|
 |
|
|
Abstract
Background
We purposed to systemically review studies investigating the prophylactic effect of both pharmacological and non-pharmacological modalities against rocuronium induced withdrawal movement (RIWM) in the Korean population.
Methods
Literature search was performed using MEDLINE, EMBASE, CENTRAL, Koreamed, KMBASE, KISS and RISS up to March 2014. Randomized controlled trials (RCTs) comparing pharmacological and non-pharmacological interventions with placebo aimed for the Korean population were included. Outcome measures were the incidence and severity of RIWM. We conducted subgroup analyses according to each intervention method.
Results
Data were analyzed from 41 RCTs totaling 4,742 subjects. The overall incidence of RIWM was about 80% (range 56-100%). Incidence and severity of RIWM were significantly reduced with lidocaine (risk ratio [RR] 0.60, 95% CI 0.49-0.74; standardized mean difference [SMD] -0.74, 95% CI -1.05 to -0.44), opioids (RR 0.28, 95% CI 0.18-0.44; SMD -1.71, 95% CI -2.09 to -1.34) and hypnotics (RR 0.36, 95% CI 0.25-0.52; SMD -2.20, 95% CI -2.62 to -1.79). Regardless of tourniquet use, lidocaine showed a prophylactic effect against incidence and severity of RIWM: tourniquet (RR 0.36, 95% CI 0.21-0.62; SMD -1.51, 95% CI -2.15 to -0.86); non-tourniquet (RR 0.58, 95% CI 0.47-0.71; SMD -0.74, 95% CI -1.05 to -0.44). Dilution and slow injection of rocuronium decreased incidence and severity of RIWM: dilution (RR 0.47, 95% CI 0.39-0.56; SMD -1.64, 95% CI -2.47 to -0.81); slow injection (RR 0.34, 95% CI 0.17-0.70; SMD -2.13, 95% CI -2.74 to -1.51).
Rocuronium is a non-depolarizing muscle relaxant with a rapid onset and an intermediate duration of action, which is used widely during induction of general anesthesia [1]. However, it is often associated with injection pain, the cause of which is unclear. A number of studies have reported rocuronium injection pain, with an incidence of up to 80% [2,3,4]. Rocuronium injection pain can appear as a withdrawal movement of patients during induction of general anesthesia [5]. Rocuronium-induced withdrawal movement (RIWM) may be dangerous because it can lead to difficult intravenous injection of the drug and increase the risk of a patient falling out of bed. Pulmonary aspiration by regurgitation of gastric content secondary to RIWM in a pediatric patient has been reported [6]. Thus, many pharmacological and non-pharmacological interventions have been investigated in an attempt to reduce RIWM; results were variable. Interestingly, most studies of RIWM prevention were aimed at the Korean population. A previous meta-analysis evaluated the incidence of RIWM by pharmacological intervention, but the literature search was based only on international databases and participant race was not restricted [7].
Therefore, we systemically reviewed studies that investigated the prophylactic effect of both pharmacological and non-pharmacological methods against RIWM in the Korean population using international and domestic databases. The prophylactic effect against the incidence of RIWM of the proposed intervention methods was the primary outcome; RIWM severity was also assessed.
This study was conducted using the protocol recommended by the Cochrane Collaboration and the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement [8,9].
The search was performed in March 2014 through: domestic databases including Koreamed (http://www.koreamed.org), KMBASE (http://kmbase.medric.or.kr), KISS (http://kiss.kstudy.com), and RISS (http://riss4u.net); international databases including EMBASE, MEDLINE, and CENTRAL were also searched. The international database search was performed using the terms "rocuronium" AND ("injection pain" OR "withdrawal"). It was difficult to identify potentially relevant literature in the domestic database; therefore, the domestic database search was performed using the term "rocuronium" to increase sensitivity, and articles were excluded manually. The reference lists of all identified reports were also searched manually.
Studies included in our analysis were selected based on the following criteria. (1) Peer-reviewed randomized controlled trials (RCTs), (2) studies that compared any type of intervention group with a control group (no treatment or normal saline) to prevent or decrease RIWM, (3) reported outcome measures for the incidence or severity of RIWM and (4) written in Korean or English. Review articles, case reports, case-series, letters to the editor, commentaries, proceedings, laboratory science studies, and any non-relevant studies were excluded from the analysis. Two authors (G.J.C. and S.G.P.) independently selected eligible studies, and discussed any differences of opinion to arrive at a consensus as to whether a study should be included or excluded. Disagreement over inclusion or exclusion was settled by discussion with another author (H.K.). Based on the predetermined selection criteria, two of the authors (S.L. and J.H.L.) independently selected all trials retrieved from the databases and bibliographies. Disagreements between evaluators were resolved by discussion or in consultation with a third author (H.K.).
