 |
 |
|
|
Abstract
We report a case of 29-year-old, morbidly obese, diabetic primigravida who had undergone previously primary percutaneous coronary intervention with stent placement for an inferior wall myocardial infarction at 10 weeks of gestation. She remained asymptomatic with medication during the remainder of her pregnancy, but preoperative echocardiography revealed left ventricular dilation and a restrictive diastolic dysfunction with a preserved ejection fraction (46%). She developed acute pulmonary edema associated with hypertension after an elective Cesarean delivery under continuous epidural anesthesia despite the meticulous restriction of fluid.
Immediate postpartum involves the greatest cardiac output of pregnancy because of the enhanced venous return due to the relief of vena caval compression and a shift of the blood from the contracting emptied uterus into the systemic circulation. Although well tolerated in most gravidae, the increased hemodynamic load in the postpartum period can result in heart failure (HF) in women with cardiac disease.
Acute pulmonary edema in patients with coronary artery disease is a dramatic manifestation of HF. It has recently recognized that patients with acute pulmonary edema are frequently hypertensive on presentation and have a preserved systolic function even after coronary revascularization [1,2]. More than 90% of patients who had HF have a diastolic dysfunction regardless of the left ventricular ejection fraction (LVEF) [3]. These findings suggest that the diastolic dysfunction is an important contributor to acute hypertensive pulmonary edema in patients with coronary artery disease.
We report a primigravida with an antepartum acute myocardial infarction (AMI) who developed acute hypertensive pulmonary edema after an elective Cesarean section under epidural anesthesia at 37 weeks of gestation.
A 29-year-old primigravida with previous antepartum AMI was admitted for delivery at 37 weeks of gestation. She had a six-year history of metabolic syndrome (type 2 diabetes mellitus, hyperlipidemia and central obesity) and a family history of ischemic heart disease. She had undergone primary percutaneous coronary intervention (PCI) with the placement of a stent in the right coronary artery due to a ST-segment elevation myocardial infarction (STEMI) in the inferior wall at 10 weeks of gestation. The postcatheterization echocardiogram showed hypokinesia of the inferior walls and LV enlargement with LVEF of 55%. She remained asymptomatic with beta-blocker, aspirin and clopidogrel medication during the remainder of her pregnancy.
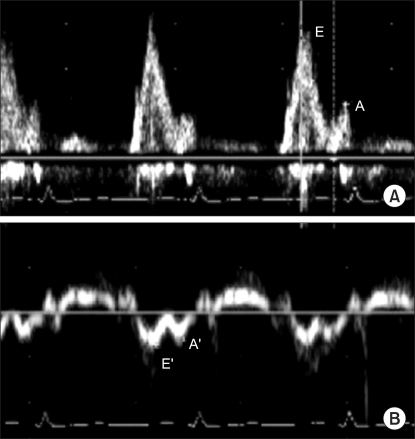
Considering the fetopelvic disproportion, a decision was made to perform an elective Cesarean section under epidural anesthesia at 37 weeks. Upon admission for surgery, her weight was 126 kg (height 170 cm, BMI 44 kg/m2). She showed neither dyspnea nor hypotension in the supine position. The preoperative echocardiogram showed a more enlarged LV (LV end-diastolic/systolic dimension 67/51 mm2) with a restrictive diastolic dysfunction despite a LVEF of 46% without a newly developed regional wall motion abnormality (Fig. 1). The chest radiograph showed cardiomegaly without consolidation in both lungs. ECG demonstrated a Q wave in leads II, III and aVF, and inverted T wave in leads III and V1. Her blood tests were within the normal range except for the HbA1c level (7.6%).
Aspirin and clopidogrel were withheld 7 days before surgery. The patient continued with the other cardiac medication except on the morning of surgery and received ranitidine 150 mg and metoclopramide 10 mg intravenously as premedication. She was monitored with a five-electrode ECG (II, V5), pulse oximetry and intraarterial blood pressure in the operating room. Upon arrival in the operating room, her blood pressure was 130/90 mmHg and heart rate was 75 beats/min. Her saturation was 100% with supplemental oxygen (5 L/min) via a face mask. An isosorbide dinitrate infusion was run at rate of 0.1 ┬Ąg/kg/min to prevent a recurrent ischemic attack. An intravenous infusion of 50 ml of crystalloid solution was started prior to commencement of the regional block. An epidural catheter was inserted at the L3-4 interspace using the midline approach followed a test dose of 3 ml of 1.5% lidocaine in the left lateral position. Epinephrine was not included in the local anesthetic solution because an unintentional intravascular injection of epinephrine can produce maternal tachycardia. A total 30 ml of 2% lidocaine mixed with fentanyl 100 ┬Ąg was titrated to achieve a T4 sensory block in the supine position with a wedge under the right hip.
A 4,130 g live male baby with Apgar scores of 8/10 at 1/5 minutes was delivered, which was followed by 100 ┬Ąg of intravenous carbetocin to control the postpartum hemorrhage. Her heart rate was 70-91 beats/min and the arterial blood pressure was 120/60-140/90 mmHg throughout the procedure. The estimated blood loss and urine output was 600 ml and 200 ml, respectively. A total 250 ml of crystalloid fluid was administrated intravenously during the 50-minute operation.
After surgery, the patient was admitted to the intensive care unit. The pain scores were within the acceptable ranges with patient controlled epidural analgesia using 0.1% levobupivacaine with sufentanil. Although she continued isosorbide dinitrate infusion, she developed hypertension to 180/110 mmHg and tachycardia to 112 beats/min 2 hours after surgery and complained of orthopnea. A follow-up ECG showed sinus tachycardia without arrhythmia or ischemia. The postoperative cardiac biochemical markers were within normal range, but the plasma B-type natriuretic petide (BNP) was 435 pg/ml. Although the oxygen saturation was >95% with the administration of O2 (5 L/min) through a face mask, the chest x-ray showed bilateral pulmonary congestion and cardiomegaly (Fig. 2). The patient was treated with intravenous isosorbide dinitrate, labetalol and furosemide as well as fluid restriction. Her blood pressure and heart rate decreased immediately to 135-140/50-70 mmHg and 90-100 beats/min, respectively, and her oxygen saturation increased to 98%, which was maintained throughout the ICU stay. The clinical and radiologic evidence of pulmonary edema were resolved with the control of hypertension and diuresis of 2,200 ml for the subsequent 5 hours. The intake and output for 24 hours postpartum were 2,470 ml and 4,320 ml, respectively. The patient was transferred to the maternal ward 48 hours postpartum and discharged on the seventh day postoperatively with a prescription of a beta-blocker and clopidogrel.
