuscitation and endotracheal intubation were performed immediately. The breathing sound was severe rale. The electrocardiogram recordings showed increase in ST segment of II, III, and aVF leads (Fig. 1). At first, air embolism due to catheter removal was not diagnosed. With a diagnosis of acute myocardial infarction at the inferior wall, coronary angiography (CAG) was performed immediately.
The angiography showed slow blood flow pattern which was compatible with thrombolysis in myocardial infarction (TIMI) grade 1 with multiple air bubbles in the mid segment of the right coronary artery (Fig. 2, Video 1). After the air passed, blood flow in the artery totally recovered to TIMI grade 3. Narrowing site or thrombus was not found in coronary arteries. The final diagnosis was acute myocardial infarction due to coronary air embolism because any narrowing site or thrombus was not found in coronary arteries as the cause of obstruction except for air bubbles.
Chest radiograph after the CAG revealed severe pulmonary edema (Fig. 3). A temporary pacemaker was placed. However, the patient was still pulseless. To assist the patient's impaired cardiopulmonary function, extracorporeal membrane oxygenation was performed and he was transferred to the ICU. Cardiac arrest began again and he expired.
Coronary air embolism is a rare complication of coronary catheterization, with a reported incidence of about 0.1%. [1]. Also, it can occur as a result of venous air embolism by many factors other than coronary catheterization. In one report, venous air embolism that occurred from uterus exteriorization during a cesarean section caused coronary air embolism through patent foramen ovale (PFO) [2]. The mechanism by which venous air penetrate into the arterial circulation is responsible for the paradoxical embolism by PFO. In the case presented, echocardiography was not performed because air embolism was not suspected to be the cause of cardiac arrest at first. And after the CAG, no diagnostic procedure for further evaluation could be performed due to low possibility of resuscitation. It was an imperfective point of our case that we were unable to demonstrate the presence of a PFO.
It is possible that the patient in our case had a PFO. If not, the filtrating property of the lungs is limited to the amount of air into the vein. Therefore, air in the vein can be transported to the artery by physiologic shunting of systemic circulation via pulmonary capillaries [3]. In one report, air embolism in the left ventricle occurred by the removal of a central venous catheter with echocardiographic finding which showed no PFO [4]. Air emboli in the arterial system can reach any organ. It is especially risky to have air bubbles in arteries supplying the brain or the heart because these organs are most vulnerable to hypoxia.
Signs and symptoms of coronary air embolism include chest pain, hypotension, transient electrocardiography changes consistent with myocardial ischemia, arrhythmias (bradycardia, heart block, ventricular tachycardia, and ventricular fibrillation), and even cardiac arrest [5]. The severity of cardiac impairment caused by coronary air embolism can be different from the amount of air injected into coronary artery and the patient's cardiac function state. A study in dogs showed that injection of 0.02 ml/kg air into the coronary artery resulted in a 28% rate of mortality [6]. Also, Khan et al. [7] classified the magnitude of air emboli based on the angiographic findings and severity of symptoms. They defined "massive emboli" as the injection of air bubbles greater than 3–5 ml, with the condition associated with serious complications. The present case had 3.5 ml of air bubbles in the right coronary artery, as estimated by the amount of air bubbles in the length of the right coronary mid segment observed in the CAG, the artery diameter, and the time of air passage. Considering the amount of air into the flow and fatal complications like acute coronary syndrome and severe pulmonary edema, the present case is consistent with the definition of massive air emboli.
Coronary air embolism can be conclusively diagnosed by the observation of air passing through the coronary artery in fluoroscopy or angiography. In general, air emboli passing distal to coronary arterial circulation results in slow flow phenomenon in epicardial vessels [8]. The treatment of coronary air embolism is most often the forceful injection of the patient's blood or saline to propel the air bubbles to the terminal portion of the coronary artery during angiography. Other treatments are breaking the air bubbles using guide wires or aspirating the air using a thrombectomy catheter [5]. If the amount of air is minimal, administration of 100% oxygen for 30 minutes can displace nitrogen from the air emboli, which causes shrinkage of the air bubbles [9].
Venous air embolism by CVC removal occurs when a fistula is formed between the skin and the vena cava due to a long-standing catheter. Fibrin tract may be formed around the catheter, usually after a long stay, but sometimes even within 24 hours [10]. Fibrin tract can be visible more than 24 hours after catheter removal and air occlusive dressing [11]. Therefore, the time of CVC should be as short as possible to avoid fistula formation between the central vein and the skin. Dressing should be done with air occlusive gauze and using inert ointment. Pressure should be firmly applied on the exit site for more than 10 minutes and dressing maintained for at least 24 hours.
Even if an air embolism occurs, the symptoms (from asymptomatic to fatal) depend on the volume and speed of the air. The factors determining the volume and speed of the air bubbles are the size of catheter lumen and the pressure gradient between the atmosphere and the patient's central vein. In the case of humans, death can occur when 200–300 ml of air is delivered into the right ventricle [12]. A 14 gauge catheter enables gas flow at the rate of 100 ml/s and a pressure of 5 cmH2O [13]. A double lumen hemodialysis catheter (12 French, 8.5 gauge) was used in the present case. Supposing that the pressure difference is 5 cmH2O, the volume flow rate of air capable passing through the catheter is calculated to be about 1280.99 ml/s by a modification of Poiseuille's law (Q = πr4(P1-P2)/8 nl, r = radius of tube, P1-P2 = pressure difference, n = viscosity of fluid, l = length of tube). When the air penetrates the vascular system some extent, air inflow will be blocked due to reversal of the pressure difference between the central vein and the atmosphere. However, it should be kept in mind that the velocity of air influx will be extremely fast even though the diameter of the catheter increases a little bit according to Poiseuille's law, in which the fluid speed is proportional to the four-square of the radius. It can be helpful to place the patient in the Trendelenburg position with the head pointing down at an angle of 10–30° during catheter removal. Because the venous pressure increases and therefore, the pressure difference decreases, less air will enter the blood. Using the same principle, in the case of co-operational patients, it is much safer to have the patient hold their breath or expire air during catheter removal.
When air embolism is detected, it is important to discover abnormal air in the heart, vena cava, or brain through echocardiography or CT scan images. In our case, CT or echocardiography was not performed because air embolism was not suspected at first. In general, when venous air embolism occurs, an electrocardiogram shows right ventricular strain pattern (T wave inversion, ST segment depression) as right ventricular outflow tract is obstructed. But the electrocardiogram at cardiac arrest in our case showed ST segment elevation, which was suggestive of acute myocardial infarction and the patient had a nephrotic syndrome accompanied by thrombosis. Therefore, he was first diagnosed with acute myocardial infarction due to thrombus and an emergent CAG was performed. If he were diagnosed with venous air embolism at first, CT or echocardiography would have been done, which would likely have discovered any air bubbles in the cardiac cavities. A chest X-ray taken after the CAG shows severe pulmonary edema as the distinctive "butterfly" pattern. This is thought to be the result of non-cardiogenic pulmonary edema from secretion of vasoactive mediators by pulmonary vascular obstruction and the cardiogenic pulmonary edema from acute myocardial infarction by coronary air embolism.
Venous air embolism can be treated as follows. First, stop the procedure that is creating the air embolism and turn the patient to the left lateral Trendelenburg position, to maintain air in the right atrium and prevent further air entry to the right ventricle. A second treatment is to remove air bubbles in the right atrium using CVC. Third, hyperbaric oxygen treatment is the most efficient way. It decreases the size of air bubbles by denitrification and improves the arterial oxygen gas saturation treating organ from ischemic damage.
CVC insertion is commonly used and is helpful in critical care. When CVC is inserted, special care and attention is necessary from a highly qualified specialist. However, there are many cases in which a relatively inexperienced physician removes CVC and neglects prevention. In the present case, a hemodialysis double lumen catheter was inserted to treat acute kidney injury, and it was well treated. But, air embolism was not prevented during catheter removal, resulting in the patient's death. The physician should have thought about the possibility about any fibrin tract due to a long-standing catheter and taken more attention to use air occlusive dressing, ensure the best position for the patient, and keep pressure on the exit site for the required length of time. Prevention is more important than treatment of air embolism. Thorough education about CVC removal complication and its prevention is needed.
References
1. Dodek A, Boone JA, Hooper RO, Kavanagh-Gray D, Macdonald IL, Peretz DI. Complications of coronary arteriography. Can Med Assoc J 1983; 128: 934-936. PMID: 6831339.