Two authors (G.J.C. and S.G.P.) independently extracted the following data using standardized data extraction form from the studies included in the final analysis: patient characteristics, age, rocuronium dose, intervention categories (pharmacological and non-pharmacological), intervention method, and grading system for RIWM severity. In cases of insufficient or missing data, data were selected either from the text or from tables, or when possible, calculated from the relevant data within the study.
The intervention categories were: pharmacological, including lidocaine, opioids (remifentanil, alfentanil, and fentanyl), sodium bicarbonate, hypnotics (thiopental and ketamine), inhalation anesthetics and other drugs; non-pharmacological, including venous occlusion, dilution, administration velocity, and temperature. The methods of intervention were: pretreatment (pharmacological administration before rocuronium injection); admixture (pharmacological administration mixed with rocuronium); inhalation; administration with venous occlusion using a tourniquet; dilution of rocuronium; and different velocities of rocuronium administration (slow or fast); and low temperature rocuronium.
The incidence of RIWM was the primary outcome in this meta-analysis, and severity of RIWM was the secondary outcome. We calculated means and standard deviations to evaluate RIWM severity for studies that used an ordinal scale with incidence. We combined all interventional groups for dose-dependency studies that included more than one interventional group to avoid multiple counting of the same individuals in the control group.
The quality of eligible studies was assessed independently by two authors (S.L. and J.H.L.) using the "risk of bias" tool provided in the Review Manager ver. 5.1 software (The Cochrane Collaboration, Oxford, UK). Quality was evaluated based on the following six potential sources of bias: random sequence generation; allocation concealment; blinding of participants; blinding of the outcome assessors; incomplete outcome data; and selective reporting. The methodology for each trial was graded as "high", "low" or "unclear", to reflect a high risk of bias, low risk of bias, or uncertainty of bias, respectively.
We computed the pooled risk ratio (RR) with corresponding 95% confidence interval (CI) for dichotomous data, and standardized mean difference (SMD) or mean difference (MD) with a 95% CI for continuous data. We used the chi-square (chi2) test for homogeneity and the I2 test for heterogeneity. We regarded a level of 10% significance (P < 0.1) in the chi-square statistic or an I2 > 50% as considerable heterogeneity, and used the Mantel-Haenszel random-effect model. Otherwise, the Mantel-Haenszel fixed-effect model was applied [8,10].
Subgroup analyses were performed according to the following characteristics: intervention method; opioid type; tourniquet use; and age group (adults or children). We also conducted a sensitivity analysis to evaluate the influence of the individual studies on the overall effect estimate by excluding one study at a time for outcomes with considerable heterogeneity.
Funnel plots were drawn for each dataset as a measure of publication bias across studies, and were assessed visually for symmetry. Considering the small study effect, we also estimated publication bias using Egger's linear regression test. If the funnel plot was asymmetrical or if the P value was < 0.1 by Egger's test, the presence of publication bias was considered, and trim and fill analyses were performed.
We performed all analyses using the Review Manager ver. 5.1 software (The Cochrane Collaboration, Oxford, UK) and Comprehensive Meta-analysis ver. 2.0 software (Biostat, Englewood, NJ, USA).
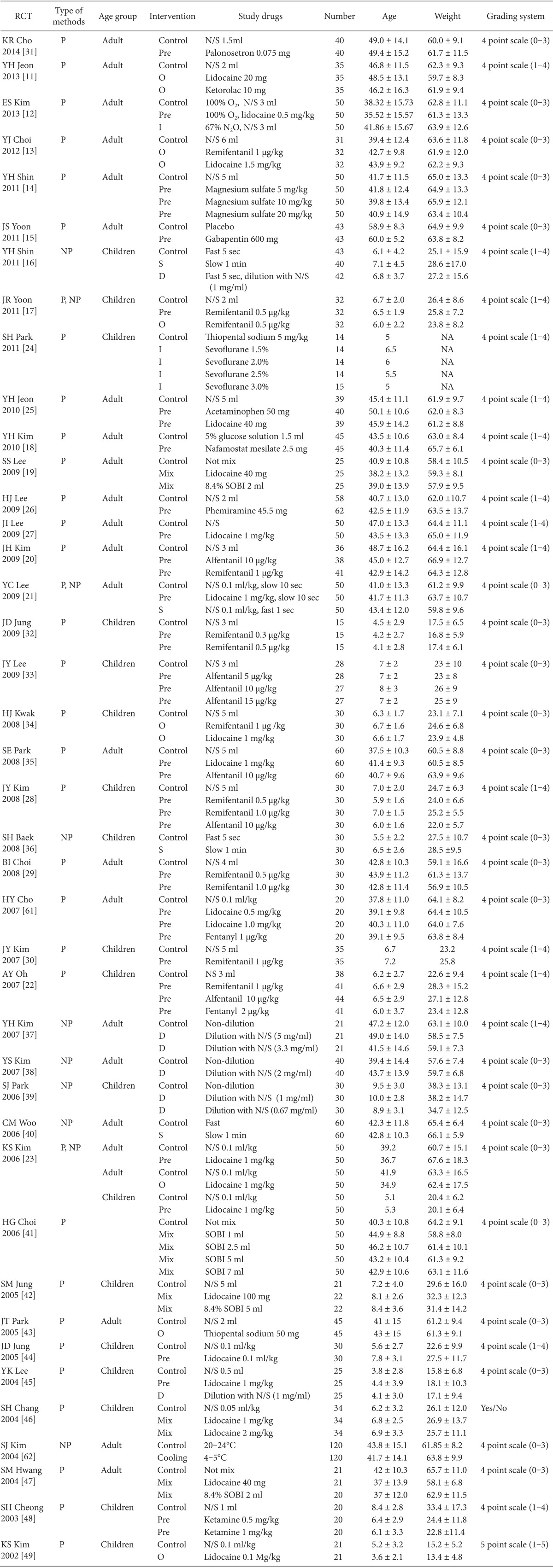
We extracted 262 records from international databases and 44 records from domestic databases. We excluded 47 duplicates between the international and domestic databases. After 183 articles were excluded by screening the titles and abstracts, 76 full-text articles were evaluated. Of the 76 full-text articles, 35 were excluded because of the following: not of interest (n = 24); no control group (n = 9); meta-analysis (n = 1); and letter to the editor (n = 1). Finally, 41 articles were included in this systematic review (Fig. 1). Relevant characteristics of the trials included are given in Table 1.
Thirteen studies mentioned the method of random sequence generation [11,12,13,14,15,16,17,18,19,20,21,22,23], and 10 studies used allocation concealment [11,14,23,24,25,26,27,28,29,30]. Patients were blinded in 39 studies [11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49], whereas outcome assessors were blinded in 25 studies [11,12,13,16,17,18,19,20,21,22,23,24,25,27,28,29,30,31,34,35,36,37,38,40,42]. Seven studies presented a clear explanation for withdrawals and dropouts in each group [12,13,15,16,24,25,33] (Fig. 2).
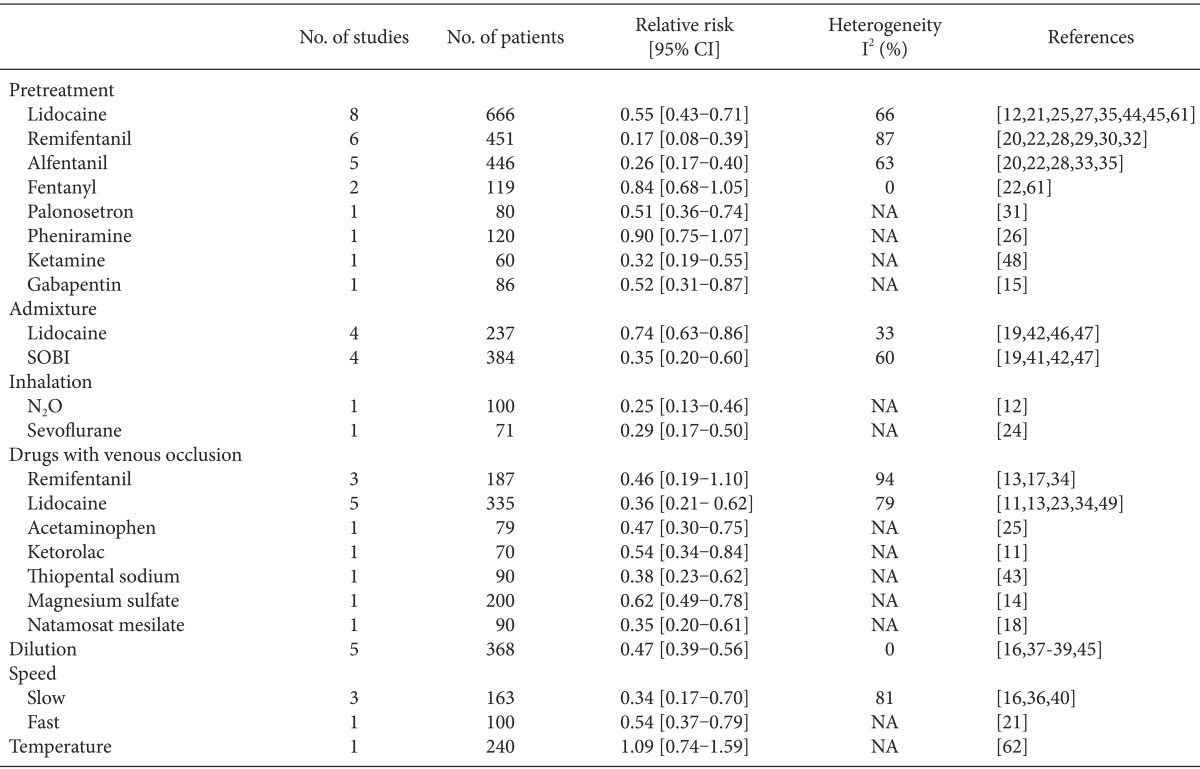
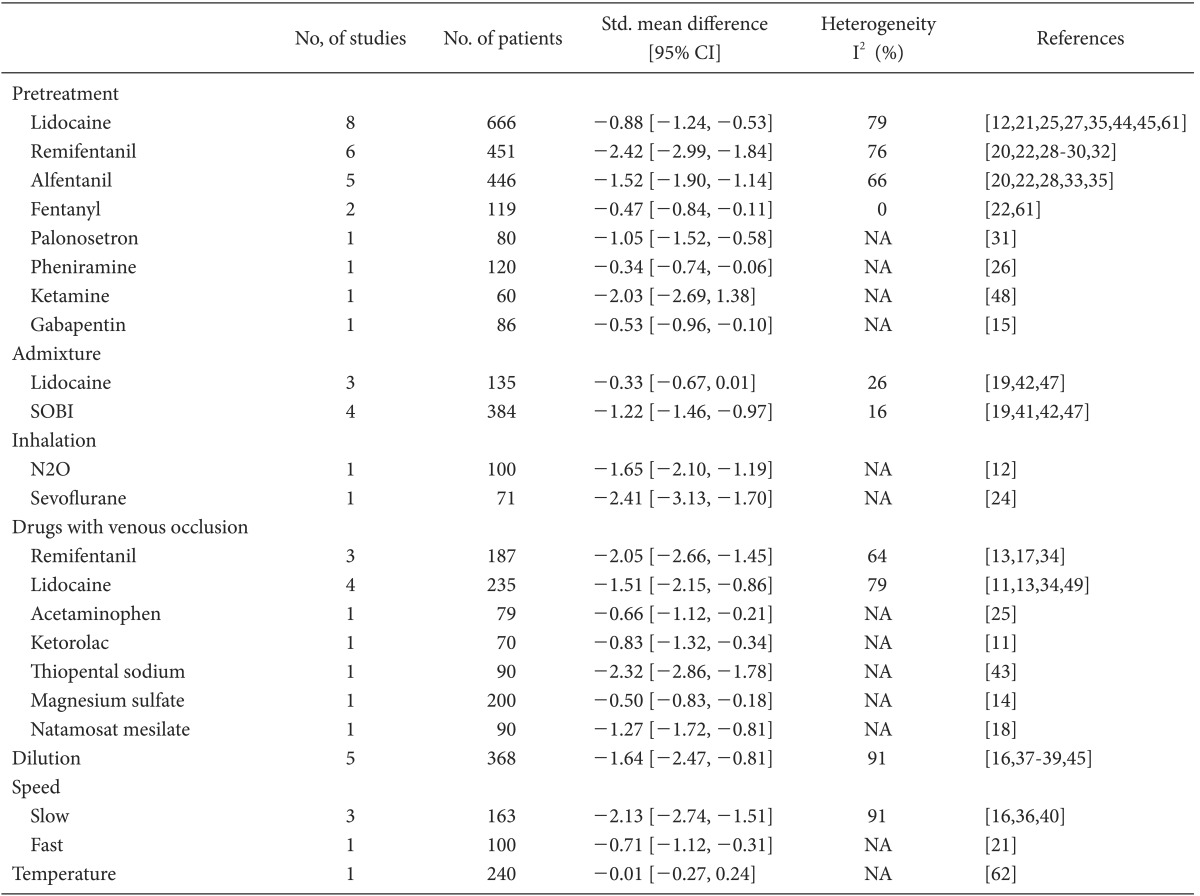
The incidence and severity of RIWM according to the method of intervention are presented in Tables 2 and 3.