Coronary artery disease has recently become the main cause of acute hypertensive pulmonary edema [1,2]. An ischemic insult to the myocardium induces a series of changes in shape, size and thickness in both the infracted and noninfarcted segment of the LV, which is referred to LV remodeling. LV remodeling is a dynamic process that begins in the acute phase with an infarct expansion and subsequent LV dilation leading to HF, and is affected by changes in the hemodynamic load and neurohormonal activation [4].
Early reperfusion and sustained patency of an infarct-related coronary artery is known to attenuate LV remodeling by limiting the infarct expansion and preserving the LV function [5,6]. However, recent studies reported that LV dilation occurred in a considerable proportion of patients with AMI, who were treated successfully with primary PCI despite the persistent patency of the infarct-related artery [7,8].
Our patient remained asymptomatic during the reminder of her pregnancy but showed progressive LV dilation and restrictive diastolic dysfunction on echocardiography 6 months after STEMI. This shows that the symptoms alone are not a good indicator of the LV remodeling process. Despite the early reperfusion with primary PCI and subsequent medication with beta blocker, the increases in hemodynamic load during pregnancy in conjunction with metabolic syndrome might have contributed to the post-MI LV remodeling in this patient.
Acute pulmonary congestion in a patient with coronary artery disease results from a transient ischemic LV systolic and/or diastolic dysfunction, or acute mitral regurgitation. Although HF is believed to arise primarily from a depressed systolic function, patients with pulmonary edema due to an ischemic LV systolic function or acute mitral regurgitation are unable to generate a high systolic pressure. Most patients with hypertensive pulmonary edema have a preserved systolic function, and the LVEF and regional wall motion are similar both during the acute episode and after the resolution of symptoms [1,2]. In addition, the severity of HF is related to the degree of diastolic dysfunction, even in patients with reduced LVEF [3]. This indicates that hypertension contributes to a worsening diastolic dysfunction, resulting in pulmonary congestion in patients with coronary artery disease.
The main diastolic abnormality is impaired relaxation in a patient with AMI, but a restrictive filling pattern may be observed due to an increase in resistance to LV filling or in chamber stiffness [9]. Hypertension usually results from neuroendocrine stimulation due to pain or stress response, increased preload and tachycardia, which impairs the LV relaxation and reduces the diastolic compliance further [10]. In these patients, a small increase in blood volume might be associated with a marked increase in diastolic pressure due to the stiff ventricle.
Our patient maintained adequate epidural anesthesia and stable hemodynamics in the absence of volume and phenylephrine infusion during surgery under titrated epidural anesthesia. However, she developed pulmonary congestion with an abrupt increase in blood pressure despite the acceptable range of postoperative epidural analgesia in the immediate postpartum period. Considering the clinical findings and the negative fluid balance, acute pulmonary congestion occurred as a result of the increased blood volume from the empty uterus and the relief of vena caval compression that normally occurs during the early postpartum period in a post-MI patient with a diastolic dysfunction. In addition, hypertension due to the increased preload and/or adrenergic activation accompanying the respiratory distress of pulmonary edema exacerbated the diastolic dysfunction. Although the cardiac function was not assessed with echocardiography and invasive hemodynamic monitoring, the diastolic dysfunction was correlated with an elevation of the BNP levels during the acute episode [3]. A transient ischemic systolic dysfunction may have contributed to the diastolic dysfunction without causing a measurable decrease in the LVEF or regional wall motion [11]. However, it was not considered to be the primary cause of pulmonary congestion in our patient because there was no change in the serial measurements of ECG and the cardiac biomarker enzyme levels.
Treatment of this condition includes the control of hypertension, cautious diuresis, and ventilatory support if necessary. Strict control of hypertension appears to be of paramount importance because lowering the systolic pressure allows the LV to eject to a smaller end-systolic volume, and rapidly reducing the pulmonary congestion caused by a diastolic dysfunction [2]. Our patient was treated with meticulous restriction of fluid and a combination of intravenous nitroglycerin, diuretic, and beta-adrenergic blockers. The nitroglycerin and diuretics would be expected to reduce the blood volume and pulmonary venous return, thereby reduce the pulmonary congestion. A beta-adrenergic blocker, while generally contraindicated in the management of HF in patients with a systolic dysfunction, plays an important role in the treatment of diastolic dysfunction by decreasing the heart rate and prolonging the diastolic filling time, thereby facilitating LV relaxation and filling. In patients with pulmonary edema, hypertension is associated with a better short-term prognosis than hypotension because of the reversible and transient diastolic dysfunction [12].
In conclusion, careful management of the intravascular volume and the control of hypertension is essential for preventing acute pulmonary edema due to the hemodynamic changes that occur during the immediate postpartum period in a post-myocardial infarction parturient.
References
1. Kramer K, Kirkman P, Kitzman D, Little WC. Flash pulmonary edema: association with hypertension and reoccurrence despite coronary revascularization. Am Heart J 2000; 140: 451-455. PMID: 10966547.


