1. Hamilton MA, Cecconi M, Rhodes A. A systematic review and meta-analysis on the use of preemptive hemodynamic intervention to improve postoperative outcomes in moderate and high-risk surgical patients. Anesth Analg 2011; 112: 1392-402.


3. Ameloot K, Palmers PJ, Malbrain ML. The accuracy of noninvasive cardiac output and pressure measurements with finger cuff: a concise review. Curr Opin Crit Care 2015; 21: 232-9.


4. Bubenek-Turconi SI, Craciun M, Miclea I, Perel A. Noninvasive continuous cardiac output by the Nexfin before and after preload-modifying maneuvers: a comparison with intermittent thermodilution cardiac output. Anesth Analg 2013; 117: 366-72.


5. Heusdens JF, Lof S, Pennekamp CW, Specken-Welleweerd JC, de Borst GJ, van Klei WA, et al. Validation of non-invasive arterial pressure monitoring during carotid endarterectomy. Br J Anaesth 2016; 117: 316-23.


6. Fischer MO, Avram R, Cârjaliu I, Massetti M, Gérard JL, Hanouz JL, et al. Non-invasive continuous arterial pressure and cardiac index monitoring with Nexfin after cardiac surgery. Br J Anaesth 2012; 109: 514-21.


7. Critchley LA, Lee A, Ho AM. A critical review of the ability of continuous cardiac output monitors to measure trends in cardiac output. Anesth Analg 2010; 111: 1180-92.


8. Sangkum L, Liu GL, Yu L, Yan H, Kaye AD, Liu H. Minimally invasive or noninvasive cardiac output measurement: an update. J Anesth 2016; 30: 461-80.


9. Westerhof N, Lankhaar JW, Westerhof BE. The arterial Windkessel. Med Biol Eng Comput 2009; 47: 131-41.


10. Broch O, Renner J, Gruenewald M, Meybohm P, Schöttler J, Caliebe A, et al. A comparison of the Nexfin® and transcardiopulmonary thermodilution to estimate cardiac output during coronary artery surgery. Anaesthesia 2012; 67: 377-83.


11. Suzuki T, Suzuki Y, Okuda J, Minoshima R, Misonoo Y, Ueda T, et al. Cardiac output and stroke volume variation measured by the pulse wave transit time method: a comparison with an arterial pressure-based cardiac output system. J Clin Monit Comput 2019; 33: 385-92.


12. Boisson M, Poignard ME, Pontier B, Mimoz O, Debaene B, Frasca D. Cardiac output monitoring with thermodilution pulse-contour analysis vs. non-invasive pulse-contour analysis. Anaesthesia 2019; 74: 735-40.


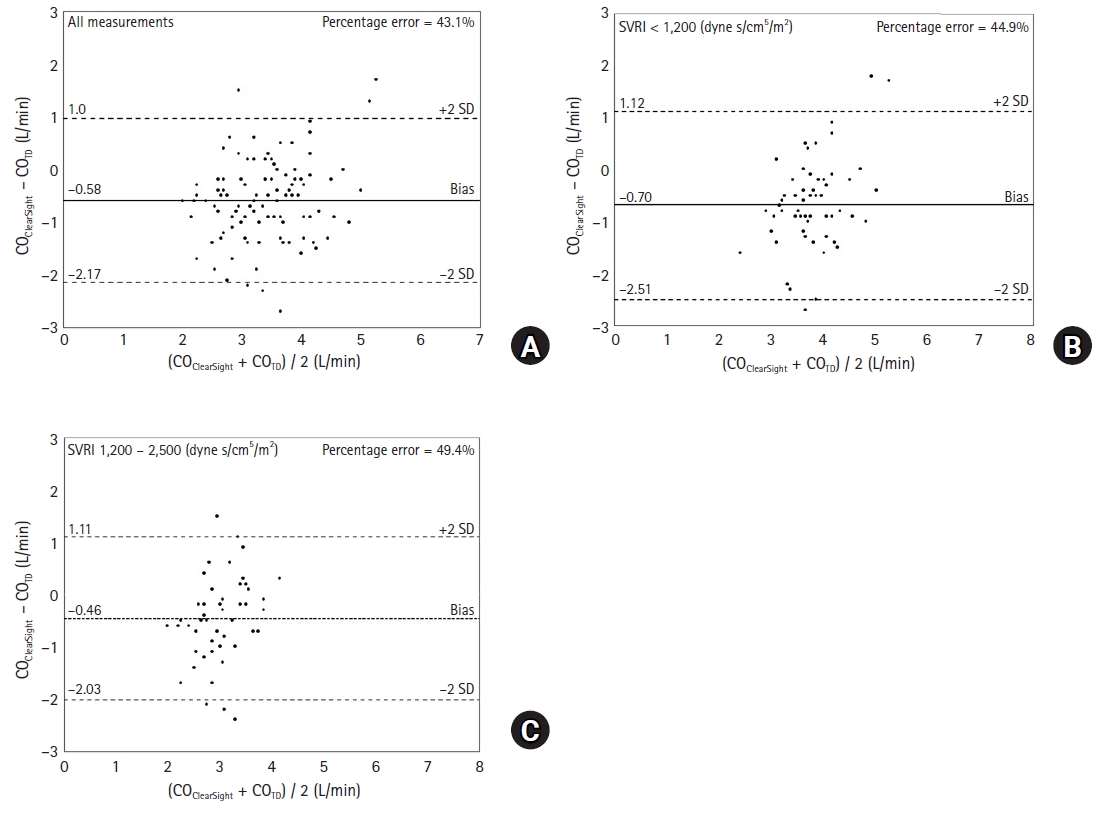
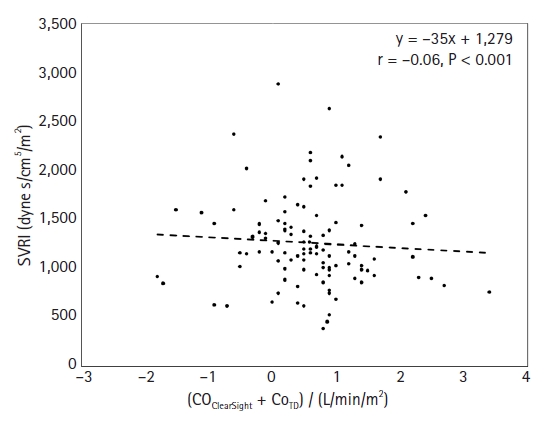
13. Mukai A, Suehiro K, Kimura A, Tanaka K, Yamada T, Mori T, et al. Effect of systemic vascular resistance on the reliability of noninvasive hemodynamic monitoring in cardiac surgery. J Cardiothorac Vasc Anesth 2021; 35: 1782-91.