Lidocaine showed a prophylactic effect against both the incidence and severity of RIWM (RR, 0.60; 95% CI, 0.49-0.74, pchi2 < 0.0001, I2 = 72%; SMD, -0.74; 95% CI, -1.05 to -0.44, pchi2 < 0.00001, I2 = 76%, respectively). Both the incidence and severity of RIWM decreased significantly in the lidocaine group compared with that in the control group when lidocaine was administered as a pretreatment (RR, 0.55; 95% CI, 0.43-0.71, pchi2 = 0.004, I2 = 66%; SMD, -0.88; 95% CI, -1.24 to -0.53, pchi2 < 0.00001, I2 = 79%, respectively; Tables 2 and 3 and Fig. 3). The lidocaine group had a lower incidence of RIWM than the control group when lidocaine was administered as an admixture (RR, 0.74; 95% CI, 0.63-0.86, pchi2 = 0.21, I2 = 33%; Table 2 and Fig. 3), whereas severity was not different between the lidocaine and control groups (SMD, -0.33; 95% CI, -0.67-0.01, pchi2 = 0.26, I2 = 26%; Table 3). Regardless of tourniquet use, lidocaine had a prophylactic effect in terms of the incidence and severity of RIWM: tourniquet (RR, 0.36; 95% CI, 0.21-0.62, pchi2 = 0.0007, I2 = 79%; SMD, -1.51; 95% CI, -2.15 to -0.86, pchi2 = 0.003, I2 = 79%); non-tourniquet (RR, 0.58; 95% CI, 0.47-0.71, pchi2 < 0.00001, I2 = 78%; SMD, -0.74; 95% CI, -1.05 to -0.44, pchi2 < 0.00001, I2 = 76%, respectively). Regardless of age group (adults vs. children), lidocaine reduced the incidence and severity of RIWM; adults (RR, 0.57; 95% CI, 0.48-0.68, pchi2 = 0.02, I2 = 51%; SMD, -0.79; 95% CI, -1.04 to -0.53, pchi2 = 0.0006, I2 = 68%, respectively); children (RR, 0.54; 95% CI, 0.36-0.81, pchi2 < 0.00001, I2 = 90%; SMD, -1.40; 95% CI, -2.47 to -0.34, pchi2 < 0.00001, I2 = 91%, respectively).
Opioids showed a prophylactic effect against both the incidence and severity of RIWM (RR, 0.28; 95% CI, 0.18-0.44, pchi2 < 0.00001, I2 = 90%; SMD, -1.71; 95% CI, -2.09 to -1.34; pchi2 < 0.00001, I2 = 87%, respectively; Fig. 4). Opioids-including fentanyl, remifentanil, and alfentanil-were administered as pretreatments. Fentanyl did not show a prophylactic effect against the incidence of RIWM (RR, 0.84; 95% CI, 0.68-1.05, pchi2 = 0.39, I2 = 0%; Table 2 and Fig. 4), whereas it showed a prophylactic effect against the severity of RIWM (SMD, -0.47; 95% CI, -0.84 to -0.11, pchi2 = 0.33, I2 = 0%; Table 3). Both remifentanil and alfentanil had a prophylactic effect against RIWM incidence and severity: remifentanil (RR, 0.17; 95% CI, 0.08-0.39, pchi2 < 0.00001, I2 = 87%; SMD, -2.42; 95% CI, -2.99 to -1.84, pchi2 = 0.002, I2 = 76%, respectively; Tables 2 and 3, and Fig. 4); alfentanil (RR, 0.26; 95% CI, 0.17-0.40, pchi2 = 0.03, I2 = 63; SMD, -1.52; 95% CI, -1.90 to -1.14, pchi2 = 0.02, I2 = 66%, respectively; Tables 2 and 3, and Fig. 4). Remifentanil with venous occlusion showed no prophylactic effect against the incidence of RIWM (RR, 0.46; 95% CI, 0.19-1.10, pchi2 < 0.00001, I2 = 94%; Table 2), while remifentanil with tourniquet reduced the severity of RIWM (SMD, -2.05; 95% CI, -2.66 to -1.45, pchi2 = 0.06, I2 = 64%; Table 3). Remifentanil without tourniquet decreased the incidence and severity of RIWM (RR, 0.20; 95% CI, 0.15-0.26, pchi2 < 0.00001, I2 = 83%; SMD, -2.30; 95% CI, -2.57 to -2.02, pchi2 = 0.002, I2 = 76%, respectively).
Sodium bicarbonate given as an admixture had a prophylactic effect against both the incidence and severity of RIWM (RR, 0.35; 95% CI, 0.20-0.60, pchi2 = 0.06, I2 = 60%; SMD, -1.22; 95% CI, -1.46 to -0.97, pchi2 = 0.31, I2 = 16%, respectively; Tables 2 and 3).
The hypnotics including ketamine and thiopental, which were given as pretreatments, had a prophylactic effect against both the incidence and severity of RIWM (RR, 0.36; 95% CI, 0.25-0.52, pchi2 = 0.65, I2 = 0%; SMD, -2.20; 95% CI, -2.62 to -1.79, pchi2 = 0.51, I2 = 0%, respectively).
Diluting rocuronium decreased the incidence and severity of RIWM (RR, 0.47; 95% CI, 0.39-0.56, pchi2 = 0.86, I2 = 0%; SMD, -1.64; 95% CI, -2.47 to -0.81, pchi2 < 0.00001, I2 = 91%, respectively; Tables 2 and 3). Regardless of age group (adults vs. children), diluting rocuronium reduced the incidence and severity of RIWM; adults (RR, 0.41; 95% CI, 0.27-0.62, pchi2 = 0.80, I2 = 0%; SMD, -0.88; 95% CI, -1.23 to -0.53, pchi2 = 0.30, I2 = 9%, respectively); children (RR, 0.49; 95% CI, 0.41-0.59, pchi2 = 0.68, I2 = 0%; SMD, -1.73; 95% CI, -1.94 to -1.52, pchi2 = 0.25, I2 = 27%, respectively).
Slow (>30-s injection time) and fast (<1-s injection time) administration of rocuronium decreased the incidence and severity of RIWM: slow administration (RR, 0.34; 95% CI, 0.17-0.70, pchi2 = 0.005, I2 = 81%; SMD, -2.13; 95% CI, -2.74 to -1.51, pchi2 < 0.00001, I2 = 91%; Tables 2 and 3); fast administration (RR, 0.54; 95% CI, 0.37-0.79; MD, -0.71; 95% CI, -1.12 to -0.31). Lowering the temperature of rocuronium did not reduce either the incidence or severity of RIWM (RR, 1.09; 95% CI, 0.74-1.59; MD, -0.01; 95% CI, -0.27-0.24).
We assessed publication bias using a funnel plot and Egger's test for the incidence of RIWM by intervention method using lidocaine, opioids, sodium bicarbonate, and diluted rocuronium. The funnel plots were asymmetric in the comparisons of lidocaine and opioids, whereas the funnel plots were symmetric in those using sodium bicarbonate and diluted rocuronium. Egger's test resulted in the following: lidocaine (P = 0.01); opioids (P < 0.001); sodium bicarbonate (P = 0.55); diluted rocuronium (P = 0.51). Based on the assumption that the intervention groups using lidocaine and opioids showed publication bias, we performed trim and fill analyses to evaluate the influence of publication bias (Figs. 5 and 6). The RR with 95% CIs remained significant: lidocaine (RR, 0.71; 95% CI, 0.59-0.85); opioids (RR, 0.56; 95% CI, 0.41-0.78).
The various types of pharmacological and non-pharmacological interventions demonstrated in the 41 RCTs with 4,742 subjects in the Korean population were associated with a reduced incidence or severity of RIWM, with the exception of RIWM severity when a lidocaine admixture was used and the incidence of RIWM when fentanyl was used as a pretreatment.