2. Gandhi SK, Powers JC, Nomeir AM, Fowle K, Kitzman DW, Rankin KM, et al. The pathogenesis of acute pulmonary edema associated with hypertension. N Engl J Med 2001; 344: 17-22. PMID: 11136955.


3. Brucks S, Little WC, Chao T, Kitzman DW, Wesley-Farrington D, Gandhi S, et al. Contribution of left ventricular diastolic dysfunction to heart failure regardless of ejection fraction. Am J Cardiol 2005; 95: 603-606. PMID: 15721099.


4. Cohn JN, Ferrari R, Sharpe N. Cardiac remodeling--concepts and clinical implications: a consensus paper from an international forum on cardiac remodeling. Behalf of an International Forum on Cardiac Remodeling. J Am Coll Cardiol 2000; 35: 569-582. PMID: 10716457.


5. Keeley EC, Boura JA, Grines CL. Primary angioplasty versus intravenous thrombolytic therapy for acute myocardial infarction: a quantitative review of 23 randomised trials. Lancet 2003; 361: 13-20. PMID: 12517460.


6. Lopes LR, Joao I, Vinhas H, Simoes O, Cotrim C, Catarino C, et al. Evaluation of systolic and systo-diastolic function: the Tei index in acute myocardial infarction treated with acute reperfusion therapy--early and late evaluation. Rev Port Cardiol 2007; 26: 649-656. PMID: 17849948.

7. Bolognese L, Neskovic AN, Parodi G, Cerisano G, Buonamici P, Santoro GM, et al. Left ventricular remodeling after primary coronary angioplasty: patterns of left ventricular dilation and long-term prognostic implications. Circulation 2002; 106: 2351-2357. PMID: 12403666.


8. Loboz-Grudzień K, Kowalska A, Brzezińka B, Sokalski L, Jaroch J. Early predictors of adverse left ventricular remodelling after myocardial infarction treated by primary angioplasty. Cardiol J 2007; 14: 238-245. PMID: 18651467.

9. Cerisano G, Pucci PD, Valenti R, Boddi V, Migliorini A, Tommasi MS, et al. Comparison of the usefulness of Doppler-derived deceleration time versus plasma brain natriuretic peptide to predict left ventricular remodeling after mechanical revascularization in patients with ST-elevation acute myocardial infarction and left ventricular systolic dysfunction. Am J Cardiol 2005; 95: 930-934. PMID: 15820157.


10. Leite-Moreira AF, Gillebert TC. Nonuniform course of left ventricular pressure fall and its regulation by load and contractile state. Circulation 1994; 90: 2481-2491. PMID: 7955206.


12. Tribouilloy C, Rusinaru D, Leborgne L, Mahjoub H, Szymanski C, Houpe D, et al. In-hospital mortality and prognostic factors in patients admitted for new-onset heart failure with preserved or reduced ejection fraction: a prospective observational study. Arch Cardiovasc Dis 2008; 101: 226-234. PMID: 18654097.


Fig.┬Ā1
Pulsed wave Doppler analysis showing a restrictive diastolic dysfunction. (A) Transmitral Doppler flow velocity revealed elevated early diastolic filling (E-wave; 14 cm/s) relative to late diasrolic filling (A-wave; 6 cm/s) with an increased E/A ratio (>2.0) and short deceleration time (<150 ms). (B) Doppler tissue imaging of the mitral annulus velocity revealed a decreased early (E'-wave; 7 cm/s) and late (A'-wave; 6 cm/s) diastolic tissue velocity with an E/E' ratio >15.
- TOOLS
-
METRICS

-
- 0 Crossref
- 1 Scopus
- 2,739 View
- 21 Download