2. Nims M, Hallonquist H, Camann W. Coronary arterial air embolus occurring during cesarean delivery. Int J Obstet Anesth 2006; 15: 166-169. PMID: 16431100.


3. van Hulst RA, Klein J, Lachmann B. Gas embolism: pathophysiology and treatment. Clin Physiol Funct Imaging 2003; 23: 237-246. PMID: 12950319.


4. Cho DS, Kim MH, Lee DH, Lee HW, Kim EB, Kim SH, et al. Air embolism in the left ventricle after the removal of a central venous catheter. Korean J Crit Care Med 2013; 28: 318-322.

5. Dib J, Boyle AJ, Chan M, Resar JR. Coronary air embolism: a case report and review of the literature. Catheter Cardiovasc Interv 2006; 68: 897-900. PMID: 17086533.


6. Stegmann T, Daniel W, Bellmann L, Trenkler G, Oelert H, Borst HG. Experimental coronary air embolism. Assessment of time course of myocardial ischemia and the protective effect of cardiopulmonary bypass. Thorac Cardiovasc Surg 1980; 28: 141-149. PMID: 6156501.



7. Khan M, Schmidt DH, Bajwa T, Shalev Y. Coronary air embolism: incidence, severity, and suggested approaches to treatment. Cathet Cardiovasc Diagn 1995; 36: 313-318. PMID: 8719380.


8. Van Blankenstein JH, Slager CJ, Schuurbiers JC, Strikwerda S, Verdouw PD. Heart function after injection of small air bubbles in coronary artery of pigs. J Appl Physiol (1985) 1993; 75: 1201-1207. PMID: 8226530.


9. Groom AC, Farhi LE. Cutaneous diffusion of atmospheric N2 during N2 washout in the dog. J Appl Physiol 1967; 22: 740-745. PMID: 6023187.


10. Badin J, Coudroy R, Velasco S, Chatellier D, Robert R. Repetitive gas embolisms after central catheter removal. Intensive Care Med 2011; 37: 887-888. PMID: 21318435.


11. Marco M, Roman-Pognuz E, Anna B, Alessio S. Air embolism after central venous catheter removal: fibrin sheath as the portal of persistent air entry. Case Rep Crit Care 2013; 2013: 403243PMID: 24829822.




12. Toung TJ, Rossberg MI, Hutchins GM. Volume of air in a lethal venous air embolism. Anesthesiology 2001; 94: 360-361. PMID: 11176104.


13. Flanagan JP, Gradisar IA, Gross RJ, Kelly TR. Air embolus--a lethal complication of subclavian venipuncture. N Engl J Med 1969; 281: 488-489. PMID: 5796967.