Although numerous studies have reported rocuronium-induced injection pain, the underlying mechanism remains unclear. The low pH of rocuronium is a possible cause of the pain, as rocuronium bromide is formulated with sodium acetate, sodium chloride, or acetic acid to produce a solution of pH 4.0 [50]. The low pH and osmolality of rocuronium solution could stimulate the chemo-nociceptors in the vessel wall or result in release of pain-triggering factors such as bradykinins. This may be correlated with the prophylactic effect of sodium bicarbonate (given as an admixture to increase the pH of the solution) against the incidence and severity of RIWM in the present meta-analysis (RR, 0.35; 95% CI, 0.20-0.60; SMD, -1.22; 95% CI, -1.46 to -0.97). This observation is in agreement with the report by Kwak et al. [7] that a NaHCO3-rocuronium admixture is one of the most effective methods of preventing RIWM (RR, 0.15; 95% CI, 0.06-0.34).
Dilution of the rocuronium also decreased the incidence and severity of RIWM (RR, 0.47; 95% CI, 0.39-0.56; SMD, -1.64; 95% CI. -2.47 to -0.81). Kim and Yoon reported that the incidence and severity of RIWM were significantly lower when rocuronium was diluted with 0.9% normal saline, although they did not report a difference in the pH or osmolality of the rocuronium solution [37]. They concluded that diluting rocuronium with 0.9% normal saline might decrease stimulation of the vessel wall. Furthermore, the increased volume of rocuronium solution due to dilution may result in a slower injection, which has been suggested to decrease the withdrawal response. This observation is in agreement with our report that a slow speed of injection decreased the incidence of RIWM (RR, 0.34; 95% CI, 0.17-0.70).
Rocuronium injection pain can be assessed by investigators through withdrawal movement in unconscious patients after induction of anesthesia. We did not include study collecting data on pain by asking conscious patients directly due to a possible ethical controversy. In 1995, Moorthy reported that a patient complained of severe burning pain after injecting a sub-paralyzing dose (10% of the intubating dose) of rocuronium before loss of consciousness after induction of general anesthesia [51], which was the first documentation of rocuronium-induced pain since first used clinically. That report recommended that rocuronium should be administered after loss of consciousness during induction of general anesthesia.
Pre-treatment application of a venous tourniquet has been used for rocuronium injection pain. The venous occlusion technique is suitable for studying the peripheral action of pretreatment drugs with local anesthetic properties, such as lidocaine, ondansetron, or tramadol, but is not useful for opioids that act centrally. A tourniquet may prevent delivery of centrally acting drugs to effect sites, which can delay the drug effect. This is correlated with results of our meta-analysisIn our study. Lidocaine showed a prophylactic effect regardless of tourniquet use, but remifenatnil in conjunction with a tourniquet did not show a prophylactic effect against the incidence of RIWM (RR, 0.46; 95% CI, 0.19-1.10). However, when using remifentanil with a tourniquet, low tendency for incidence in RIWM and significant reduction for severity in RIWM (SMD, -2.05, 95% CI, -2.66 to -1.45), may be possibly due to the short onset of action of remifentanil.
Propofol, similarly to rocuronium, is used frequently during induction of general anesthesia, and also associated with injection pain. The effect of prophylactic modalities against propofol injection-induced pain using lidocaine or opioids has been reported [52]; this is in agreement with the results of the present meta-analysis. Moreover, lidocaine or opioids can be applied to attenuate stimulation by tracheal intubation. Thus, we suggest that lidocaine and opioids should be used during induction of anesthesia due to their reduction of pain associated with injection of both propofol and rocuronium and their promotion of hemodynamic stability during tracheal intubation.
We found a substantial number of RIWM clinical trials in the Korean literature. About half of the studies since 1995 (29 of 53 articles assessed for eligibility identified in the international database) were carried out by medical staff in Korea. This may be because Korean medical staff have a greater ability to detect RIWM, or have a medical mission that aims to reduce RIWM, as Koreans may be particularly sensitive to rocuronium-injection-induced pain. The overall incidence of RIWM in this study was 80% (range, 56-100%) [27,39,42,46,49], which is higher than those of a previous meta-analysis [7] and reports [2,53,54,55]. This might be why RIWM study protocols are relatively simple compared with those used in other studies.
As a number of RIWM studies have shown that various interventions have prophylactic effects, conducting an RIWM study would not be novel or significant. It is necessary to compile the results of those studies and introduce effective intervention methods in an appropriate manner in a clinical setting. The present systematic review will contribute to reducing the number of studies of the known association with RIWM.