14. Mantha S, Roizen MF, Fleisher LA, Thisted R, Foss J. Comparing methods of clinical measurement: reporting standards for bland and altman analysis. Anesth Analg 2000; 90: 593-602.


15. Bland JM, Altman DG. Agreement between methods of measurement with multiple observations per individual. J Biopharm Stat 2007; 17: 571-82.


16. Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986; 1: 307-10.


17. Critchley LA, Critchley JA. A meta-analysis of studies using bias and precision statistics to compare cardiac output measurement techniques. J Clin Monit Comput 1999; 15: 85-91.

18. Critchley LA, Yang XX, Lee A. Assessment of trending ability of cardiac output monitors by polar plot methodology. J Cardiothorac Vasc Anesth 2011; 25: 536-46.


19. Peyton PJ, Chong SW. Minimally invasive measurement of cardiac output during surgery and critical care: a meta-analysis of accuracy and precision. Anesthesiology 2010; 113: 1220-35.


20. Kanda Y. Investigation of the freely available easy-to-use software 'EZR' for medical statistics. Bone Marrow Transplant 2013; 48: 452-8.


21. Sumiyoshi M, Maeda T, Miyazaki E, Hotta N, Sato H, Hamaguchi E, et al. Accuracy of the ClearSight™ system in patients undergoing abdominal aortic aneurysm surgery. J Anesth 2019; 33: 364-71.


22. Kanazawa H, Maeda T, Miyazaki E, Hotta N, Ito S, Ohnishi Y. Accuracy and trending ability of blood pressure and cardiac index measured by ClearSight system in patients with reduced ejection fraction. J Cardiothorac Vasc Anesth 2020; 34: 3293-9.


23. Grube E, Sinning JM. The "Big Five” complications after transcatheter aortic valve replacement: do we still have to be afraid of them? JACC Cardiovasc Interv 2019; 12: 370-2.


24. Schraverus P, Kuijpers MM, Coumou J, Boly CA, Boer C, van Kralingen S. Level of agreement between cardiac output measurements using Nexfin® and thermodilution in morbidly obese patients undergoing laparoscopic surgery. Anaesthesia 2016; 71: 1449-55.


25. Ostergaard M, Nielsen J, Rasmussen JP, Berthelsen PG. Cardiac output--pulse contour analysis vs. pulmonary artery thermodilution. Acta Anaesthesiol Scand 2006; 50: 1044-9.


26. Bartels SA, Stok WJ, Bezemer R, Boksem RJ, van Goudoever J, Cherpanath TG, et al. Noninvasive cardiac output monitoring during exercise testing: nexfin pulse contour analysis compared to an inert gas rebreathing method and respired gas analysis. J Clin Monit Comput 2011; 25: 315-21.


27. Bogert LW, Wesseling KH, Schraa O, van Lieshout EJ, de Mol BA, van Goudoever J, et al. Pulse contour cardiac output derived from non-invasive arterial pressure in cardiovascular disease. Anaesthesia 2010; 65: 1119-25.