We should address the importance of publication bias, which may have influenced the results of the meta-analysis. The fact that studies may or may not report negative results may result in publication bias. In this study, the Begg's funnel plot for lidocaine and opioids on the incidence of RIWM suggested a bias towards publishing small-sample-size studies with findings in favor of intervention, and the results of the Egger's tests were significant (P = 0.013, P < 0.001, respectively). As a marked concentration of studies was observed on the left lower part of the plot, this asymmetry suggests a publication bias toward a larger effect size in smaller studies. It was inferred that smaller studies with larger effect sizes were more likely to be published; therefore, they had a higher probability of demonstrating statistical significance. Duval and Tweedie's trim and fill analyses were used to provide an adjusted estimate of the effects of pharmacological and non-pharmacological interventions on RIWM by considering the role of unpublished studies [56]. This nonparametric method revealed that a significant effect of lidocaine and opioids on RIWM was observed after considering publication bias (RR, 0.71; 95% CI, 0.59-0.85; RR, 0.56; 95% CI, 0.41-0.78, respectively).
The strength of the present meta-analysis lies in the large sample size (41 studies and 4,742 subjects) and application of a rigorous methodology with a sensitive search strategy to provide the first systematic review assessing the prophylactic effect of pharmacological and non-pharmacological methods against the incidence and severity of RIWM, particularly in the Korean population. Two investigators identified the articles independently, extracted the data, and verified and resolved all discrepancies. Furthermore, our findings were stable and robust in subgroup and sensitivity analyses.
However, several limitations to this meta-analysis should be noted. First, the methodological quality of the studies was generally poor. The randomization method was not stated clearly in most trials, and few trials reported blinding of assessors of outcomes. Blinding and allocation concealment were not reported in these RCTs, indicating a potential risk of bias [57,58]. These findings suggest that many Korean anesthesiologists are not familiar with or are not paying attention to the study methodology (e.g., CONSORT or Risk of Bias). This finding also suggests that researchers carry out RCTs while lacking the methodological skills to execute such studies, or failed to report the methodology used despite the fact that the clinical trials were conducted under strict RCT principles. Thus, it is essential that well-designed RCTs using a rigorous methodology should be designed, conducted, and reported. Second, we cannot be certain that our search was all-inclusive despite our effort to retrieve all relevant articles. As there is no standard search filter in domestic databases, it was difficult to conduct an efficient and complete search for this study. Therefore, a domestic search was performed using the term "rocuronium" in all possible relevant databases to increase sensitivity, and unnecessary articles were excluded manually. Third, we did not search for unpublished studies, so only published studies were included in our meta-analysis. Further, all studies included in our meta-analysis were conducted on a Korean population, and a large proportion of them were published in Korean. Therefore, there was a considerable possibility of publication bias [59,60]. Efforts were made to limit the impact of publication bias on findings. Graphical (funnel plots) and statistical (Egger's test) methods were used to assess possible publication bias and its potential impact on the findings. While the funnel plot indicated a potential for publication bias (i.e., unpublished trials with non-significant or negative findings), the analysis revealed that a significant effect remained after consideration of publication bias. This allowed us to have some confidence in our findings despite the omission of data from unpublished studies. Finally, heterogeneity was substantial in almost all intervention analyses. This may have arisen from the different characteristics of the subjects and the various methods used to prevent RIWM. Furthermore, the methodology used to measure the incidence and severity of RIWM may have increased the heterogeneity.
In conclusion, the use of lidocaine, opioids, sodium bicarbonate, hypnotics, and inhalation anesthetics were effective in terms of decreasing the incidence and severity of RIWM in a Korean population. Pretreatment and admixture administration of lidocaine, remifentanil, alfentanil, fentanyl, and sodium bicarbonate reduced the incidence and severity of RIWM except for the severity of RIWM when using lidocaine admixture and the incidence of RIWM when using fentanyl pretreatment. Lidocaine reduced the incidence and severity of RIWM regardless of venous occlusion, but remifentanil with venous occlusion did not decrease the incidence of RIWM. Dilution and slow injection of rocuronium reduced the incidence and severity of IPWM. Administration of lidocaine or opioids during induction of anesthesia is recommended unless contraindicated, as it seems to have certain advantages.
References
1. Hunter JM. Rocuronium: the newest aminosteroid neuromuscular blocking drug. Br J Anaesth 1996; 76: 481-483. PMID: 8652315.



2. Ahmad N, Choy CY, Aris EA, Balan S. Preventing the withdrawal response associated with rocuronium injection: a comparison of fentanyl with lidocaine. Anesth Analg 2005; 100: 987-990. PMID: 15781511.


3. Steegers MA, Robertson EN. Pain on injection of rocuronium bromide. Anesth Analg 1996; 83: 203PMID: 8659757.

4. Cheong KF, Wong WH. Pain on injection of rocuronium: influence of two doses of lidocaine pretreatment. Br J Anaesth 2000; 84: 106-107. PMID: 10740559.



5. Borgeat A, Kwiatkowski D. Spontaneous movements associated with rocuronium: is pain on injection the cause? Br J Anaesth 1997; 79: 382-383. PMID: 9389860.



6. Lui JT, Huang SJ, Yang CY, Hsu JC, Lui PW. Rocuronium-induced generalized spontaneous movements cause pulmonary aspiration. Chang Gung Med J 2002; 25: 617-620. PMID: 12479624.

7. Kwak HJ, Kim JY, Kim YB, Min SK, Moon BK, Kim JY. Pharmacological prevention of rocuronium-induced injection pain or withdrawal movements: a meta-analysis. J Anesth 2013; 27: 742-749. PMID: 23519582.


8. Higgins JP, Green S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0. The Cochrane Collaboration. 2011, updated March 2011.
9. Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ 2009; 339: b2700PMID: 19622552.



10. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ 2003; 327: 557-560. PMID: 12958120.



11. Jeon Y, Ha JH, Lee JE, Lee HC, Ryu T, Kwak KH. Rocuronium-induced withdrawal movement: influence of ketorolac or a combination of lidocaine and ketorolac pretreatment. Korean J Anesthesiol 2013; 64: 25-28. PMID: 23372882.



12. Kim E, Kim CH, Kim HK, Kwon JY, Lee do W, Kim HY. Effect of nitrous oxide inhalation on pain after propofol and rocuronium injection. J Anesth 2013; 27: 868-873. PMID: 23982855.


13. Choi YJ, Park HS, Lee H, Yoon SZ. Single pretreatment of remifentanil may reduce pain after propofol and rocuronium injection in rapid sequence induction. Korean J Anesthesiol 2012; 63: 413-418. PMID: 23198034.



14. Shin YH, Choi SJ, Jeong HY, Kim MH. Evaluation of dose effects of magnesium sulfate on rocuronium injection pain and hemodynamic changes by laryngoscopy and endotracheal intubation. Korean J Anesthesiol 2011; 60: 329-333. PMID: 21716962.



15. Yoon JS, Jeon HJ, Cho SS, Lee JD, Kang KO, Ryu SW, et al. Effect of pretreatment with gabapentin on withdrawal movement associated with intravenous rocuronium injection. Korean J Anesthesiol 2011; 61: 367-371. PMID: 22148083.



16. Shin YH, Kim CS, Lee JH, Sim WS, Ko JS, Cho HS, et al. Dilution and slow injection reduces the incidence of rocuronium-induced withdrawal movements in children. Korean J Anesthesiol 2011; 61: 465-469. PMID: 22220222.



17. Yoon JR, Jeon Y, Yoo Y, Shin HJ, Ahn JH, Lim CH. The analgesic effect of remifentanil on prevention of withdrawal response associated with the injection of rocuronium in children: no evidence for a peripheral action. J Int Med Res 2010; 38: 1795-1800. PMID: 21309495.


18. Kim YH, Go YK, Lee JU, Chung WS, Shin YS, Han KC, et al. Pretreatment with nafamostat mesilate, a kallikrein inhibitor, to decrease withdrawal response associated with rocuronium. J Anesth 2010; 24: 549-552. PMID: 20499255.


19. Lee SS, Yoon H. A comparison of the effect of lidocaine or sodium bicarbonate mixed with rocuronium on withdrawal movement, mean arterial pressure and heart rate during rocuronium injection. J Korean Acad Nurs 2009; 39: 270-278. PMID: 19411798.


20. Kim JH, Kim JH, Han SH, Hwang JW, Oh AY. Alfentanil is comparable to remifentanil in preventing withdrawal movement following rocuronium injection. J Clin Anesth 2009; 21: 9-12. PMID: 19232934.


21. Lee YC, Jang YH, Kim JM, Lee SG. Rapid injection of rocuronium reduces withdrawal movement on injection. J Clin Anesth 2009; 21: 427-430. PMID: 19833276.


22. Oh AY, Seo KS, Goo EK, Park YO, Kim SJ, Kim JH. Prevention of withdrawal movement associated with injection of rocuronium in children: comparison of remifentanil, alfentanil and fentanyl. Acta Anaesthesiol Scand 2007; 51: 1190-1193. PMID: 17711566.


23. Kim KS, Kim YS, Jeon WJ, Yeom JH. Prevention of withdrawal associated with the injection of rocuronium in adults and children. J Clin Anesth 2006; 18: 334-338. PMID: 16905077.


24. Park SH, Oh AY, Goo EK, Nahm FS, Min SW, Hwang JW, et al. A short period of inhalation induction with sevoflurane prevents rocuronium-induced withdrawal in children. Acta Anaesthesiol Scand 2011; 55: 87-91. PMID: 21126238.


25. Jeon Y, Baek SU, Park SS, Kim SO, Baek WY, Yeo JS. Effect of pretreatment with acetaminophen on withdrawal movements associated with injection of rocuronium: a prospective, randomized, double-blind, placebo controlled study. Korean J Anesthesiol 2010; 59: 13-16. PMID: 20651992.



26. Lee HJ, Han SJ, Kim H, Lee IO, Kong MH, Kim NS, et al. Antihistamine pretreatment to reduce incidence of withdrawal movement after rocuronium injection. J Korean Med Sci 2009; 24: 879-882. PMID: 19794987.



27. Lee JI, Lim SH, Lee SE, Kim YH, Lee JH, Lee KM, et al. Priming technique can alleviate the withdrawal responses associated with intravenous administration of rocuronium. Korean J Anesthesiol 2009; 56: 628-633.


28. Kim JY, Kwak HJ, Kim JY, Park KS, Song JS. Prevention of rocuronium-induced withdrawal movement in children: a comparison of remifentanil with alfentanil. Paediatr Anaesth 2008; 18: 245-250. PMID: 18230068.


29. Choi BI, Choi SH, Shin YS, Lee SJ, Yoon KB, Shin SK, et al. Remifentanil prevents withdrawal movements caused by intravenous injection of rocuronium. Yonsei Med J 2008; 49: 211-216. PMID: 18452256.



30. Kim JY, Kim JY, Kim YB, Kwak HJ. Pretreatment with remifentanil to prevent withdrawal after rocuronium in children. Br J Anaesth 2007; 98: 120-123. PMID: 17065169.



31. Cho K, Lee SH, Lee W, Chu BK, Kim MH, Lim SH, et al. Effect of pretreatment with palonosetron on withdrawal movement associated with rocuronium injection. Korean J Anesthesiol 2014; 66: 23-27. PMID: 24567809.



32. Jung JD, An TH, Song HS. The dose-dependent effect of remifentanil for withdrawal responses on injection of recuronium in children. Anesth Pain Med 2009; 4: 170-173.
33. Lee JY, Lee KC, Kim HS, Bang MS, Chang YJ. The dose-dependent effect of alfentanil on the rocuronium induced withdrawal in children. Anesth Pain Med 2009; 4: 348-351.
34. Kwak HJ, Kim YB, Kim EJ, Kim JY. Prevention of rocuronium-induced withdrawal movement in children: a comparison of remifentanil with lidocaine. Korean J Anesthesiol 2008; 54: 437-440.

35. Park SE, Park SH, Cho YW, Son HW, Lee JH, Kim DY. Hemodynamic Responses of Pretreatments Designed to Reduce Withdrawal Movements Following Rocuronium Injection: A Comparison of Alfentanil with Lidocaine. Korean J Anesthesiol 2008; 54: 507-512.

36. Baek SH, Woo CM, Lee HJ, Yoon JY, Kwon JY, Shin SW. Rocuronium-induced withdrawal movements associated with different Rocuronium injection method. Paediatr Anaesth 2008; 18: 515-519. PMID: 18312520.


37. Kim YH, Yoon MJ. Dilution of rocuronium with 0.9% NaCl reduces withdrawal movement during anesthetic induction. Anesth Pain Med 2007; 2: 22-25.
38. Kim YS, Lee JA. The effect of dilution with 0.9% normal saline on reducing withdrawal movement induced by rocuronium injection. Anesth Pain Med 2007; 2: 26-29.
39. Park SJ, Lee JY, Jee DL. The effect of rocuronium diluted with 0.9% NaCl on withdrawal response during injection in pediatric patients. Korean J Anesthesiol 2006; 51: 157-161.

40. Woo CM, Baek SH, Kim CH, Kwon JY, Kim HK, Shin SW. Comparison of withdrawal movement and intubating condition by the methods of rocuronium injection. Korean J Anesthesiol 2006; 51: 663-668.

41. Choi HG, Kim DS, Chang TH, Kim SH, Kim KH, Ryu SJ. Appropriate dosage of 8.4% sodium bicarbonate for preventing injection pain of rocuronium during anesthetic induction. Korean J Anesthesiol 2006; 51: 162-166.

42. Jung SM, Kim SH, Lim YS, Kwon HU, Kang PS, Park CW. The effect of sodium bicarbonate or lidocaine mixed with rocuronium on withdrawal movement in pediatric patient. Korean J Anesthesiol 2005; 48: 514-519.

43. Park JT, Choi JC, Yoo YS, Lee YB, Kim SY, Lim HK. The effect of pretreatment with thiopental on reducing pain induced by rocuronium injection. Yonsei Med J 2005; 46: 765-768. PMID: 16385651.



44. Jung JD, An TH, Yang IH, Wee HW, Eun SS, Han SY. The effect of pretreatment with lidocaine on withdrawal movement associated with injection of rocuronium in pediatric patients. Korean J Pediatr Anesth 2005; 9: 31-35.
45. Lee YK, Choi WJ, Kang WC, Leem JG, Yang HS. The effects of lidocaine on pain due to rocuronium. Korean J Anesthesiol 2004; 46: 145-150.

46. Chang SH, Kim HY, Park JY, Lee HW, Lim HJ, Yoon SM. The preventive effect of lidocaine on the withdrawal associated with the injection of rocuronium in children. Korean J Anesthesiol 2004; 46: 665-669.

47. Hwang SM, Oh MS, Lim SY. Effect of sodium bicarbonate or lidocaine mixed with rocuronium on withdrawal movement during rocuronium injection. Korean J Anesthesiol 2004; 46: 160-163.

48. Cheong SH, Kim SY, Lee KM, Choe YK, Kim YJ, Park JW, et al. The effect of pretreatment with ketamine on vascular pain due to an intravenous rocuronium injection in pediatric patients. Korean J Anesthesiol 2003; 45: 447-450.

49. Kim KS, Chae YJ, Pak SY, Cho HB, Kim JS. The effect of pretreatment with lidocaine for the withdrawal movement associated with the injection of rocuronium in children. Korean J Anesthesiol 2002; 43: 572-574.

50. Lockey D, Coleman P. Pain during injection of rocuronium bromide. Anaesthesia 1995; 50: 474PMID: 7793569.

51. Moorthy SS, Dierdorf SF. Pain on injection of rocuronium bromide. Anesth Analg 1995; 80: 1067PMID: 7726421.

52. Jalota L, Kalira V, George E, Shi YY, Hornuss C, Radke O, et al. Prevention of pain on injection of propofol: systematic review and meta-analysis. BMJ 2011; 342: d1110PMID: 21406529.


53. Akkaya T, Toygar P, Bedirli N, Yazicioglu D, Gumus H. Effects of pretreatment with lidocaine or ketamine on injection pain and withdrawal movements of rocuronium. Turk J Med Sci 2008; 38: 577-582.
54. Asida SM, Megalla SA, Korany A. Effect of fentanyl, lidocaine or, ketorolac on the prevention of pain during rocuronium injection. Egyptian Journal of Anaesthesia 2009; 25: 31-40.
55. Yavascaoglu B, Kaya FN, Ozcan B. Esmolol pretreatment reduces the frequency and severity of pain on injection of rocuronium. J Clin Anesth 2007; 19: 413-417. PMID: 17967668.


56. Duval S, Tweedie R. Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 2000; 56: 455-463. PMID: 10877304.


57. Wood L, Egger M, Gluud LL, Schulz KF, Juni P, Altman DG, et al. Empirical evidence of bias in treatment effect estimates in controlled trials with different interventions and outcomes: meta-epidemiological study. BMJ 2008; 336: 601-605. PMID: 18316340.



58. He J, Du L, Liu G, Fu J, He X, Yu J, et al. Quality assessment of reporting of randomization, allocation concealment, and blinding in traditional Chinese medicine RCTs: a review of 3159 RCTs identified from 260 systematic reviews. Trials 2011; 12: 122PMID: 21569452.




59. Williamson PR, Gamble C, Altman DG, Hutton JL. Outcome selection bias in meta-analysis. Stat Methods Med Res 2005; 14: 515-524. PMID: 16248351.


60. Jüni P, Holenstein F, Sterne J, Bartlett C, Egger M. Direction and impact of language bias in meta-analyses of controlled trials: empirical study. Int J Epidemiol 2002; 31: 115-123. PMID: 11914306.



Fig. 1
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow chart showing the numbers of abstracts and articles identified and evaluated during the review process.
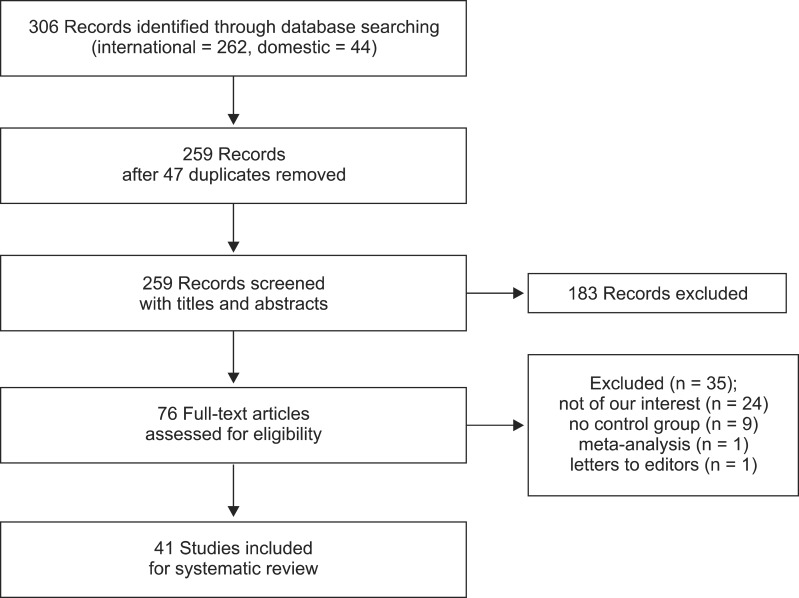
Fig. 2
Risks of bias summary table: review author's judgments of each risk of bias item for each of the studies included.

Fig. 3
Forest plot showing the overall effect of lidocaine pretreatment on the incidence of rocuronium-induced withdrawal movement (RIWM). Subgroup analyses according to methods of lidocaine administration: pretreatment vs. admixture. M-H: Mantel-Haenszel.
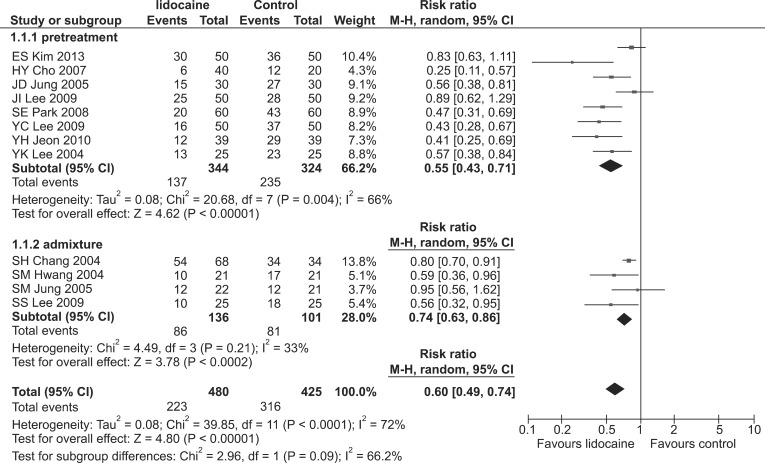
Fig. 4
Forest plot showing an overall effect of opioid pretreatment on the incidence of rocuronium-induced withdrawal movement (RIWM). Subgroup analyses according to kind of opioid: fentanyl vs. remifentanil vs. alfentanil. M-H: Mantel-Haenszel.
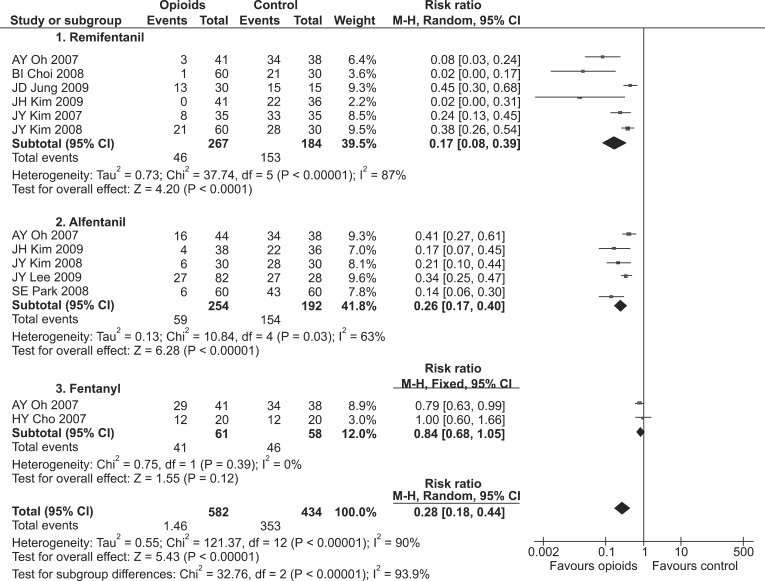
Fig. 5
Funnel plot of the comparisons: opioid vs. control, outcome: reduction in rocuronium-induced withdrawal movement (RIWM). White circles: comparisons included. Black circles: inputted comparisons using the trim-and-fill method. White diamond: pooled observed log risk ratio. Black diamond: pooled inputted log risk ratio.
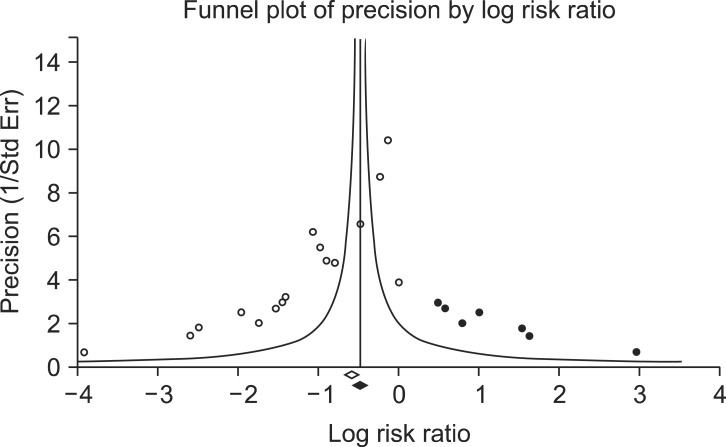
Fig. 6
Funnel plot of the comparisons: lidocaine vs. control, outcome: reduction in rocuronium-induced withdrawal movement (RIWM). White circles: comparisons included. Black circles: inputted comparisons using the trim-and-fill method. White diamond: pooled observed log risk ratio. Black diamond: pooled inputted log risk ratio.
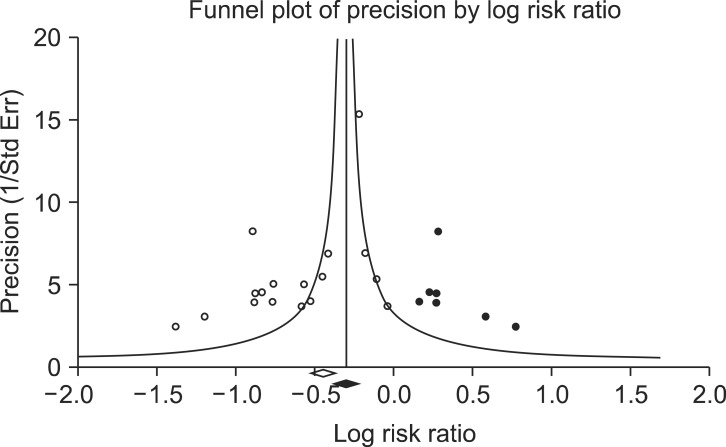
Table 1
Characteristics of Studies of the Efficacy of Pharmacological or Non-pharmacological Interventions in Terms of Reducing or Preventingrocuronium-induced withdrawal Movement
- TOOLS